A rational framework for 3D bioprinting of organoids: From assembly mechanisms and material properties to functional outcomes

Organoids hold great promise for modeling development, disease, and patient-specific therapy, but their translation is limited by poor vascularization, lack of immune and neural components, low reproducibility, and difficulties in scale-up. 3D bioprinting can address these bottlenecks by enabling digitally guided, spatially precise deposition of bioinks to construct multiscale architectures and perfusable channel networks, yet matching printing principles with material systems remains a central challenge. Unlike existing reviews that focus on isolated technologies or materials, this review introduces a cohesive paradigm that systematically links assembly mechanisms, material properties, and organoid functions along a mechanism–material–function axis. Its core breakthrough shifts the field from empirical guesswork to on-demand engineered design. We analyzed physicochemical mechanisms, such as ionic crosslinking, hydrophobic interactions, dynamic covalent bonding, and photoinitiated polymerization, and mapped them to tunable metrics, including modulus, degradation kinetics, mass-transport capacity, and bioactive delivery. Based on this mapping, we developed a full-chain decision framework for technology selection, material design, and process-parameter optimization, and proposed a predictive, reproducible formulation strategy. By transforming organoid fabrication from an empirical practice into an engineering discipline, this framework enables predictable and scalable organoid fabrication for regenerative medicine, drug discovery, and disease modeling applications through standardized process-parameter optimization and material recipe design.
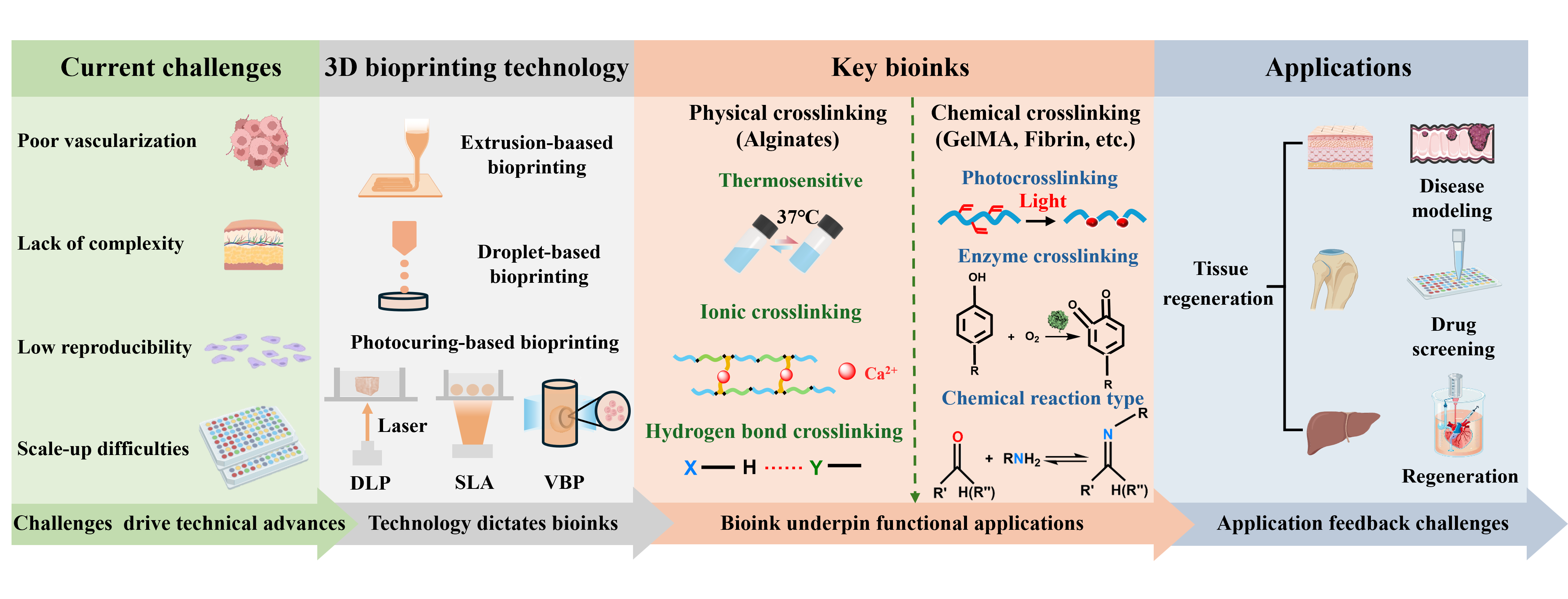
- Zhu Z, Cheng Y, Liu X, et al. Advances in the Development and Application of Human Organoids: Techniques, Applications, and Future Perspectives. Cell Transplant. 2025;34:1–20. doi: 10.1177/09636897241303271
- Septiana WL, Pawitan JA. Potential Use of Organoids in Regenerative Medicine. Tissue Eng Regener Med. 2024;21(8):1125–1139. doi: 10.1007/s13770-024-00672-y
- Wolff L, Hendrix S. Rethinking Matrigel: The Complex Journey to Matrix Alternatives in Organoid Culture. Adv Sci. 2025;12(47):e08734. doi: 10.1002/advs.202508734
- Musah S, Arzaghi H. Unleashing the power of biomaterials to enhance organoid differentiation and function. Nat Methods. 2024;21(9):1575–1577. doi: 10.1038/s41592-024-02393-5
- Low LA, Mummery C, Berridge BR, Austin CP, Tagle DA. Organs-on-chips: into the next decade. Nat Rev Drug Discovery. 2021;20(5):345–361. doi: 10.1038/s41573-020-0079-3
- Chang M, Bogacheva MS, Lou YR. Challenges for the Applications of Human Pluripotent Stem Cell-Derived Liver Organoids. Front Cell Dev Biol. 2021;9:748576. doi: 10.3389/fcell.2021.748576
- Andrews MG, Kriegstein AR. Challenges of Organoid Research. Annu Rev Neurosci. 2022;45:23–39. doi: 10.1146/annurev-neuro-111020-090812
- Shrestha S, Lekkala VKR, Acharya P, Kang S-Y, Vanga MG, Lee M-Y. Reproducible generation of human liver organoids (HLOs) on a pillar plate platform via microarray 3D bioprinting. Lab Chip. 2024;24(10):2747–2761. doi: 10.1039/d4lc00149d
- Zhang S, Wan Z, Kamm RD. Vascularized organoids on a chip: strategies for engineering organoids with functional vasculature. Lab Chip. 2021;21(3):473–488. doi: 10.1039/D0LC01186J
- Nam S-W, Lee H, Jeon D-G, Son M-Y. 3D Organoid Culturing Devices to Induce in Vitro Models of Human Intestinal Inflammation. Electron Mater Lett. 2025;21(4):504–512. doi: 10.1007/s13391-025-00561-z
- Gao D, Li R, Pan J, et al. 3D bioprinting bone/cartilage organoids: construction, applications, and challenges. J Orthop Translat. 2025;55:75–93. doi: 10.1016/j.jot.2025.08.008
- Layrolle P, Payoux P, Chavanas S. Message in a Scaffold: Natural Biomaterials for Three-Dimensional (3D) Bioprinting of Human Brain Organoids. Biomolecules. 2023;13(1):25. doi: 10.3390/biom13010025
- Leung MC, Laksman Z. 3D Bioprinting Functional Engineered Heart Tissues. Int J Mol Sci. 2025;26(21):10707. doi: 10.3390/ijms262110707
- D’Antoni C, Mautone L, Sanchini C, et al. Unlocking Neural Function with 3D In Vitro Models: A Technical Review of Self-Assembled, Guided, and Bioprinted Brain Organoids and Their Applications in the Study of Neurodevelopmental and Neurodegenerative Disorders. Int J Mol Sci. 2023;24(13):10762.
doi: 10.3390/ijms241310762
- Chen C, Rengarajan V, Kjar A, Huang Y. A matrigel-free method to generate matured human cerebral organoids using 3D-Printed microwell arrays. Bioact Mater. 2021;6(4):1130– 1139. doi: 10.1016/j.bioactmat.2020.10.003
- Kim E, Jeong E, Hong YM, et al. Magnetically reshapable 3D multi-electrode arrays of liquid metals for electrophysiological analysis of brain organoids. Nat Commun. 2025;16(1):2011. doi: 10.1038/s41467-024-55752-3
- Huang MS, Christakopoulos F, Roth JG, Heilshorn SC. Organoid bioprinting: from cells to functional tissues. Nat Rev Bioeng. 2025;3(2):126–142. doi: 10.1038/s44222-024-00268-0
- Groll J, Burdick JA, Cho D-W, et al. A definition of bioinks and their distinction from biomaterial inks. Biofabrication. 2018;11(1):013001. doi: 10.1088/1758-5090/aaec52
- Guvendiren M, Molde J, Soares R, Kohn J. Designing Biomaterials for 3D Printing. ACS Biomater Sci Eng. 2016;2:1679−1693. doi: 10.1021/acsbiomaterials.6b00121
- Zhang T, Sheng S, Cai W, et al. 3-D bioprinted human-derived skin organoids accelerate full-thickness skin defects repair. Bioact Mater. 2024;42:257–269. doi: 10.1016/j.bioactmat.2024.08.036
- Skylar-Scott MA, Uzel SG, Nam LL, et al. Biomanufacturing of organ-specific tissues with high cellular density and embedded vascular channels. Sci Adv. 2019;5(9):eaaw2459. doi: 10.1126/sciadv.aaw2459
- Sorrentino G, Rezakhani S, Yildiz E, et al. Mechano-modulatory synthetic niches for liver organoid derivation. Nat Commun. 2020;11(1):3416. doi: 10.1038/s41467-020-17161-0
- Cui H, Nowicki M, Fisher JP, Zhang LG. 3D Bioprinting for Organ Regeneration. Adv Healthcare Mater. 2017;6(1):1601118. doi: 10.1002/adhm.201601118
- Zhang B, Gao L, Ma L, Luo Y, Yang H, Cui Z. 3D Bioprinting: A Novel Avenue for Manufacturing Tissues and Organs. Engineering. 2019;5(4):777–794. doi: 10.1016/j.eng.2019.03.009
- Bebiano LB, Presa R, Fernandes L, Lourenço BN, Chaudhuri O, Pereira RF. Bioinks with varying densities of physical and chemical crosslinks modulate cellular responses in 3D by altering the viscoelasticity of the cell microenvironment. Mater Today. 2025;86:146–161. doi: 10.1016/j.mattod.2025.03.019
- GhavamiNejad A, Ashammakhi N, Wu XY, Khademhosseini A. Crosslinking Strategies for 3D Bioprinting of Polymeric Hydrogels. Small. 2020;16(35):2002931. doi: 10.1002/smll.202002931
- Xie M, Sun Y, Wang J, et al. Thermo-sensitive Sacrificial Microsphere-based Bioink for Centimeter-scale Tissue with Angiogenesis. IJB. 2022;8(4):599. doi: 10.18063/ijb.v8i4.599
- Wang Y, Yue H, Liu A, et al. Dual crosslinkable bioink for direct and embedded 3D bioprinting at physiological temperature. Mater Today. 2025;85:1–16. doi: 10.1016/j.mattod.2025.02.005
- Chai M, Zhong W, Yan S, et al. Diffusion-induced phase separation 3D printed scaffolds for dynamic tissue repair. BMEMat. 2024;2(3):e12108. doi: 10.1002/bmm2.12108
- Wu L, Wu G, Wu D, et al. Stiffness-Tunable Hydrogel Microfluidic Chip Reveals the Role of Stiffness in Cholangiocarcinoma Invasion and Pre-Metastatic Niche Formation. Adv Healthcare Mater. 2025:e03515. doi: 10.1002/adhm.202503515
- Nerger BA, Sinha S, Lee NN, et al. 3D Hydrogel Encapsulation Regulates Nephrogenesis in Kidney Organoids. Adv Mater. 2024;36(14):2308325. doi: 10.1002/adma.202308325
- Yang H, Zhang J, Li Y, et al. Multiscale Organization of Neural Networks in a 3D Bioprinted Matrix. Adv Sci. 2025;12(30):e04455. doi: 10.1002/advs.202504455
- Shin J, Tabatabaei Rezaei N, Choi S, Li Z, Kim D-H, Kim K. Photocrosslinkable Kidney Decellularized Extracellular Matrix-Based Bioink for 3D Bioprinting. Adv Healthcare Mater. 2025;14(24):2501616. doi: 10.1002/adhm.202501616
- Wang C, Wang Z, Lin Y, Ouyang L. Off-the-Shelf Granular Microtissue Bioinks: Long-Term Preserved Cellularized Porous Microgels with Enhanced Cell Activity for Shelf- Ready Biofabrication. Adv Mater. 2025;37(44):e06616. doi: 10.1002/adma.202506616
- Liu M, Jiang S, Witman N, et al. Intrinsically cryopreservable, bacteriostatic, durable glycerohydrogel inks for 3D bioprinting. Matter. 2023;6(3):983–999. doi: 10.1016/j.matt.2022.12.013
- Brown NC, Ames DC, Mueller J. Multimaterial extrusion 3D printing printheads. Nat Rev Mater. 2025;10(11):807– 825. doi: 10.1038/s41578-025-00809-y
- Tarassoli SP, Jessop ZM, Jovic T, Hawkins K, Whitaker IS. Candidate Bioinks for Extrusion 3D Bioprinting—A Systematic Review of the Literature. Front Bioeng Biotechnol. 2021;9:616753. doi: 10.3389/fbioe.2021.616753
- Miller JS, Stevens KR, Yang MT, et al. Rapid casting of patterned vascular networks for perfusable engineered three-dimensional tissues. Nat Mater. 2012;11(9):768–774. doi: 10.1038/nmat3357
- Hu Y, Zhu T, Cui H, Cui H. Integrating 3D Bioprinting and Organoids to Better Recapitulate the Complexity of Cellular Microenvironments for Tissue Engineering. Adv Healthcare Mater. 2025;14(3):2403762. doi: 10.1002/adhm.202403762
- Flégeau K, Puiggali-Jou A, Zenobi-Wong M. Cartilage tissue engineering by extrusion bioprinting utilizing porous hyaluronic acid microgel bioinks. Biofabrication. 2022;14(3):034105. doi: 10.1088/1758-5090/ac6b58
- Lawlor KT, Vanslambrouck JM, Higgins JW, et al. Cellular extrusion bioprinting improves kidney organoid reproducibility and conformation. Nat Mater. 2021;20(2):260–271. doi: 10.1038/s41563-020-00853-9
- Wu H, Xu F, Jin H, et al. 3D Nanofiber-Assisted Embedded Extrusion Bioprinting for Oriented Cardiac Tissue Fabrication. ACS Biomater Sci Eng. 2024;10(11):7256–7265. doi: 10.1021/acsbiomaterials.4c01611
- Bera AK, Rizvi MS, Kn V, Pati F. Engineering anisotropic tissue analogues: harnessing synergistic potential of extrusion-based bioprinting and extracellular matrix-based bioink. Biofabrication. 2025;17(1):015003. doi: 10.1088/1758-5090/ad86ec
- Han J, Jeong H-J, Choi J, et al. Bioprinted Patient-Derived Organoid Arrays Capture Intrinsic and Extrinsic Tumor Features for Advanced Personalized Medicine. Adv Sci. 2025;12(20):2407871. doi: 10.1002/advs.202407871
- Yogeshwaran S, Goodarzi Hosseinabadi H, Gendy DE, Miri AK. Design considerations and biomaterials selection in embedded extrusion 3D bioprinting. Biomater Sci. 2024;12(18):4506–4518. doi: 10.1039/D4BM00550C
- Gupta D, Derman ID, Xu C, Huang Y, Ozbolat IT. Droplet-based bioprinting. Nat Rev Methods Primers. 2025;5(1):25. doi: 10.1038/s43586-025-00394-y
- Hwang S, Yang K, Yoon HS, Roh YH. 3D printed pectin–chitosan polyelectrolyte hydrogels for controlled gastrointestinal release and colon-targeted delivery. Carbohydr Polym. 2025;376:124802. doi: 10.1016/j.carbpol.2025.124802
- Yoon WH, Lee H-R, Kim S, et al. Use of inkjet-printed single cells to quantify intratumoral heterogeneity. Biofabrication. 2020;12(3):035030. doi: 10.1088/1758-5090/ab9491
- Liu H, Tao T, Gan Z, et al. Organoid in droplet: Production of uniform pancreatic cancer organoids from single cells. Mater Today Bio. 2025;32:101765. doi: 10.1016/j.mtbio.2025.101765
- Zhao Y, Paul R, Li Q, et al. DOSS: Microfluidic engineered system enables automated intelligent sorting and chemo-evaluation of droplet-based colorectal adenocarcinoma organoids. Chem Eng J. 2025;522:166597. doi: 10.1016/j.cej.2025.166597
- Yang H, Li J, Zheng Y, et al. Ultra-small tissue-compatible organoid printer for rapid and controllable modeling of respiratory organoids. Device. 2024;2(8):100420. doi: 10.1016/j.device.2024.100420
- Gu Z, Fu J, Lin H, He Y. Development of 3D bioprinting: From printing methods to biomedical applications. Asian J Pharm Sci. 2020;15(5):529–557. doi: 10.1016/j.ajps.2019.11.003
- Singh YP, Moses JC, Kim MH, et al. Three-tier framework for high-throughput biofabrication: Integrating 3D bioprinting, assistive platforms, and translational opportunities. Bioact Mater. 2026;57:726–753. doi: 10.1016/j.bioactmat.2025.11.024
- Li Z, Chen L, Wu J, et al. A review of 3D bioprinting for organoids. Med Rev. 2025;5(4):318–338. doi: 10.1515/mr-2024-0089
- Gehlen J, Qiu W, Schädli GN, Müller R, Qin X-H. Tomographic volumetric bioprinting of heterocellular bone-like tissues in seconds. Acta Biomater. 2023;156:49–60. doi: 10.1016/j.actbio.2022.06.020
- Wang J, Wu Y, Li G, et al. Engineering Large-Scale Self-Mineralizing Bone Organoids with Bone Matrix- Inspired Hydroxyapatite Hybrid Bioinks. Adv Mater. 2024;36(30):2309875. doi: 10.1002/adma.202309875
- Bernal PN, Bouwmeester M, Madrid-Wolff J, et al. Volumetric Bioprinting of Organoids and Optically Tuned Hydrogels to Build Liver-Like Metabolic Biofactories. Adv Mater. 2022;34(15):2110054. doi: 10.1002/adma.202110054
- Wang M, Li W, Hao J, et al. Biomaterial-minimalistic photoactivated bioprinting of cell-dense tissues. Cell. 2025;189:1–17. doi: 10.1016/j.cell.2025.11.012
- Wang D, Guo Y, Zhu J, et al. Hyaluronic acid methacrylate/ pancreatic extracellular matrix as a potential 3D printing bioink for constructing islet organoids. Acta Biomater. 2023;165:86–101. doi: 10.1016/j.actbio.2022.06.036
- Hasenauer A, Bevc K, McCabe MC, et al. Volumetric printed biomimetic scaffolds support in vitro lactation of human milk-derived mammary epithelial cells. Sci Adv. 2025;11(23):eadu5793. doi: 10.1126/sciadv.adu5793
- Jain P, Kathuria H, Dubey N. Advances in 3D bioprinting of tissues/organs for regenerative medicine and in-vitro models. Biomaterials. 2022;287:121639. doi: 10.1016/j.biomaterials.2022.121639
- Cidonio G, Glinka M, Dawson JI, Oreffo ROC. The cell in the ink: Improving biofabrication by printing stem cells for skeletal regenerative medicine. Biomaterials. 2019;209:10– 24. doi: 10.1016/j.biomaterials.2019.04.009
- Zhou X, Yu X, You T, et al. 3D Printing-Based Hydrogel Dressings for Wound Healing. Adv Sci. 2024;11(47):2404580. doi: 10.1002/advs.202404580
- Shapira A, Dvir T. 3D Tissue and Organ Printing—Hope and Reality. Adv Sci. 2021;8(10):2003751. doi: 10.1002/advs.202003751
- Yan J, Li Z, Guo J, Liu S, Guo J. Organ-on-a-chip: A new tool for in vitro research. Biosens Bioelectron. 2022;216:114626. doi: 10.1016/j.bios.2022.114626
- Zhao D, Xu H, Ye Z, et al. Sequential-crosslinking facilitated droplet-droplet collision inkjet 3D printing of soft biomaterials. Addit Manuf. 2025;106:104809. doi: 10.1016/j.addma.2025.104809
- Zhang P, Teng Z, Zhou M, et al. Upconversion 3D Bioprinting for Noninvasive In Vivo Molding. Adv Mater. 2024;36(14):2310617. doi: 10.1002/adma.202310617
- Brassard JA, Nikolaev M, Hübscher T, Hofer M, Lutolf MP. Recapitulating macro-scale tissue self-organization through organoid bioprinting. Nat Mater. 2021;20(1):22–29. doi: 10.1038/s41563-020-00803-5
- Xie D, Chen B, Xue Y, et al. Printable and biocompatible hydrogels for residual-free and high-throughput printing patient-derived organoid biochips. Sci China Mater. 2024;67(8):2505–2514. doi: 10.1007/s40843-024-2933-8
- Corzo IJM, Fonseca JHLD, Ferman V. Optimizing biomaterial inks: A study on the printability of Carboxymethyl cellulose- Laponite nanocomposite hydrogels and dental pulp stem cells bioprinting. Bioprinting. 2024;43:e00358. doi: 10.1016/j.bprint.2024.e00358
- Gan Z, Qin X, Liu H, Liu J, Qin J. Recent advances in defined hydrogels in organoid research. Bioact Mater. 2023;28:386– 401. doi: 10.1016/j.bioactmat.2023.06.004
- Cho Y, You J, Lee JH. Natural Polymer-Based Hydrogel Platforms for Organoid and Microphysiological Systems: Mechanistic Insights and Translational Perspectives. Polymers. 2025;17(15):2109. doi: 10.3390/polym17152109
- Zhang Z, Zhu M, Luo H, Zeng F, Qi Z. Organoid scaffold materials: research and application. Front Bioeng Biotechnol. 2025;13:1637456. doi: 10.3389/fbioe.2025.1637456
- Li Y, Saiding Q, Wang Z, Cui W. Engineered biomimetic hydrogels for organoids. Prog Mater Sci. 2024;141:101216. doi: 10.1016/j.pmatsci.2023.101216
- Zhang C, Shen Y, Huang M, et al. Dynamic hydrogel mechanics in organoid engineering: From matrix design to translational paradigms. Bioact Mater. 2026;55:144–170. doi: 10.1016/j.bioactmat.2025.09.021
- Luo L, Liu L, Ding Y, Dong Y, Ma M. Advances in biomimetic hydrogels for organoid culture. Chem Commun. 2023;59(64):9675–9686. doi: 10.1039/d3cc01274c
- Hosseinzadeh B, Ahmadi M. Degradable Hydrogels: Design Mechanisms and Versatile Applications. Mater Today Sustain. 2023;23:100468. doi: 10.1016/j.mtsust.2023.100468
- Santhamoorthy M, Kim SC. A Review of the Development of Biopolymer Hydrogel-Based Scaffold Materials for Drug Delivery and Tissue Engineering Applications. Gels. 2025;11(3):178. doi: 10.3390/gels11030178
- Zhao G, Ge Y, Jin Y, et al. Research progress and potential of organoids based on biomimetic hydrogel materials. Colloids Surf B Biointerfaces. 2026;257:115168. doi: 10.1016/j.colsurfb.2025.115168
- Jergitsch M, Soiunov R, Selinger F, et al. Fabrication and validation of an affordable DIY coaxial 3D extrusion bioprinter. Sci Rep. 2025;15(1):22978. doi: 10.1038/s41598-025-06478-9
- Yang L, Shang J, Sun S, et al. Hierarchical Hydrogen Bond- Metal Coordination for High-Strength, High-Dissipation Elastomers over a Broad Temperature Range. Small. 2025;21(39):e06269. doi: 10.1002/smll.202506269
- Rajabi M, McConnell M, Cabral J, Ali MA. Chitosan hydrogels in 3D printing for biomedical applications. Carbohydr Polym. 2021;260:117768. doi: 10.1016/j.carbpol.2021.117768
- Szekalska M, Puciłowska A, Szymańska E, Ciosek P, Winnicka K. Alginate: Current Use and Future Perspectives in Pharmaceutical and Biomedical Applications. Int J Polym Sci. 2016;2016(1):7697031. doi: 10.1155/2016/7697031
- Gao Q, Kim B-S, Gao G. Advanced Strategies for 3D Bioprinting of Tissue and Organ Analogs Using Alginate Hydrogel Bioinks. Mar Drugs. 2021;19(12):708. doi: 10.3390/md19120708
- Levato R, Jungst T, Scheuring RG, Blunk T, Groll J, Malda J. From Shape to Function: The Next Step in Bioprinting. Adv Mater. 2020;32(12):1906423. doi: 10.1002/adma.201906423
- Daly AC, Cunniffe GM, Sathy BN, Jeon O, Alsberg E, Kelly DJ. 3D Bioprinting of Developmentally Inspired Templates for Whole Bone Organ Engineering. Adv Healthcare Mater. 2016;5(18):2353–2362. doi: 10.1002/adhm.201600182
- Wang S, Bai L, Hu X, et al. 3D bioprinting of neurovascular tissue modeling with collagen‐based low‐viscosity composites. Adv Healthcare Mater. 2023;12(25):2300004. doi: 10.1002/adhm.202300004
- Kang S-M, Lee J-H, Huh YS, Takayama S. Alginate microencapsulation for three-dimensional in vitro cell culture. ACS Biomater Sci Eng. 2020;7(7):2864–2879. doi: 10.1021/acsbiomaterials.0c00457
- Zhou Y, Gao X, Zhao M, Li L, Liu M. Three-dimensional printed sodium alginate clay nanotube composite scaffold for bone regeneration. Compos Sci Technol. 2024;250:110537. doi: 10.1016/j.compscitech.2024.110537
- Fu Z, Ouyang L, Xu R, Yang Y, Sun W. Responsive biomaterials for 3D bioprinting: A review. Mater Today. 2022;52:112–132. doi: 10.1016/j.mattod.2022.01.001
- Xu X, Lyu Y, Liu D, et al. Skin-Mountable Thermo-responsive Structured Hydrogel for Optical and Adhesion Coupled Functional Sensing. Small. 2025;21(7):2411808. doi: 10.1002/smll.202411808
- Waidi YO, Kariim I, Datta S. Bioprinting of gelatin-based materials for orthopedic application. Front Bioeng Biotechnol. 2024;12:30. doi: 10.3389/fbioe.2024.1357460
- Bociaga D, Bartniak M, Grabarczyk J, Przybyszewska K. Sodium Alginate/Gelatine Hydrogels for Direct Bioprinting—The Effect of Composition Selection and Applied Solvents on the Bioink Properties. Materials. 2019;12(17):254–271. doi: 10.3390/ma12172669
- Gao T, Gillispie GJ, Copus JS, et al. Optimization of gelatin– alginate composite bioink printability using rheological parameters: A systematic approach. Biofabrication. 2018;10(3):034106. doi: 10.1088/1758-5090/aacdc7
- Yue K, Santiago TD, Alvarez MM, Tamayol A, Annabi N, Khademhosseini A. Synthesis, properties, and biomedical applications of gelatin methacryloyl (GelMA) hydrogels. Biomaterials. 2015;73:254–271. doi: 10.1016/j.biomaterials.2015.08.045
- Hull SM, Brunel LG, Heilshorn SC. 3D bioprinting of cell‐laden hydrogels for improved biological functionality. Adv Mater. 2022;34(2):2103691. doi: 10.1002/adma.202103691
- Kim SA, Lee Y, Park K, et al. 3D printing of mechanically tough and self-healing hydrogels with carbon nanotube fillers. Int J Bioprint. 2023;9(5):765. doi: 10.18063/ijb.765
- Karvinen J, Kellomäki M. 3D-bioprinting of self-healing hydrogels. Eur Polym J. 2024;209:112864. doi: 10.1016/j.eurpolymj.2024.112864
- Majstorović N, Zahedtalaban M, Agarwal S. Printable Poly (N-acryloyl glycinamide) Nanocomposite Hydrogel Formulations. Polym J. 2023;55(10):1085–1095. doi: 10.1038/s41428-023-00798-1
- Wang H, Zhu H, Fu W, et al. A high strength self‐healable antibacterial and anti‐inflammatory supramolecular polymer hydrogel. Macromol Rapid Commun. 2017;38(9):1600695. doi: 10.1002/marc.201600695
- Yang M, Chu L, Zhuang Y, et al. Multi-Material Digital Light Processing (DLP) Bioprinting of Heterogeneous Hydrogel Constructs with Perfusable Networks. Adv Funct Mater. 2024;34(32):2316456. doi: 10.1002/adfm.202316456
- Ma W, Lu H, Xiao Y, Wu C. Advancing organoid development with 3D bioprinting. Organoid Res. 2025;1(1):025040004. doi: 10.36922/OR025040004
- Zhang D, Huerta-López C, Heilshorn SC. Organoid bioprinting to pattern the matrix microenvironment. Curr Opin Biomed Eng. 2025;35:100607. doi: 10.1016/j.cobme.2025.100607
- Huang L, Guo Z, Yang X, et al. Advancements in GelMA bioactive hydrogels: Strategies for infection control and bone tissue regeneration. Theranostics. 2025;15(2):460. doi: 10.7150/thno.103725
- Ghosh RN, Thomas J, Janardanan A, Namboothiri PK, Peter M. An insight into synthesis, properties and applications of gelatin methacryloyl hydrogel for 3D bioprinting. Mater Adv. 2023;4(22):5496–5529. doi: 10.1039/D3MA00715D
- Chalard AE, Dixon AW, Taberner AJ, Malmström J. Visible-light stiffness patterning of GelMA hydrogels towards in vitro scar tissue models. Front Cell Dev Biol. 2022;10:946754. doi: 10.3389/fcell.2022.946754
- Loukelis K, Koutsomarkos N, Mikos AG, Chatzinikolaidou M. Advances in 3D bioprinting for regenerative medicine applications. Regener Biomater. 2024;11:rbae033. doi: 10.1093/rb/rbae033
- Mierke CT. Bioprinting of cells, organoids and organs-on-a-chip together with hydrogels improves structural and mechanical cues. Cells. 2024;13(19):1638. doi: 10.3390/cells13191638
- Zhou F, Hong Y, Liang R, et al. Rapid printing of bio-inspired 3D tissue constructs for skin regeneration. Biomaterials. 2020;258:120287. doi: 10.1016/j.biomaterials.2020.120287
- Ma C, Hua B, Wang H, Ma T, Lv Q, Yan Z. Functionalized hydrogels of CeO2 and Urolithin A synergistically scavenge ROS and activate mitophagy for cartilage repair. Mater Today Bio. 2026;37:102785. doi: 10.1016/j.mtbio.2026.102785
- Gungor-Ozkerim PS, Inci I, Zhang YS, Khademhosseini A, Dokmeci MR. Bioinks for 3D bioprinting: an overview. Biomater Sci. 2018;6(5):915–946. doi: 10.1039/C7BM00765E
- Cruz-Acuña R, Quirós M, Huang S, et al. PEG-4MAL hydrogels for human organoid generation, culture, and in vivo delivery. Nat Protoc. 2018;13(9):2102–2119. doi: 10.1038/s41596-018-0079-5
- Lai W, Geliang H, Bin X, Wang W. Effects of hydrogel stiffness and viscoelasticity on organoid culture: a comprehensive review. Mol Med. 2025;31(1):83. doi: 10.1186/s10020-025-01131-7
- Huang B, Kim M, Zhang P, Oduro E, Rau DA, Cai LH. Additive Manufacturing of Molecular Architecture Encoded Stretchable Polyethylene Glycol Hydrogels and Elastomers. Adv Mater. 2025;38:e12806. doi: 10.1002/adma.202512806
- Mao L, Xarpidin B, Shi R, et al. Natural Enzyme-Loaded Polymeric Stealth Coating-Armed Engineered Probiotics by Disrupting Tumor Lactate Homeostasis to Synergistic Metabolism-Immuno-Enzyme Dynamic Therapy. Adv Sci. 2025;12(16):2417172. doi: 10.1002/advs.202417172
- Luo Z, Jiang S, Wu YL, Li Z. Nanozyme-based therapeutic systems for diabetic wound treatment. Ther Deliv. 2023;14(3):179–182. doi: 10.4155/tde-2023-0008
- Luo Z, Fan X, Chen Y, et al. Mitochondria targeted composite enzyme nanogels for synergistic starvation and photodynamic therapy. Nanoscale. 2021;13(42):17737– 17745. doi: 10.1039/d1nr06214j
- Luo Z, Cao Y, Liao Z, et al. Mitochondria-Targeted Gold Biometallization for Photoacoustically Visualized Photothermal Cancer Therapy. ACS Nano. 2024;18(43):29667–29677. doi: 10.1021/acsnano.4c08567
- Li Z, Fan X, Luo Z, et al. Nanoenzyme-chitosan hydrogel complex with cascade catalytic and self-reinforced antibacterial performance for accelerated healing of diabetic wounds. Nanoscale. 2022;14(40):14970–14983. doi: 10.1039/d2nr04171e
- Hu H, Lin Y, Yang B, et al. Biomineralization-inspired functional biomaterials: From principles to practice. Chem Eng J. 2025;504:158624. doi: 10.1016/j.cej.2024.158624
- Fan X, Luo Z, Chen Y, et al. Oxygen self-supplied enzyme nanogels for tumor targeting with amplified synergistic starvation and photodynamic therapy. Acta Biomater. 2022;142:274–283. doi: 10.1016/j.actbio.2022.01.056
- Wang Q, Luo Z, Li Z, et al. In-situ oxygen-supplying ROS nanopurifier for enhanced healing of MRSA-infected diabetic wounds via microenvironment modulation. Acta Biomater. 2025;193:334–347. doi: 10.1016/j.actbio.2024.12.044
- Palani N, Mendonce KC, Syed Altaf RR, et al. Next-generation smart wound dressings: AI integration, biosensors, and electrospun nanofibers for chronic wound therapy. J Biomater Sci Polym Ed. 2025;1–51. doi: 10.1080/09205063.2025.2540362
- Zheng Y, Mao L, Wang Q, et al. Mitochondria-Targeted ROS Scavenging Natural Enzyme Cascade Nanogels for Periodontitis Treatment via Hypoxia Alleviation and Immunomodulation. Adv Sci. 2025;12(29):e07481. doi: 10.1002/advs.202507481
- Stepanovska J, Supova M, Hanzalek K, Broz A, Matejka R. Collagen bioinks for bioprinting: a systematic review of hydrogel properties, bioprinting parameters, protocols, and bioprinted structure characteristics. Biomedicines. 2021;9(9):1137. doi: 10.3390/biomedicines9091137
- Shpichka A, Osipova D, Efremov Y, et al. Fibrin-based Bioinks: New Tricks from an Old Dog. Int J Bioprint. 2024;6(3):269. doi: 10.18063/ijb.v6i3.269
- Li T, Ma Z, Zhang Y, et al. Regeneration of Humeral Head Using a 3D Bioprinted Anisotropic Scaffold with Dual Modulation of Endochondral Ossification. Adv Sci. 2023;10(12):2205059. doi: 10.1002/advs.202205059
- Fisch P, Broguiere N, Finkielsztein S, Linder T, Zenobi- Wong M. Bioprinting of Cartilaginous Auricular Constructs Utilizing an Enzymatically Crosslinkable Bioink. Adv Funct Mater. 2021;31(16):2008261. doi: 10.1002/adfm.202008261
- Porcionatto MA. Strategies to use Fibrinogen as Bioink for 3D Bioprinting Fibrin-Based Soft and Hard Tissues. Acta Biomater. 2020;117:60–76. doi: 10.1016/j.actbio.2020.09.024
- Yan Y, Li X, Gao Y, et al. 3D bioprinting of human neural tissues with functional connectivity. Network Daily News. 2024;31(2):260–274.e267. doi: 10.1016/j.stem.2023.12.009
- Bhat SM, Badiger VA, Vasishta S, et al. 3D tumor angiogenesis models: recent advances and challenges. J Cancer Res Clin Oncol. 2021;147(12):3477–3494. doi: 10.1007/s00432-021-03814-0
- Xianyu B, Xu H. Dynamic covalent bond-based materials: From construction to biomedical applications. Supramol Mater. 2024;3:100070. doi: 10.1016/j.supmat.2024.100070
- Du W, Li H, Luo J, et al. Dynamic Schiff base linkage-based double-network hydrogels with injectable, self-healing, and pH-responsive properties for bacteria-infected wound healing. Cellulose. 2024;31(10):6373–6385. doi: 10.1007/s10570-024-05972-z
- Hidaka M, Sakai S. Photo-and Schiff Base-Crosslinkable Chitosan/Oxidized Glucomannan Composite Hydrogel for 3D Bioprinting. Polysaccharides. 2025;6(1):19. doi: 10.3390/polysaccharides6010019
- Willems C, Qi F, Trutschel M-L, Groth T. Functionalized gelatin/polysaccharide hydrogels for encapsulation of hepatocytes. Gels. 2024;10(4):231. doi: 10.3390/gels10040231
- Zhang C, Fu Z, Liu Q, et al. Bioprinted M2 macrophage-derived extracellular vesicle mimics attenuate foreign body reaction and enhance vascularized tissue regeneration. Biofabrication. 2025;17(3):035007. doi: 10.1088/1758-5090/add49f
- Wang H, Zhang J, Bai H, et al. 3D printed cell-free bilayer porous scaffold based on alginate with biomimetic microenvironment for osteochondral defect repair. Biomater Adv. 2025;167:214092. doi: 10.1016/j.bioadv.2024.214092
- Maia FR, Oliveira JM, Reis RL. Handbook of the Extracellular Matrix: Biologically-Derived Materials. Springer; 2024. doi: 10.1007/978-3-031-56363-8
- Mazzoldi EL, Gaudenzi G, Ginestra PS, Ceretti E, Giliani SC. Evaluating cells metabolic activity of bioinks for bioprinting: the role of cell-laden hydrogels and 3D printing on cell survival. Front Bioeng Biotechnol. 2024;12:1450838. doi: 10.3389/fbioe.2024.1450838
- Cavallo A, Al Kayal T, Mero A, et al. Fibrinogen-based bioink for application in skin equivalent 3D bioprinting. J Funct Biomater. 2023;14(9):459. doi: 10.3390/jfb14090459
- Guo K, van den Beucken T. Advances in drug-induced liver injury research: in vitro models, mechanisms, omics and gene modulation techniques. Cell Biosci. 2024;14(1):134. doi: 10.1186/s13578-024-01317-2
- Jiao W, Shan J, Gong X, et al. GelMA hydrogel: A game-changer in 3D tumor modeling. Mater Today Chem. 2024;38:102111. doi: 10.1016/j.mtchem.2024.102111
- Maisumu G, Willerth S, Nestor MW, et al. Brain organoids: Building higher-order complexity and neural circuitry models. Trends Biotechnol. 2025;43(7):1583–1598. doi: 10.1016/j.tibtech.2025.02.009
- de Paula AGP, de Lima JD, Bastos TSB, et al. Decellularized extracellular matrix: the role of this complex biomaterial in regeneration. ACS Omega. 2023;8(25):22256–22267. doi: 10.1021/acsomega.2c06216
- Walejewska E, Melchels FP, Paradiso A, et al. Tuning physical properties of GelMA hydrogels through microarchitecture for engineering osteoid tissue. Biomacromolecules. 2023;25(1):188–199. doi: 10.1021/acs.biomac.3c00909
- Roth JG, Brunel LG, Huang MS, et al. Spatially controlled construction of assembloids using bioprinting. Nat Commun. 2023;14(1):4346. doi: 10.1038/s41467-023-40006-5
- Wu M, Liu H, Zhu Y, et al. Bioinspired soft-hard combined system with mild photothermal therapeutic activity promotes diabetic bone defect healing via synergetic effects of immune activation and angiogenesis. Theranostics. 2024;14(10):4014. doi: 10.7150/thno.97335
- Torras N, Zabalo J, Abril E, Carré A, García-Díaz M, Martinez E. A bioprinted 3D gut model with crypt-villus structures to mimic the intestinal epithelial-stromal microenvironment. Biomater Adv. 2023;153:213534. doi: 10.1016/j.bioadv.2023.213534
- Xiao H, Liang Z, Gong X, et al. Application of instant assembly of collagen to bioprint cardiac tissues. APL Bioeng. 2025;9(2):026124. doi: 10.1063/5.0252746
- Zhe M, Wu X, Yu P, et al. Recent advances in decellularized extracellular matrix-based bioinks for 3D bioprinting in tissue engineering. Materials. 2023;16(8):3197. doi: 10.3390/ma16083197
- Hwang DG, Choi Y-m, Jang J. 3D bioprinting-based vascularized tissue models mimicking tissue-specific architecture and pathophysiology for in vitro studies. Front Bioeng Biotechnol. 2021;9:685507. doi: 10.3389/fbioe.2021.685507
- Shin J, Tabatabaei Rezaei N, Choi S, Li Z, Kim DH, Kim K. Photocrosslinkable Kidney Decellularized Extracellular Matrix‐Based Bioink for 3D Bioprinting. Adv Healthcare Mater. 2025;14(24):2501616. doi: 10.1002/adhm.202501616
- Kim JJ, Cho D-W. Advanced strategies in 3D bioprinting for vascular tissue engineering and disease modelling using smart bioinks. Virtual Phys Prototyp. 2024;19(1):e2395470. doi: 10.1080/17452759.2024.2395470
- Bansal AM, Horowitz LF, Yeung M, Gujral TS, Folch A. Bioprinting of microdissected tumor “cuboids” in hydrogels. bioRxiv Preprint posted online 2025;671932. doi: 10.1101/2025.09.05.671932
- Leonardo M, Prajatelistia E, Judawisastra H. Alginate-based bioink for organoid 3D bioprinting: A review. Bioprinting. 2022;28:e00246. doi: 10.1016/j.Bprint.2022.E00246
- He C, Yan J, Fu Y, Guo J, Shi Y, Guo J. Organoid bioprinting strategy and application in biomedicine: A review. Int J Bioprint. 2023;9(6):0112. doi: 10.36922/ijb.0112
- Ren Y, Yuan C, Liang Q, et al. 3D Bioprinting for Engineering Organoids and Organ‐on‐a‐Chip: Developments and Applications. Med Res Rev. 2025;45:1630–1650. doi: 10.1002/med.22121
- Jones CFE, Di Cio S, Connelly JT, Gautrot JE. Design of an Integrated Microvascularized Human Skin-on-a- Chip Tissue Equivalent Model. Front Bioeng Biotechnol. 2022;10:915702. doi: 10.3389/fbioe.2022.915702
- Oftadeh R, Azadi M, Donovan M, et al. Poroelastic behavior and water permeability of human skin at the nanoscale. PNAS Nexus. 2023;2(8):pgad240. doi: 10.1093/pnasnexus/pgad240
- Yu S-y, Wu T, Xu K-h, et al. 3D bioprinted biomimetic MOF-functionalized hydrogel scaffolds for bone regeneration: Synergistic osteogenesis and osteoimmunomodulation. Mater Today Bio. 2025;32:101740. doi: 10.1016/j.mtbio.2025.101740
- Yu B, Zhou D, Wang F, Chen X, Li M, Su J. Organoids for tissue repair and regeneration. Mater Today Bio. 2025;33:102013. doi: 10.1016/j.mtbio.2025.102013
- Zhang C-J, Jin Z-B. Turning point of organoid transplantation: first-in-human trial of iPSC-derived retinal organoid grafting in patients with retinitis pigmentosa. Sci China Life Sci. 2024;67(5):1082–1084. doi: 10.1007/s11427-023-2531-3
- Poling HM, Sundaram N, Fisher GW, et al. Human pluripotent stem cell-derived organoids repair damaged bowel in vivo. Cell Stem Cell. 2024;31(10):1513–1523.e1517. doi: 10.1016/j.stem.2024.08.009
- Jun Y, Nguyen-Ngoc K-V, Sai S, et al. Engineered vasculature induces functional maturation of pluripotent stem cell-derived islet organoids. Dev Cell. 2025;60(18):2455–2469. doi: 10.1016/j.devcel.2025.04.024
- Budharaju H, Singh RK, Kim H-W. Bioprinting for drug screening: A path toward reducing animal testing or redefining preclinical research? Bioact Mater. 2025;51:993– 1017. doi: 10.1016/j.bioactmat.2025.07.006
- Ma Y, He R, Deng B, et al. Advanced 3D bioprinted liver models with human-induced hepatocytes for personalized toxicity screening. J Tissue Eng. 2025;16:1–12. doi: 10.1177/20417314241313341
- Yang J, Wang L, Wu R, et al. 3D Bioprinting in Cancer Modeling and Biomedicine: From Print Categories to Biological Applications. ACS Omega. 2024;9(44):25. doi: 10.1021/acsomega.4c06051
- Jung M, Poltavets V, Skhinas JN, et al. High-throughput 3D engineered paediatric tumour models for precision medicine. Mol Syst Biol. 2025;21(12):1748–1777. doi: 10.1038/s44320-025-00152-y
- Li Z, Li K, Zhang C, et al. Bioprinted Organoids: An Innovative Engine in Biomedicine. Adv Sci. 2025;12(33):e07317. doi: 10.1002/advs.202507317
- Chliara MA, Elezoglou S, Zergioti I. Bioprinting on Organ-on-Chip: Development and Applications. Biosensors. 2022;12(12):1135. doi: 10.3390/bios12121135
- Mitrofanova O, Nikolaev M, Xu Q, et al. Bioengineered human colon organoids with in vivo-like cellular complexity and function. Cell Stem Cell. 2024;31(8):1175–1186.e1177. doi: 10.1016/j.stem.2024.05.007
- Li G, He J, Shi J, et al. Bioprinting functional hepatocyte organoids derived from human chemically induced pluripotent stem cells to treat liver failure. Gut. 2025;74(7):1150–1164. doi: 10.1136/gutjnl-2024-333885
- Son J, Li S, Jeong W. Bioprinting Vascularized Constructs for Clinical Relevance: Engineering Hydrogel Systems for Biological Maturity. Gels. 2025;11(8):636. doi: 10.3390/gels11080636

