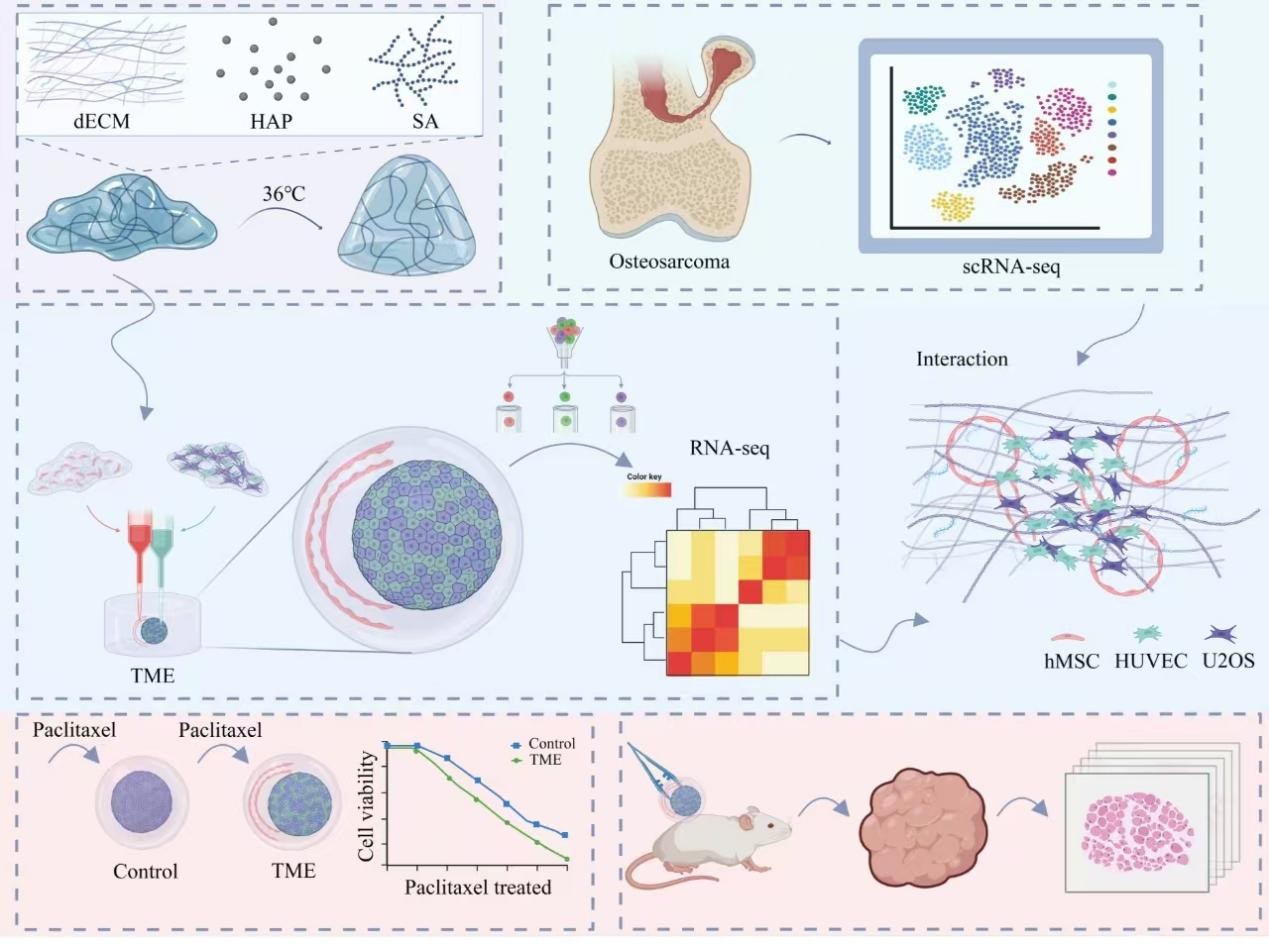
A vascularized 3D-bioprinted model of the osteosarcoma microenvironment reveals a proliferation-to-invasion switch that confers chemoresistance

The tumor microenvironment (TME) is a major driver of osteosarcoma progression, metastasis, and therapeutic resistance, yet conventional models fail to effectively recapitulate the multicellular interactions and biomechanical cues of the bone niche. In this study, we developed a vascularized 3D-bioprinted osteosarcoma model using a biomimetic hydrogel composed of decellularized extracellular matrix, chondroitin sulfate, and hydroxyapatite. U2OS osteosarcoma cells, human mesenchymal stem cells (HMSCs), and human umbilical vein endothelial cells were co-cultured within the printed constructs. Confocal imaging, RNA sequencing, xenograft validation, and integration with clinical single-cell RNA-sequencing data were used to define TME-driven changes in tumor behavior. The engineered TME induced a proliferation-to-invasion switch characterized by G0/G1 arrest, reduced proliferation activity, and enhanced invasiveness, angiogenic potential, and extracellular-matrix remodeling. Transcriptomic profiling showed downregulation of cell-cycle programs, including E2F and MYC targets, together with activation of TGF-β signaling, epithelial–mesenchymal transition (EMT), and hypoxia-related pathways. Mechanistically, HMSCs were identified as key regulatory cells that promoted migratory programs in tumor cells, partly through CXCL chemokine signaling engaging CXCR2. The TME-induced state also conferred robust chemoresistance to paclitaxel, associated with cell-cycle quiescence and pro-survival signaling, such as NF-κB activation. Critically, the transcriptional signature of these quiescent-invasive cells closely mirrored that of a clinically observed osteosarcoma subpopulation with low proliferation and high EMT activity. This high-fidelity vascularized 3D-bioprinted model recapitulates major TME-dependent features of osteosarcoma and provides a biologically relevant platform for studying tumor plasticity and evaluating therapies targeting microenvironment-driven treatment resistance.
- Mirabello L, Troisi RJ, Savage SA. Osteosarcoma incidence and survival rates from 1973 to 2004: data from the Surveillance, Epidemiology, and End Results Program. Cancer. 2009;115(7):1531–43. doi: 10.1002/cncr.24121
- Xu G, Wu H, Zhang Y, et al. Risk and Prognostic Factors for Different Organ Metastasis in Primary Osteosarcoma: A Large Population-Based Analysis. Orthop Surg. 2022;14(4):714–719.doi: 10.1111/os.13243
- Zou Y, Kang J, Zhu S, et al. The osteosarcoma immune microenvironment in progression: PLEK as a prognostic biomarker and therapeutic target. Front Immunol. 2025;16:1651858. doi: 10.3389/fimmu.2025.1651858
- Rodrigues J, Sarmento B, Pereira CL. Osteosarcoma tumor microenvironment: the key for the successful development of biologically relevant 3D in vitro models. Vitro Model. 2022;1(1):5–27. doi: 10.1007/s44164-022-00008-x
- Zhu Y, Chen J, Chen C, et al. Deciphering mechanical cues in the microenvironment: from non-malignant settings to tumor progression. Biomark Res. 2025;13(1):11. doi: 10.1186/s40364-025-00727-9
- Biray Avci C, Goker Bagca B, Nikanfar M, Takanlou LS, Takanlou MS, Nourazarian A. Tumor microenvironment and cancer metastasis: molecular mechanisms and therapeutic implications. Front Pharmacol. 2024;15:1442888. doi: 10.3389/fphar.2024.1442888
- Mak IW, Evaniew N, Ghert M. Lost in translation: animal models and clinical trials in cancer treatment. Am J Transl Res. 2014;6(2):114–8.
- Andreu-Sanz D, Gregor L, Carlini E, Scarcella D, Marr C, Kobold S. Predictive value of preclinical models for CAR-T cell therapy clinical trials: a systematic review and meta-analysis. J ImmunoTherapy Cancer. 2025;13(6):e011698. doi: 10.1136/jitc-2025-011698
- Munoz-Garcia J, Jubelin C, Loussouarn A, et al. In vitro three-dimensional cell cultures for bone sarcomas. J Bone Oncol. 2021;30:100379. doi: 10.1016/j.jbo.2021.100379
- Sandhu V, Bakkalci D, Wei S, Cheema U. Enhanced Biomimetics of Three-Dimensional Osteosarcoma Models: A Scoping Review. Cancers. 2023;16(1):164. doi: 10.3390/cancers16010164
- Kortam S, Lu Z, Zreiqat H. Recent advances in drug delivery systems for osteosarcoma therapy and bone regeneration. Commun Mater. 2024;5(1):168. doi: 10.1038/s43246-024-00612-2
- Mandeda Madaiah P, Ghosh RN, Namboothiri PK, Peter M. Advancement in Scaffold-Based 3D Cell Culture Models for Osteosarcoma Drug Screening. ACS Biomater Sci Eng. 2025;11(11):6426–6442. doi: 10.1021/acsbiomaterials.5c01174
- Yu L, Zhang J, Li Y. Effects of microenvironment in osteosarcoma on chemoresistance and the promise of immunotherapy as an osteosarcoma therapeutic modality. Front Immunol. 2022;13:871076. doi: 10.3389/fimmu.2022.871076
- Tu B, Zhu J, Liu S, et al. Mesenchymal stem cells promote osteosarcoma cell survival and drug resistance through activation of STAT3. Oncotarget. 2016;7(30):48296–48308. doi: 10.18632/oncotarget.10219
- Zhou B, Sun M, Yang M, Cui W, Yang H. Construction and application of multicellular tumor microenvironment models based on three-dimensional bioprinting technology. Hepatobiliary Surg Nutr. 2025;14(4):692–696. doi: 10.21037/hbsn-2025-504
- Son J, Li S, Jeong W. Bioprinting Vascularized Constructs for Clinical Relevance: Engineering Hydrogel Systems for Biological Maturity. Gels. 2025;11(8):636. doi: 10.3390/gels11080636
- Du S, Yang J, Li H, Men S, Xu D, Wang H. Three-dimensional culture of tumor cells (Review). Oncol Lett. 2026;31(3):110. doi: 10.3892/ol.2026.15464
- Domingues MF, Carreira MC, Santos MS, et al. A 3D Bioprinted Spheroid-Laden dECM-Enriched Osteosarcoma Model for Enhanced Drug Testing and Therapeutic Discovery. Adv Healthc Mater. 2026:e03633. doi: 10.1002/adhm.202503633
- Tharakan S, Raja I, Pietraru A, et al. The Use of Hydrogels for the Treatment of Bone Osteosarcoma via Localized Drug- Delivery and Tissue Regeneration: A Narrative Review. Gels. 2023;9(4):274. doi: 10.3390/gels9040274
- Leonteva A, Kazakova A, Berezutskaya E, et al. Heterotypic 3D Model of Breast Cancer Based on Tumor, Stromal and Endothelial Cells: Cytokines Interaction in the Tumor Microenvironment. Cells. 2026;15(2):145. doi: 10.3390/cells15020145
- Cristini N, Tavakoli M, Sanati M, Yavari SA. Exploring bone-tumor interactions through 3D in vitro models: Implications for primary and metastatic cancers. J Bone Oncol. 2025;53:100698. doi: 10.1016/j.jbo.2025.100698
- Chen J, Yang H. Convergent bioprinting and microfluidics: toward next-generation biomimetic tumor models. Hepatobiliary Surg Nutr. 2026;15(1):18. doi: 10.21037/hbsn-2026-1-0021
- Barajaa MA, Otsuka T, Ghosh D, Kan H-M, Laurencin CT. Development of porcine skeletal muscle extracellular matrix–derived hydrogels with improved properties and low immunogenicity. Proc Natl Acad Sci USA 2024;121(19):e2322822121. doi: 10.1073/pnas.2322822121
- Li C, An N, Song Q, et al. Enhancing organoid culture: harnessing the potential of decellularized extracellular matrix hydrogels for mimicking microenvironments. J Biomed Sci. 27 2024;31(1):96. doi: 10.1186/s12929-024-01086-7
- Klara J, Marczak A, Łatkiewicz A, Horak W, Lewandowska-Łańcucka J. Lysine-functionalized chondroitin sulfate improves the biological properties of collagen/chitosan-based injectable hydrogels. Int J Biol Macromol. 2022;202:318–331. doi: 10.1016/j.ijbiomac.2022.01.026
- Fontana F, Marzagalli M, Sommariva M, Gagliano N, Limonta P. In Vitro 3D Cultures to Model the Tumor Microenvironment. Cancers. 2021;13(12):2970. doi: 10.3390/cancers13122970
- Loessner D, Stok KS, Lutolf MP, Hutmacher DW, Clements JA, Rizzi SC. Bioengineered 3D platform to explore cell– ECM interactions and drug resistance of epithelial ovarian cancer cells. Biomaterials. 2010;31(32):8494–8506. doi: 10.1016/j.biomaterials.2010.07.064
- Friedrich J, Ebner R, Kunz-Schughart LA. Experimental anti-tumor therapy in 3-D: spheroids--old hat or new challenge? Int J Radiat Biol. 2007;83(11-12):849–871. doi: 10.1080/09553000701727531
- Hendrikse CSE, Theelen PMM, van der Ploeg P, et al. The potential of RAS/RAF/MEK/ERK (MAPK) signaling pathway inhibitors in ovarian cancer: A systematic review and meta-analysis. Gynecol Oncol. 2023;171:83–94. doi: 10.1016/j.ygyno.2023.01.038
- Sahai E, Olson MF, Marshall CJ. Cross-talk between Ras and Rho signalling pathways in transformation favours proliferation and increased motility. Embo J. 2001;20(4):755– 766. doi: 10.1093/emboj/20.4.755
- Witze ES, Litman ES, Argast GM, Moon RT, Ahn NG. Wnt5a control of cell polarity and directional movement by polarized redistribution of adhesion receptors. Science. 2008;320(5874):365–369. doi: 10.1126/science.1151250
- Weeraratna AT, Jiang Y, Hostetter G, et al. Wnt5a signaling directly affects cell motility and invasion of metastatic melanoma. Cancer Cell. 2002;1(3):279–288. doi: 10.1016/s1535-6108(02)00045-4
- Bahar ME, Kim HJ, Kim DR. Targeting the RAS/RAF/ MAPK pathway for cancer therapy: from mechanism to clinical studies. Signal Transduct Target Ther. 2023;8(1):455. doi: 10.1038/s41392-023-01705-z
- Braicu C, Buse M, Busuioc C, et al. A Comprehensive Review on MAPK: A Promising Therapeutic Target in Cancer. Cancers. 2019;11(10):1618. doi: 10.3390/cancers11101618
- Zhang T, Ma C, Zhang Z, Zhang H, Hu H. NF-κB signaling in inflammation and cancer. MedComm. 2021;2(4):618–653. doi: 10.1002/mco2.104
- Zhang W, Wang H, Sun M, et al. CXCL5/CXCR2 axis in tumor microenvironment as potential diagnostic biomarker and therapeutic target. Cancer Commun. 2020;40(2-3):69– 80. doi: 10.1002/cac2.12010
- Han ZJ, Li YB, Yang LX, Cheng HJ, Liu X, Chen H. Roles of the CXCL8-CXCR1/2 Axis in the Tumor Microenvironment and Immunotherapy. Molecules. 2021;27(1):137. doi: 10.3390/molecules27010137
- Miyake M, Goodison S, Urquidi V, Gomes Giacoia E, Rosser CJ. Expression of CXCL1 in human endothelial cells induces angiogenesis through the CXCR2 receptor and the ERK1/2 and EGF pathways. Lab Investig. 2013;93(7):768–778. doi: 10.1038/labinvest.2013.71
- Zhou Y, Yang D, Yang Q, et al. Single-cell RNA landscape of intratumoral heterogeneity and immunosuppressive microenvironment in advanced osteosarcoma. Nat Commun. 2020;11(1):6322. doi: 10.1038/s41467-020-20059-6
- Odri GA, Tchicaya-Bouanga J, Yoon DJY, Modrowski D. Metastatic Progression of Osteosarcomas: A Review of Current Knowledge of Environmental versus Oncogenic Drivers. Cancers. 2022;14(2):360. doi: 10.3390/cancers14020360
- Patrașcu AV, Țarcă E, Lozneanu L, et al. The Role of Epithelial- Mesenchymal Transition in Osteosarcoma Progression: From Biology to Therapy. Diagnostics. 2025;15(5):644. doi: 10.3390/diagnostics15050644
- Syga S, Jain HP, Krellner M, Hatzikirou H, Deutsch A. Evolution of phenotypic plasticity leads to tumor heterogeneity with implications for therapy. PLoS Comput Biol. 2024;20(8):e1012003. doi: 10.1371/journal.pcbi.1012003
- Böttger K, Hatzikirou H, Voss-Böhme A, Cavalcanti-Adam EA, Herrero MA, Deutsch A. An Emerging Allee Effect Is Critical for Tumor Initiation and Persistence. PLoS Comput Biol. 2015;11(9):e1004366. doi: 10.1371/journal.pcbi.1004366
- Xue J, Yang XR, Wang L. NF-κB signaling pathway in osteosarcoma: from signaling networks to targeted therapy. Front Oncol. 2025;15:1565760. doi: 10.3389/fonc.2025.1565760
- Liu G, An L, Zhang H, Du P, Sheng Y. Activation of CXCL6/ CXCR1/2 Axis Promotes the Growth and Metastasis of Osteosarcoma Cells in vitro and in vivo. Front Pharmacol. 2019;10:307. doi: 10.3389/fphar.2019.00307
- Matsuoka T, Yashiro M. Bioinformatics Analysis and Validation of Potential Markers Associated with Prediction and Prognosis of Gastric Cancer. Int J Mol Sci. 2024;25(11):5880. doi: 10.3390/ijms25115880
- Hu J, Lazar AJ, Ingram D, et al. Cell membrane-anchored and tumor-targeted IL-12 T-cell therapy destroys cancer-associated fibroblasts and disrupts extracellular matrix in heterogenous osteosarcoma xenograft models. J ImmunoTherapy Cancer. 2024;12(1):e006991. doi: 10.1136/jitc-2023-006991
- Li X, Yang Y, Zhang B, et al. Lactate metabolism in human health and disease. Signal Transduct Target Ther. 2022;7(1):305. doi: 10.1038/s41392-022-01151-3
- Russo M, Chen M, Mariella E, et al. Cancer drug-tolerant persister cells: from biological questions to clinical opportunities. Nat Rev Cancer. 2024;24(10):694–717. doi: 10.1038/s41568-024-00737-z
- Wang L, Shang Z, Zhou Y, et al. Autophagy mediates glucose starvation-induced glioblastoma cell quiescence and chemoresistance through coordinating cell metabolism, cell cycle, and survival. Cell Death Dis. 2018;9(2):213. doi: 10.1038/s41419-017-0242-x
- Wang Y, Chen W, Wang Z, et al. Deciphering metabolic reprogramming of immune cells within the tumor microenvironment. J Transl Med. 2025;23(1):1055. doi: 10.1186/s12967-025-07069-y
- Rubahamya B, Dong S, Thurber GM. Clinical translation of antibody drug conjugate dosing in solid tumors from preclinical mouse data. Sci Adv. 2024;10(22):eadk1894. doi: 10.1126/sciadv.adk1894
- Lee H, Kim B, Park J, et al. Cancer stem cells: landscape, challenges and emerging therapeutic innovations. Signal Transduct Target Ther. 2025;10(1):248. doi: 10.1038/s41392-025-02360-2
- Ozmen F, Ozmen TY, Ors A, et al. Single-cell RNA sequencing reveals different cellular states in malignant cells and the tumor microenvironment in primary and metastatic ER-positive breast cancer. npj Breast Cancer. 2025;11(1):95. doi: 10.1038/s41523-025-00808-w
- Cao K, Shi Y, Wu B, et al. Single cell RNA sequencing decodes cellular heterogeneity and identifies prognostic immune signatures in bladder cancer microenvironment. Discov Oncol. 2025;16(1):2239. doi: 10.1007/s12672-025-03878-1
- Chuprin J, Buettner H, Seedhom MO, et al. Humanized mouse models for immuno-oncology research. Nat Rev Clin Oncol. 2023;20(3):192–206. doi: 10.1038/s41571-022-00721-2
- Zheng D. Orthotopic tumours, a hot topic for xenograft models? eBioMedicine. 2019;41:11–12. doi: 10.1016/j.ebiom.2019.02.052
- Nabel CS, Vander Heiden MG. Patient-Derived Xenografts to Study Cancer Metabolism: When Does X Mark the Spot? Cancer Res. 2021;81(17):4399–4401. doi: 10.1158/0008-5472.Can-21-0770
- Peng M, Zhu Y, Hu Y, Wen J, Huang W. Advances in the regulation of macrophage polarization by the tumor microenvironment. Discov Oncol. 2025;16(1):1487. doi: 10.1007/s12672-025-03258-9
- Picco G, Garnett MJ. A Road Map for Precision Cancer Medicine Using Personalized Models. Cancer Discov. 2017;7(5):456–458. doi: 10.1158/2159-8290.CD-17-0268
- Yousefieh N, Hahto SM, Stephens AL, Ciavarra RP. Regulated expression of CCL21 in the prostate tumor microenvironment inhibits tumor growth and metastasis in an orthotopic model of prostate cancer. Cancer Microenviron. 2009;2(1):59–67. doi: 10.1007/s12307-009-0021-z

