Taxonomic characterization of bioactive hard coral-dwelling actinobacteria in Fiji

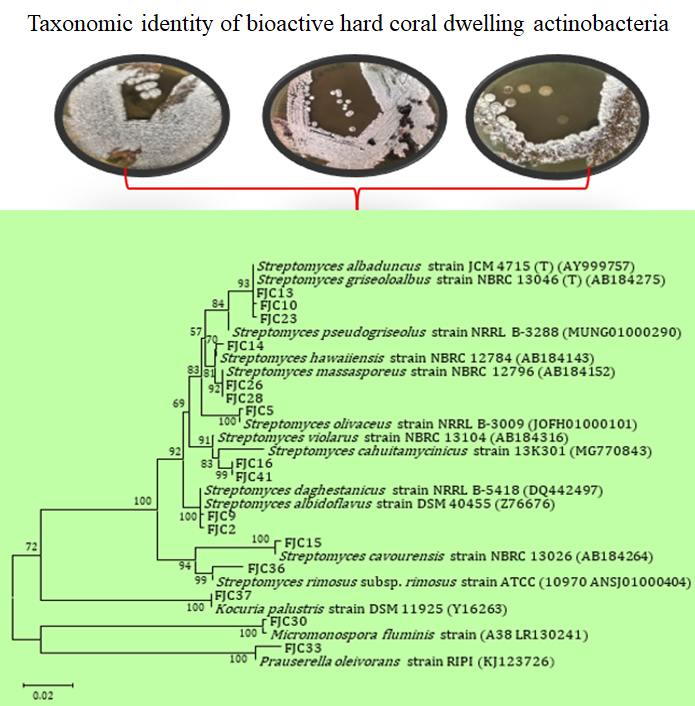
Actinobacteria produce a plethora of bioactive molecules, yet research on these bacteria in the Pacific Island nations remains scarce. Previously, we isolated 16 actinomycete strains from Fijian hard corals, with varying degrees of antimicrobial activity against common pathogenic strains. The present study aims to identify these bioactive actinomycete isolates using 16S ribosomal RNA (16S rRNA) gene sequencing and phylogenetic analysis. The 16S rRNA gene characterization of the bioactive isolates predominantly revealed members of the genus Streptomyces, alongside three rare actinomycete genera: Micromonospora, Prauserella, and Kocuria. Additionally, antiSMASH analysis of the closest phylogenetic relatives of these cultured actinobacteria with notable antimicrobial activity revealed a strong potential for antibiotic biosynthesis. Overall, these findings suggest that Fijian hard corals represent a promising reservoir of bioactive actinobacteria for the discovery of antimicrobial compounds, particularly Streptomyces spp.
- Atanasov AG, Waltenberger B, Pferschy-Wenzig EM, et al. Discovery and resupply of pharmacologically active plant-derived natural products: a review. Biotechnol Adv. 2015;33(8):1582-1614. doi: 10.1016/j.biotechadv.2015.08.001
- Chin YW, Balunas MJ, Chai HB, Kinghorn AD. Drug discovery from natural sources. AAPS J. 2006;8(2):E239-E253. doi: 10.1007/BF02854894
- Siro G, Donald L, Pipite A. The diversity of deep-sea actinobacteria and their natural products: an epitome of curiosity and drug discovery. Diversity. 2023;15(1):30. doi: 10.3390/d15010030
- Siro G, Pipite A, Srinivasan S. Uncovering novel species and bioactive compounds from psychrophilic actinobacteria: A concise review. Microbes Immun. 2025:025350087. doi: 10.36922/MI025350087
- Siro G, Pipite A. Mini-review on the antimicrobial potential of actinobacteria associated with seagrasses. Explor Drug Sci. 2024;2(1):117-125. doi: 10.37349/eds.2024.00038
- Lattanzio V. Relationship of phenolic metabolism to growth in plant and cell cultures under stress. In: Ramawat KG, Ekiert HM, Goyal S, eds. Plant Cell and Tissue Differentiation and Secondary Metabolites: Fundamentals and Applications. Springer International Publishing; 2021:837-868. doi: 10.1007/978-3-030-30185-9_8
- Watve MG, Tickoo R, Jog MM, Bhole BD. How many antibiotics are produced by the genus Streptomyces? Arch Microbiol. 2001;176(5):386-390. doi: 10.1007/s002030100345
- Manivasagan P, Venkatesan J, Sivakumar K, Kim SK. Marine actinobacterial metabolites: current status and future perspectives. Microbiol Res. 2013;168(6):311-332. doi: 10.1016/j.micres.2013.02.002
- Janardhan A, Kumar AP, Viswanath B, Saigopal VD, Narasimha G. Production of bioactive compounds by actinomycetes and their antioxidant properties. Biotechnol Res Int. 2014;2014:1-8. doi: 10.1155/2014/217030
- Zineb A, Ali Z, Ayoub E, Nadia Z, Assia M, Mohammed M. Optimization of culture conditions for growth and secondary metabolites production by Streptomyces sp. MFB27 using response surface methodology. The Microbe. 2025;8:100423. doi: 10.1016/j.microb.2025.100423
- Lane DJ. 16S/23S rRNA sequencing. In: Stackebrandt E, Goodfellow M, eds. Nucleic Acid Techniques in Bacterial Systematics. John Wiley and Sons; 1991:115-175.
- Yoon SH, Ha SM, Kwon S, et al. Introducing EzBioCloud: a taxonomically united database of 16S rRNA gene sequences and whole-genome assemblies. Int J Syst Evol Microbiol. 2017;67(5):1613-1617. doi: 10.1099/ijsem.0.001755
- Tamura K, Stecher G, Kumar S. MEGA11: molecular evolutionary genetics analysis version 11. Mol Biol Evol. 2021;38(7):3022-3027. doi: 10.1093/molbev/msab120
- Blin K, Shaw S, Kloosterman AM, et al. antiSMASH 6.0: improving cluster detection and comparison capabilities. Nucleic Acids Res. 2021;49(1):29-35. doi: 10.1093/nar/gkab335
- Siro G, Pipite A, Christi K, Srinivasan S, Subramani R. Marine actinomycetes associated with stony corals: a potential hotspot for specialized metabolites. Microorganisms. 2022;10(7):1349. doi: 10.3390/microorganisms10071349
- Nithyanand P, Manju S, Pandian SK. Phylogenetic characterization of culturable actinomycetes associated withthe mucus of the coral Acropora digitifera from Gulf of Mannar. FEMS Microbiol Lett. 2011;314(2):112-118. doi: 10.1111/j.1574-6968.2010.02149.x
- Mahmoud HM, Kalendar AA. Coral-associated actinobacteria: diversity, abundance, and biotechnological potentials. Front Microbiol. 2016;7:204. doi: 10.3389/fmicb.2016.00204
- Subramani R, Aalbersberg W. Culturable rare actinomycetes: diversity, isolation and marine natural product discovery. Appl Microbiol Biotechnol. 2013;97(21):9291-9321. doi: 10.1007/s00253-013-5229-7
- Ezeobiora CE, Igbokwe NH, Amin DH, Enwuru NV, Okpalanwa CF, Mendie UE. Uncovering the biodiversity and biosynthetic potentials of rare actinomycetes. Futur J Pharm Sci. 2022;8(1):23.doi: 10.1186/s43094-022-00410-y
- Subramani R, Sipkema D. Marine rare actinomycetes: a promising source of structurally diverse and unique novel natural products. Mar Drugs. 2019;17(5):249. doi: 10.3390/md17050249
- Dastgheib SM, Tirandaz H, Moshtaghi Nikou M, et al. Prauserella oleivorans sp. nov., a halophilic and thermotolerant crude-oil-degrading actinobacterium isolated from an oil-contaminated mud pit. Int J Syst Evol Microbiol. 2017;67(9):3381-3386. doi: 10.1099/ijsem.0.002124
- Saygin H, Ay H, Guven K, Cetin D, Sahin N. Streptomyces cahuitamycinicus sp. nov., isolated from desert soil and reclassification of Streptomyces galilaeus as a later heterotypic synonym of Streptomyces bobili. Int J Syst Evol Microbiol. 2020;70(4):2750-2759. doi: 10.1099/ijsem.0.004103
- Camacho MI, Wieme AD, Rodríguez SP, et al. Micromonospora fluminis sp. nov., isolated from mountain river sediment. Int J Syst Evol Microbiol. 2020;70(12):6428-6436. doi: 10.1099/ijsem.0.004554
- Paul B, Kavia Raj K, Murali TS, Satyamoorthy K. Species-specific genomic sequences for classification of bacteria. Comput Biol Med. 2020;123:103874. doi: 10.1016/j.compbiomed.2020.103874
- Pollock J, Glendinning L, Wisedchanwet T, Watson M. The madness of microbiome: attempting to find consensus “best practice” for 16S microbiome studies. Appl Environ Microbiol. 2018;84(7). doi: 10.1128/aem.02627-17
- Lee N, Hwang S, Kim J, Cho S, Palsson B, Cho BK. Mini review: genome mining approaches for the identification of secondary metabolite biosynthetic gene clusters in Streptomyces. Comput Struct Biotechnol J. 2020;18:1548-1556. doi: 10.1016/j.csbj.2020.06.024
- Ziemert N, Alanjary M, Weber T. The evolution of genome mining in microbes–a review. Nat Prod Rep. 2016;33(8):988-1005. doi: 10.1039/C6NP00025H
- Singh TA, Passari AK, Jajoo A, et al. Tapping into actinobacterial genomes for natural product discovery. Front Microbiol. 2021;12:655620. doi: 10.3389/fmicb.2021.655620
- Undabarrena A, Ugalde J, Seeger M, Cámara B. Genomic data mining of the marine actinobacteria Streptomyces sp. H-KF8 unveils insights into multi-stress related genes and metabolic pathways involved in antimicrobial synthesis. PeerJ. 2017;5:e2912. doi: 10.7717/peerj.2912
- Letzel A, Li J, Amos GC, et al. Genomic insights into specialized metabolism in the marine actinomycete Salinispora. Environ Microbiol. 2017;19(9):3660-3673. doi: 10.1111/1462-2920.13867
- Xu L, Ye KX, Dai WH, Sun C, Xu LH, Han BN. Comparative genomic insights into secondary metabolism biosynthetic gene cluster distributions of marine Streptomyces. Mar Drugs. 2019;17(9):498. doi: 10.3390/md17090498
- Komaki H. Recent progress of reclassification of the genus Streptomyces. Microorganisms. 2023;11(4):831. doi: 10.3390/microorganisms11040831
- Chitraselvi RPE, Kalidass S. Biocontrol activity of Streptomyces violarus bioformulation against leaf blight disease of groundnut. Trends Biosci. 2017;10(39):8249-8251.
- Therdkiattikul N, Ratpukdi T, Kidkhunthod P, ChanlekN, Siripattanakul-Ratpukdi S. Manganese-contaminated groundwater treatment by novel bacterial isolates: kinetic study and mechanism analysis using synchrotron-based techniques. Sci Rep. 2020;10(1):13391. doi: 10.1038/s41598-020-70355-w
- Lekhak B, Singh A, Bhatta DR. Antibacterial and antifungal property of actinomycetes isolates from soil and water of Nepal. J Nepal Health Res Counc. 2018;16(2):136-139. doi: 10.3126/jnhrc.v16i2.20298
- Kumar V, Bisht GS, Gusain O. Terrestrial actinomycetes from diverse locations of Uttarakhnad, India: Isolation and screening for their antibacterial activity. Iran J Microbiol. 2013;5(3):299-308.
- Sekurova ON, Schneider O, Zotchev SB. Novel bioactive natural products from bacteria via bioprospecting, genome mining and metabolic engineering. Microb Biotechnol. 2019;12(5):828-844. doi: 10.1111/1751-7915.13398

