Si–C covalent bonding-mediated interfacial charge transfer enhances photodynamic antitumor effects of selective laser sintered bone scaffolds

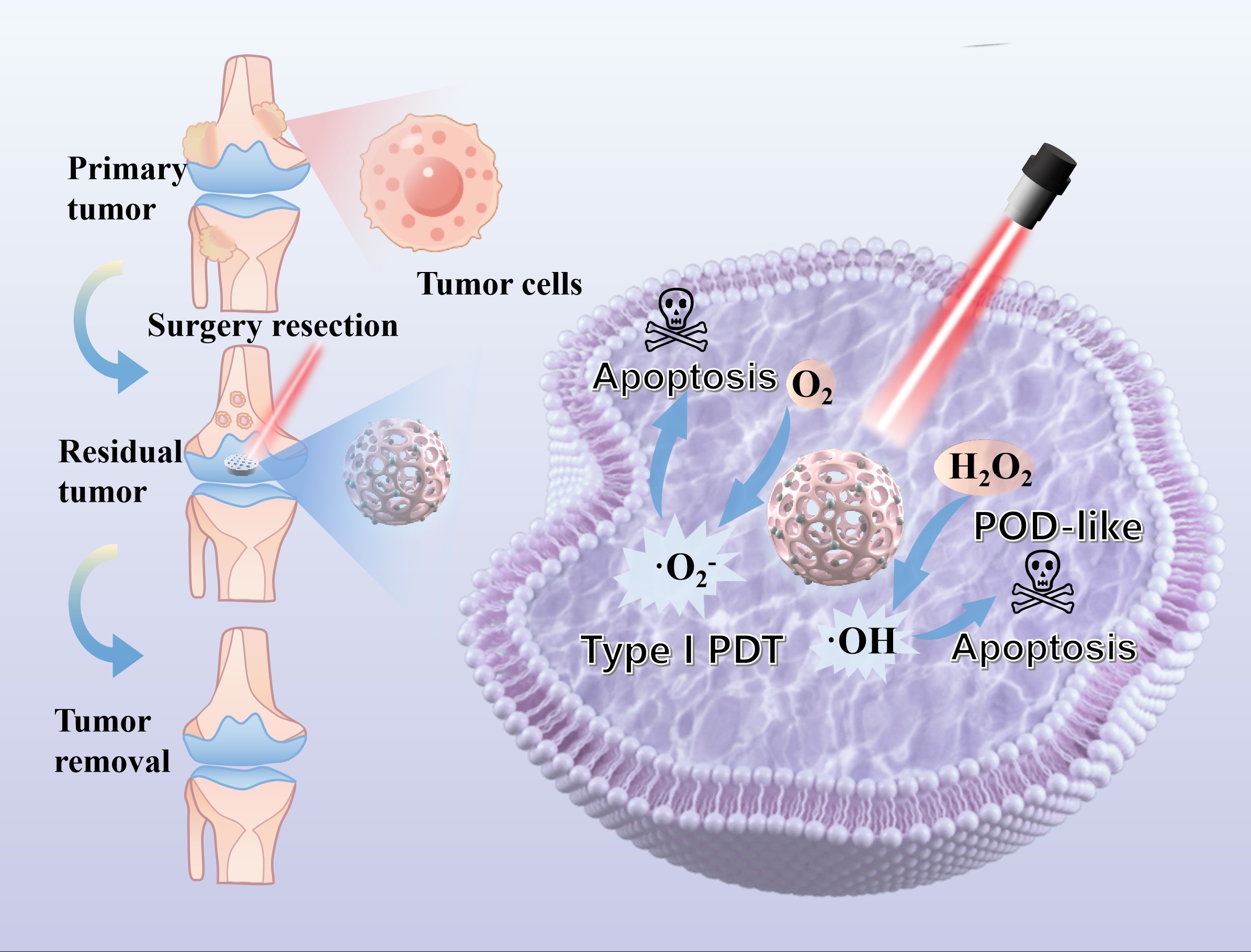
Nitrogen-doped carbon dots (NCDs) show promising potential in photodynamic antitumor applications due to their appropriate band gap and photo-responsiveness. Nevertheless, their therapeutic efficacy is limited by both a low reactive oxygen species (ROS) quantum yield and their propensity for aggregation. Herein, NCDs were encapsulated within mesoporous silica nanoparticles (MSNs) to fabricate an effective photosensitizer (NCDs@MSN) via a one-pot hydrothermal method. The covalent Si–C bonds formed between NCDs and MSNs enhanced interfacial charge transfer, thereby substantially amplifying the generation of ROS under hypoxic conditions. Meanwhile, the mesoporous structure of MSNs prevented NCD aggregation and provided a larger accessible surface area with more exposed active sites. Electron spin resonance spectroscopy confirmed the light-triggered generation of ROS, validating its potent ROS generation capacity under hypoxia. Subsequently, NCDs@ MSN were incorporated into poly-L-lactic acid to fabricate a composite scaffold via selective laser sintering, which was designed for postoperative photodynamic management of tumorous bone defects. The resulting scaffold exhibited potent photodynamic cytotoxicity against tumor cells alongside excellent biocompatibility. This work presents a potential strategy for engineering intelligent implants to prevent postoperative tumor recurrence.
- Abad-Montero D, Gandioso A, Izquierdo-García E, et al. Ruthenium(II) polypyridyl complexes containing COUBPY ligands as potent photosensitizers for the efficient phototherapy of hypoxic tumors. J Am Chem Soc. 2025;147(9):7360-7376. doi: 10.1021/jacs.4c15036
- Nosaka Y, Nosaka AY. Generation and Detection of Reactive Oxygen Species in Photocatalysis. Chem Rev. 2017;117(17):11302-11336. doi: 10.1021/acs.chemrev.7b00161
- Shuai C, Li D, Xie H, Yao X, Peng S, Gao C. Programmable lamellar eutectic Zn-2Al-Mg biodegradable implants manufactured by laser powder bed fusion for synergistic strength-ductility and osteogenesis. Adv Healthc Mater. 2025;14(32):2501917. doi: 10.1002/adhm.202501917
- Lin Y, Wu S, Li X, et al. Microstructure and performance of Z-scheme photocatalyst of silver phosphate modified by MWCNTs and Cr-doped SrTiO3 for malachite green degradation. Appl Catal B. 2018;227:557-570. doi: 10.1016/j.apcatb.2018.01.054
- Li D, Xie H, Gao C, Jiang H, Wang L, Shuai C. Harmonic heterostructured pure Ti fabricated by laser powder bed fusion for excellent wear resistance via strength-plasticity synergy. Opto-Electron Adv. 2025;8(9):250043. doi: 10.29026/oea.2025.250043
- Li Z, Zhang T, Fan F, Gao F, Ji H, Yang L. Piezoelectric materials as sonodynamic sensitizers to safely ablate tumors: a case study using black phosphorus. J Phys Chem Lett. 2020;11(4):1228-1238. doi: 10.1021/acs.jpclett.9b03769
- Lu S, Yang B. Carbon dots are shining in the world. SmartMat. 2022;3(2):207. doi: 10.1002/smm2.1132
- Gao X, Tan J, Ye T, et al. Ti3C2Tx-enhanced photothermoelectric performance of Bi2Te3 in scaffold for improved osteogenic potential. J Colloid Interface Sci. 2026;702:138794. doi: 10.1016/j.jcis.2025.138794
- Shuai C, Pan G, Wang Z, et al. Bifunctional MoS2@Cu2O heterojunction within scaffold for dual-mode synergistic antibacterial effects. Appl Surf Sci. 2025;686:162154. doi: 10.1016/j.apsusc.2024.162154
- Zhang Z, Song S, Ding Y, et al. Ultrasonic enhanced liquid-liquid interfacial reaction for improving the synthesis of Iron-doped carbon dots (Fe-CDs) for achieving superior photocatalytic performance. J Colloid Interface Sci. 2024;669:816-824. doi: 10.1016/j.jcis.2024.05.070
- Kurdyukov DA, Eurov DA, Stovpiaga EY, et al. Novel solid state of silica with ultra-high specific surface area. Mater Today. 2025;88:146-154. doi: 10.1016/j.mattod.2025.06.006
- Tang X, Wu Y, Shen Y, et al. Heterogeneous-structure based AuNBs@TiO2 nano-photosensitizers for computed tomography imaging guided NIR-II photodynamic therapy and cancer metastatic prevention. Adv Healthc Mater. 2024;13(18):2304209. doi: 10.1002/adhm.202304209
- Lee H, Han G, Na Y, et al. 3D-printed tissue-specific nanospike-based adhesive materials for time-regulated synergistic tumor therapy and tissue regeneration in vivo. Adv Funct Mater. 2024;34(48):2406237. doi: 10.1002/adfm.202406237
- Tian B, Liu S, Yu C, et al. A metal-free mesoporous carbon dots/silica hybrid type I photosensitizer with enzyme activity for synergistic treatment of hypoxic tumor. Adv Funct Mater. 2023;33(25):2300818. doi: 10.1002/adfm.202300818
- Wang L, Zhang H, Zhou X, Liu Y, Lei B. Preparation, characterization and oxygen sensing properties of luminescent carbon dots assembled mesoporous silica microspheres. J Colloid Interface Sci. 2016;478:256-262. doi: 10.1016/j.jcis.2016.06.026
- Shuai X, Yang Y, Qi F, Yang M, Shuai C. Phase-field modeling of laser sintering degradable biopolymer. Compos Commun. 2025;53:102244. doi: 10.1016/j.coco.2024.102244
- Sun B, Peng S, Zhang Z, et al. A bifunctional scaffold achieves a two-stage strategy for repairing tumorous bone defects. Colloids Surf A Physicochem Eng Asp. 2025;726:137996. doi: 10.1016/j.colsurfa.2025.137996
- Aebisher D, Czech S, Dynarowicz K, et al. Photodynamic therapy: past, current, and future. Int J Mol Sci. 2024;25(20):11325. doi: 10.3390/ijms252011325
- Aebisher D, Szpara J, Bartusik-Aebisher D. Advances in medicine: photodynamic therapy. Int J Mol Sci. 2024;25(15):8258. doi: 10.3390/ijms25158258
- Blasi M, Pagliara M, Lanza A, et al. Photodynamic Therapy in Ocular Oncology. Biomedicines. 2018;6(1):17. doi: 10.3390/biomedicines6010017
- Chou W, Sun T, Peng N, et al. Photodynamic therapy-induced anti-tumor immunity: influence factors and synergistic enhancement strategies. Pharmaceutics. 2023;15(11):2617. doi: 10.3390/pharmaceutics15112617
- Cramer GM, Cengel KA, Busch TM. Forging forward in photodynamic therapy. Cancer Res. 2022;82(4):534-536. doi: 10.1158/0008-5472.Can-21-4122
- Qu J, Zhou W, Zhang W, et al. Tunable time-dependent phosphorescent colors and evolution direction of solvent free grown carbon dots within hollow mesoporous silica for dynamic information encryption. Chem Eng J. 2025;524:169198. doi: 10.1016/j.cej.2025.169198
- Cui X, Li X, Peng C, et al. Beyond external light: on-spot light generation or light delivery for highly penetrated photodynamic therapy. ACS Nano. 2023;17(21):20776-20803. doi: 10.1021/acsnano.3c05619
- Ren Y, Fan Z. Synthesis of fluorescent probe based on molecularly imprinted polymers on nitrogen-doped carbon dots for determination of tobramycin in milk. Food Chem. 2023;416:135792. doi: 10.1016/j.foodchem.2023.135792
- Hang L, Zhang T, Wen H, et al. Controllable photodynamic performance via an acidic microenvironment based on two dimensional metal-organic frameworks for photodynamic therapy. Nano Res. 2021;14(3):660-666. doi: 10.1007/s12274-020-3093-1
- Ye J, Miao B, Xiong Y, et al. 3D printed porous magnesium metal scaffolds with bioactive coating for bone defect repair: enhancing angiogenesis and osteogenesis. J Nanobiotechnology. 2025;23(1):355. doi: 10.1186/s12951-025-03222-3
- Li CA, Nan J, Ye Q, et al. Amplifying anti-tumor immune responses via mitochondria-targeting near-infrared photodynamic therapy. Adv Sci. 2025;12(33):2505525. doi: 10.1002/advs.202505525
- Liu LG, Sun YM, Liu ZY, et al. Halogenated gallium corroles: DNA interaction and photodynamic antitumor activity. Inorg Chem. 2021;60(4):2234-2245. doi: 10.1021/acs.inorgchem.0c03016
- Fan J, Ding Z, Cai Y, et al. Revolutionizing Bone Regeneration: Vascularized Bone Tissue Engineering with Advanced 3D Printing Technology. Aggregate. Published online March 6, 2025. doi: 10.1002/agt2.731
- Nompumelelo Simelane NW, Kruger CA, Abrahamse H. Photodynamic diagnosis and photodynamic therapy of colorectal cancer in vitro and in vivo. RSC Adv. 2020;10(68):41560-41576. doi: 10.1039/d0ra08617g
- Mesquita MQ, Dias CJ, Neves MGPM, Almeida A, Faustino MAF. Revisiting Current Photoactive Materials for Antimicrobial Photodynamic Therapy. Molecules. 2018;23(10):2424. doi: 10.3390/molecules23102424
- Wang H, Ewetse MP, Ma C, et al. The “light knife” for gastric cancer: photodynamic therapy. Pharmaceutics. 2022;15(1):101. doi: 10.3390/pharmaceutics15010101
- Korbelik M. Photodynamic therapy supported by antitumor lipids. Pharmaceutics. 2023;15(12):2723. doi: 10.3390/pharmaceutics15122723
- Sun B, Chen Y, Yu H, et al. Photodynamic PEG-coated ROS sensitive prodrug nanoassemblies for core-shell synergistic chemo-photodynamic therapy. Acta Biomater. 2019;92:219-228. doi: 10.1016/j.actbio.2019.05.008
- Wang GD, Nguyen HT, Chen H, et al. X-Ray Induced Photodynamic Therapy: A Combination of Radiotherapy and Photodynamic Therapy. Theranostics. 2016;6(13):2295-2305.doi: 10.7150/thno.16141
- Wang H, Liu H, Guo Y, et al. Photosynthetic microorganisms coupled photodynamic therapy for enhanced antitumor immune effect. Bioact Mater. 2022;12:97-106. doi: 10.1016/j.bioactmat.2021.10.028
- Wei F, Rees TW, Liao X, Ji L, Chao H. Oxygen self-sufficient photodynamic therapy. Coord Chem Rev. 2021;432:213714. doi: 10.1016/j.ccr.2020.213714
- Tam LKB, Ng DKP. “Click” for precise photodynamic therapy. Mater Chem Front. 2023;7(16):3184-3193. doi: 10.1039/d3qm00431g
- Wu C, Li Y, Cheng Z, et al. Cell-penetrating riboflavin conjugate for antitumor photodynamic therapy. Chin Chem Lett. 2022;33(9):4339-4344. doi: 10.1016/j.cclet.2022.01.036
- Yu Y, Wang H, Zhuang Z, et al. Self-adaptive photodynamic-to-photothermal switch for smart antitumor photoimmunotherapy. ACS Nano. 2024;18(20):13019-13034. doi: 10.1021/acsnano.4c01600
- Zheng K, Liu H, Liu X, et al. Tumor targeting chemo- and photodynamic therapy packaged in albumin for enhanced anti-tumor efficacy. Int J Nanomedicine. 2020;15:151-167. doi: 10.2147/ijn.S227144
- Yang Y, Ji C, Zhu ZH, et al. Metal–organic frameworks based on fluorogens with aggregation-induced emission for enhanced sonodynamic therapy. Chem Mater. 2024;36(10):4955-4966. doi: 10.1021/acs.chemmater.3c02495
- Kazimierczak P, Benko A, Palka K, Canal C, Kolodynska D, Przekora A. Novel synthesis method combining a foaming agent with freeze-drying to obtain hybrid highly macroporous bone scaffolds. J Mater Sci Technol. 2020;43:52-63. doi: 10.1016/j.jmst.2020.01.006
- Yuan X, Zhu W, Yang Z, et al. Recent advances in 3D printing of smart scaffolds for bone tissue engineering and regeneration. Adv Mater. 2024;36(34):2403641. doi: 10.1002/adma.202403641
- Song P, Hu C, Pei X, et al. Dual modulation of crystallinity and macro-/microstructures of 3D printed porous titanium implants to enhance stability and osseointegration. J Mater Chem B. 2019;7(17):2865-2877. doi: 10.1039/c9tb00093c
- Wu K, El Zowalaty AE, Sayin VI, Papagiannakopoulos T. The pleiotropic functions of reactive oxygen species in cancer. Nat Cancer. 2024;5(3):384-399. doi: 10.1038/s43018-024-00738-9
- Zhong K, Zhang Z, Cheng W, et al. Photodynamic O2 economizer encapsulated with DNAzyme for enhancing mitochondrial gene-photodynamic therapy. Adv Healthc Mater. 2024;13(5):2302495. doi: 10.1002/adhm.202302495
- Chen X, Yong Z, Xiong Y, et al. Hydroxyethyl starch conjugates co-assembled nanoparticles promote photodynamic therapy and antitumor immunity by inhibiting antioxidant systems. Asian J Pharm Sci. 2024;19(5):100950. doi: 10.1016/j.ajps.2024.100950
- He S, Wang L, Wu D, et al. Dual-responsive supramolecular photodynamic nanomedicine with activatable immunomodulation for enhanced antitumor therapy. Acta Pharm Sin B. 2024;14(2):765-780. doi: 10.1016/j.apsb.2023.10.006
- Li J, Xing Y, Chen X. Intercalating of AIEgens into MoS2 nanosheets to induce crystal phase transform for enhanced photothermal and photodynamic synergetic anti-tumor therapy. Talanta. 2024;271:125677. doi: 10.1016/j.talanta.2024.125677
- Xie X, Zhang S, Liu M, et al. Boosting anti-tumor immunity with boron-based nanosheets via photodynamic-elicited pyroptosis and adjuvant delivery. J Mater Chem B. 2025;13(28):8380-8394. doi: 10.1039/d5tb00801h
- Zhang Y, Ying-Kit C, NDKP, Wing-Ping F. Enhancement of innate and adaptive anti-tumor immunity by serum obtained from vascular photodynamic therapy-cured BALB/c mouse. Cancer Immunol Immunother. 2021;70(11):3217-3233. doi: 10.1007/s00262-021-02917-4

