Immunomodulatory 3D-printed hydroxyapatite/tricalcium phosphate/polycaprolactone scaffolds promote bone regeneration via macrophage polarization

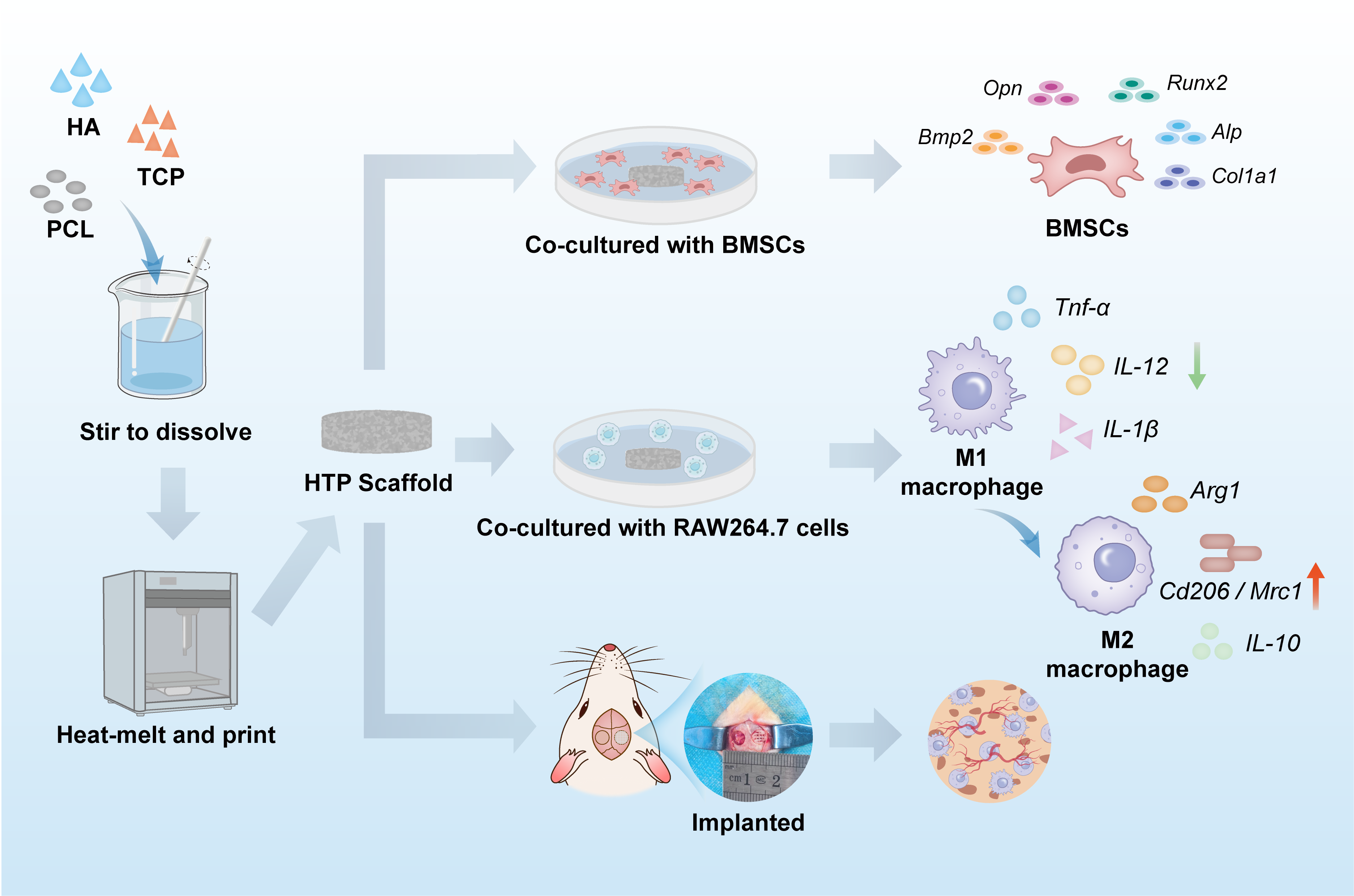
Excessive inflammation remains a significant impediment to the regeneration of critical-sized bone defects, where the local immune microenvironment plays a crucial role in osteogenesis. However, most bone scaffolds primarily emphasize mechanical support and osteoconductivity, while their immunomodulatory potential remains largely unexplored. In this study, we designed and fabricated three-dimensional–printed hydroxyapatite/β-tricalcium phosphate/polycaprolactone (HTP) composite scaffolds to regulate macrophage polarization and promote bone regeneration. The HTP scaffolds demonstrated exceptional structural integrity and mechanical strength, facilitating the adhesion, proliferation, and osteogenic differentiation of bone marrow-derived mesenchymal stem cells. Notably, the HTP scaffolds effectively modulated the immune microenvironment by inhibiting the polarization of pro-inflammatory M1 macrophages and promoting their transition toward the regenerative M2 phenotype. This immunomodulatory effect further enhanced osteogenic factor secretion, establishing a correlation between immunoregulation and osteogenesis. In a rat calvarial defect model, the HTP scaffolds significantly increased M2 macrophage infiltration, promoted angiogenesis, and accelerated new bone formation compared to other groups. This study demonstrates that three-dimensional–printed HTP composite scaffolds promote bone regeneration and angiogenesis by establishing a regenerative immune microenvironment, highlighting their potential as an advanced immunomodulatory platform for bone tissue engineering.
- Ma XY, Yuan H, Cui D, et al. Management of segmental defects post open distal femur fracture using a titanium cage combined with the masquelet technique a single-centre report of 23 cases. Injury. 2023;54(12):111130. doi: 10.1016/j.injury.2023.111130
- Zheng S, Zhong H, Cheng H, et al. Engineering multifunctional hydrogel with osteogenic capacity for critical-size segmental bone defect repair. Front Bioeng Biotechnol. 2022;10:899457. doi: 10.3389/fbioe.2022.899457
- Engel EE, Gava NF, Santos MA dos, Baldoino LG, Ismael LK, Modolo LFM. Updates on bone grafts and substitutes. Rev Bras Ortop. 2025;60(6):s00451814114. doi: 10.1055/s-0045-1814114
- Ferraz MP. Bone grafts in dental medicine: An overview of autografts, allografts and synthetic materials. Materials. 2023;16(11):4117. doi: 10.3390/ma16114117
- Ciszyński M, Dominiak S, Dominiak M, Gedrange T, Hadzik J. Allogenic bone graft in dentistry: A review of current trends and developments. Int J Mol Sci. 2023;24(23):16598. doi: 10.3390/ijms242316598
- Wang H, Kang J. Bone grafts and synthetic substitutes in dental applications: A comprehensive review of molecular mechanisms, materials evolution, and clinical perspective. Front Bioeng Biotechnol. 2026;13:1759864. doi: 10.3389/fbioe.2025.1759864
- Gharibshahian M, Salehi M, Beheshtizadeh N, et al. Recent advances on 3D-printed PCL-based composite scaffolds for bone tissue engineering. Front Bioeng Biotechnol. 2023;11:1168504. doi: 10.3389/fbioe.2023.1168504
- Sun J, Chen C, Zhang B, Yao C, Zhang Y. Advances in 3D-printed scaffold technologies for bone defect repair: Materials, biomechanics, and clinical prospects. Biomed Eng Online. 2025;24:51. doi: 10.1186/s12938-025-01381-w
- Bellen F, Carbone E, Baatsen P, Jones EAV, Kabirian F, Heying R. Improvement of endothelial cell-polycaprolactone interaction through surface modification via aminolysis, hydrolysis, and a combined approach. J Tissue Eng Regener Med. 2023;2023:5590725. doi: 10.1155/2023/5590725
- Farjaminejad S, Farjaminejad R, Hasani M, et al. Advances and challenges in polymer-based scaffolds for bone tissue engineering: a path towards personalized regenerative medicine. Polymers. 2024;16(23):3303. doi: 10.3390/polym16233303
- Ielo I, Calabrese G, De Luca G, Conoci S. Recent advances in hydroxyapatite-based biocomposites for bone tissue regeneration in orthopedics. Int J Mol Sci. 2022;23(17):9721. doi: 10.3390/ijms23179721
- Ye X, Zhang Y, Liu T, et al. Beta-tricalcium phosphate enhanced mechanical and biological properties of 3D-printed polyhydroxyalkanoates scaffold for bone tissue engineering. Int J Biol Macromol. 2022;209:1553-1561. doi: 10.1016/j.ijbiomac.2022.04.056
- Ramanathan M, Shijirbold A, Okui T, et al. In vivo evaluation of bone regenerative capacity of the novel nanobiomaterial: β-tricalcium phosphate polylactic acid-co-glycolide (β-TCP/PLLA/PGA) for use in maxillofacial bone defects. Nanomaterials. 2023;14(1):91. doi: 10.3390/nano14010091
- Wu H, Wei X, Liu Y, et al. Dynamic degradation patterns of porous polycaprolactone/β-tricalcium phosphate composites orchestrate macrophage responses and immunoregulatory bone regeneration. Bioact Mater. 2022;21:595-611. doi: 10.1016/j.bioactmat.2022.07.032
- Ruckh TT, Carroll DA, Weaver JR, Popat KC. Mineralization content alters osteogenic responses of bone marrow stromal cells on hydroxyapatite/polycaprolactone composite nanofiber scaffolds. J Funct Biomater. 2012;3(4):776-798. doi: 10.3390/jfb3040776
- Ghezzi B, Matera B, Meglioli M, et al. Composite PCL scaffold with 70% β-TCP as suitable structure for bone replacement. Int Dent J. 2024;74(6):1220-1232. doi: 10.1016/j.identj.2024.02.013
- Guder C, Gravius S, Burger C, Wirtz DC, Schildberg FA. Osteoimmunology: A current update of the interplay between bone and the immune system. Front Immunol. 2020;11:58. doi: 10.3389/fimmu.2020.00058
- Batool F, Özçelik H, Stutz C, et al. Modulation of immune-inflammatory responses through surface modifications of biomaterials to promote bone healing and regeneration. J Tissue Eng. 2021;12:20417314211041428. doi: 10.1177/20417314211041428
- Wang J, Yuan B, Yin R, Zhang H. Inflammation responses to bone scaffolds under mechanical stimuli in bone regeneration. J Funct Biomater. 2023;14(3):169. doi: 10.3390/jfb14030169
- Wang Y, Wang B, Liu D, et al. Osteoimmunology uncovered: How macrophages and biomaterials revolutionize bone healing. Mater Today Bio. 2026;36:102647. doi: 10.1016/j.mtbio.2025.102647
- Huang S, Zeng A, Yin Q, et al. Progress in immunoregulatory mechanisms during distraction osteogenesis. Front Bioeng Biotechnol. 2025;13:1665192. doi: 10.3389/fbioe.2025.1665192
- Liu W, Cheong N, He Z, Zhang T. Application of hydroxyapatite composites in bone tissue engineering: A review. J Funct Biomater. 2025;16(4):127. doi: 10.3390/jfb16040127
- Yao H, Zhu W, Zhu X, et al. Development of hydroxyapatite/ polycaprolactone composite biomaterials for laser powder bed fusion: Evaluation of powder characteristics, mechanical properties and biocompatibility. Polymers. 2024;16(6):731. doi: 10.3390/polym16060731
- Vajgel A, Mardas N, Farias BC, Petrie A, Cimões R, Donos N. A systematic review on the critical size defect model. Clin Oral Implants Res. 2014;25(8):879-893. doi: 10.1111/clr.12194
- Qi H, Zhang B, Lian F. 3D-printed bioceramic scaffolds for bone defect repair: Bone aging and immune regulation. Front Bioeng Biotechnol. 2025;13:1557203. doi: 10.3389/fbioe.2025.1557203
- Yuqiang W, Ziyan Z, Xuedi S, Chengdong P. Recent progress in immunomodulation-based strategies for bone repair. Regen Ther. 2025;31:101054. doi: 10.1016/j.reth.2025.101054
- Chen M, Chen Y, He H, Zhou X, Chen N. Structure and property evolution of microinjection molded PLA/PCL/ bioactive glass composite. Polymers. 2025;17(7):991. doi: 10.3390/polym17070991
- Li Y, Yu Z, Ai F, et al. Characterization and evaluation of polycaprolactone/hydroxyapatite composite scaffolds with extra surface morphology by cryogenic printing for bone tissue engineering. Mater Des. 2021;205:109712. doi: 10.1016/j.matdes.2021.109712
- Helaehil JV, Lourenço CB, Huang B, et al. In vivo investigation of polymer-ceramic PCL/HA and PCL/β-TCP 3D composite scaffolds and electrical stimulation for bone regeneration. Polymers. 2021;14(1):65. doi: 10.3390/polym14010065
- Ni X, Feng J, Liang M, et al. Enhancing bone repair with β-TCP-based composite scaffolds: A review of design strategies and biological mechanisms. Orthop Res Rev. 2025;17:313-340. doi: 10.2147/ORR.S525959
- Ardeshiriansharifabadi S, Asefnejad A, Azami M. 3D-printed polycaprolactone/nano-hydroxyapatite scaffold coated by zein for controlled release of tetracycline hydrochloride in bone tissue engineering. Results Eng. 2026;29:109540. doi: 10.1016/j.rineng.2026.109540
- Thuaksuban N, Monmaturapoj N, Luntheng T. Effects of polycaprolactone-biphasic calcium phosphate scaffolds on enhancing growth and differentiation of osteoblasts. Bio- Med Mater Eng. 2018;29(2):159-176. doi: 10.3233/BME-171720
- Pogonyalova MY, Popov DY, Vinokurov AY. Intracellular calcium as a regulator of polarization and target reprogramming of macrophages. Int J Mol Sci. 2025;26(24):11901. doi: 10.3390/ijms262411901
- Tollabi M, Poursalehi Z, Mehrafshar P, et al. Insight into the role of integrins and integrins-targeting biomaterials in bone regeneration. Connect Tissue Res. 2024;65(5):343-363. doi: 10.1080/03008207.2024.2396002
- Polini A, Pisignano D, Parodi M, Quarto R, Scaglione S. Osteoinduction of human mesenchymal stem cells by bioactive composite scaffolds without supplemental osteogenic growth factors. PLoS ONE. 2011;6(10):e26211. doi: 10.1371/journal.pone.0026211
- Gou Y, Qi K, Wei Y, Gu Z, Xie H. Advances of calcium phosphate nanoceramics for the osteoinductive potential and mechanistic pathways in maxillofacial bone defect repair. Nano TransMed. 2024;3:100033. doi: 10.1016/j.ntm.2024.100033
- Liang HY, Lee WK, Hsu JT, et al. Polycaprolactone in bone tissue engineering: A comprehensive review of innovations in scaffold fabrication and surface modifications. J Funct Biomater. 2024;15(9):243. doi: 10.3390/jfb15090243
- Kong L, Smith W, Hao D. Overview of RAW264.7 for osteoclastogensis study: phenotype and stimuli. J Cell Mol Med. 2019;23(5):3077-3087. doi: 10.1111/jcmm.14277
- Zhang J, Wu Q, Yin C, et al. Sustained calcium ion release from bioceramics promotes CaSR-mediated M2 macrophage polarization for osteoinduction. J Leukocyte Biol. 2021;110(3):485-496. doi: 10.1002/JLB.3MA0321-739R
- Lv L, Xie Y, Li K, et al. Unveiling the mechanism of surface hydrophilicity-modulated macrophage polarization. Adv Healthcare Mater. 2018;7(19):1800675. doi: 10.1002/adhm.201800675
- Wang M, Chen F, Tang Y, et al. Regulation of macrophage polarization and functional status by modulating hydroxyapatite ceramic micro/nano-topography. Mater Des. 2022;213:110302. doi: 10.1016/j.matdes.2021.110302
- Zhang Y, Cheng X, Jansen JA, Yang F, van den Beucken JJJP. Titanium surfaces characteristics modulate macrophage polarization. Mater Sci Eng C. 2019;95:143-151. doi: 10.1016/j.msec.2018.10.065
- Nascimento Da Conceicao V, Sun Y, Ramachandran K, et al. Resolving macrophage polarization through distinct Ca2+ entry channel that maintains intracellular signaling and mitochondrial bioenergetics. iScience. 2021;24(11):103339. doi: 10.1016/j.isci.2021.103339
- Hu L, Chen W, Qian A, Li YP. Wnt/β-catenin signaling components and mechanisms in bone formation, homeostasis, and disease. Bone Res. 2024;12(1):39. doi: 10.1038/s41413-024-00342-8
- Chen E, Liu G, Zhou X, et al. Concentration-dependent, dual roles of IL-10 in the osteogenesis of human BMSCs via P38/MAPK and NF-κB signaling pathways. FASEB J. 2018;32(9):4917-4929. doi: 10.1096/fj.201701256RRR
- Liu K, Luo X, Lv ZY, et al. Macrophage-derived exosomes promote bone mesenchymal stem cells towards osteoblastic fate through microRNA-21a-5p. Front Bioeng Biotechnol. 2022;9:801432. doi: 10.3389/fbioe.2021.801432
- Kusumbe AP, Ramasamy SK, Adams RH. Coupling of angiogenesis and osteogenesis by a specific vessel subtype in bone. Nature. 2014;507(7492):323-328. doi: 10.1038/nature13145
- Liu X, Zhang P, Gu Y, Guo Q, Liu Y. Type H vessels: Functions in bone development and diseases. Front Cell Dev Biol. 2023;11:1236545. doi: 10.3389/fcell.2023.1236545
- Zhang J, Shi H, Zhang N, Hu L, Jing W, Pan J. Interleukin-4- loaded hydrogel scaffold regulates macrophages polarization to promote bone mesenchymal stem cells osteogenic differentiation via TGF-β1/smad pathway for repair of bone defect. Cell Prolif. 2020;53(10):e12907. doi: 10.1111/cpr.12907

