Advancing wound healing with three-dimensional bioprinted hyaluronic acid-based tissue constructs: From mechanistic insights to clinical translation

Skin functions as a primary protective barrier against mechanical injury, microbial invasion, and dehydration. Extensive trauma and chronic diseases pose significant clinical challenges to wound healing. Conventional wound dressings and skin substitutes often lack the structural and biochemical sophistication to dynamically interact with the wound. Hyaluronic acid (HA), a key glycosaminoglycan in the extracellular matrix, has emerged as a versatile biomaterial for wound repair due to its biocompatibility, hydration capacity, and intrinsic bioactivity. However, native HA suffers from limitations such as rapid degradation and poor mechanical strength, necessitating advanced engineering strategies, including chemical modification, biofunctionalization, and compositing, to enhance its versatility. Three-dimensional bioprinting has recently emerged as a transformative technology, enabling the precise deposition of HA-based biomaterials to form biomimetic constructs with spatial heterogeneity. In this review, we first elucidate the mechanism by which HA orchestrates the wound healing cascade, followed by a list of engineered approaches to enhance HA’s functionality. This review then focuses on the capabilities of mainstream bioprinting technologies for fabricating HA-based wound dressings and stratified skin substitutes. Finally, we discuss prevailing challenges and outline future perspectives, emphasizing innovations in dynamic biomaterials, hybrid bioprinting strategies, and the integration of artificial intelligence to advance the clinical translation of HA products for wound healing.
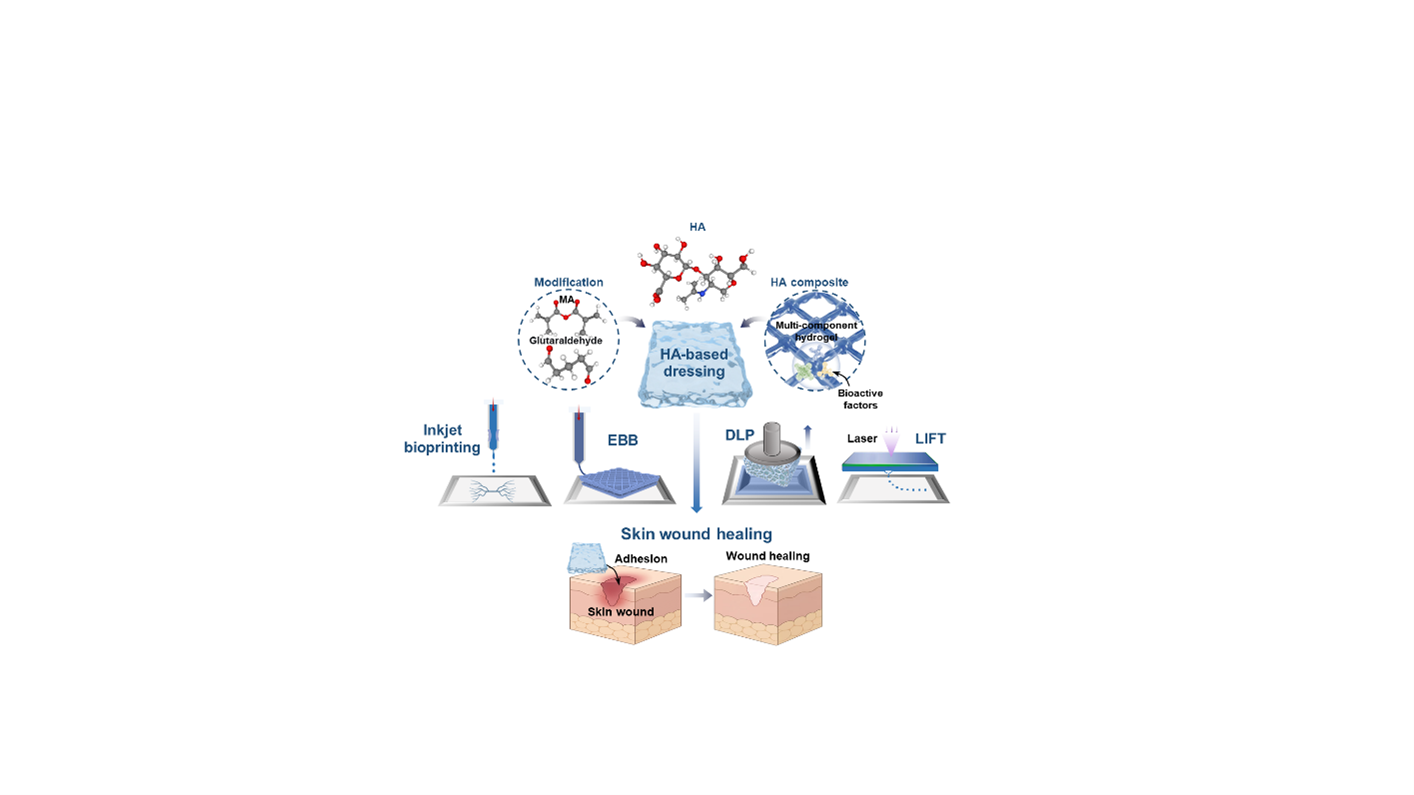
- Deepa C, Bhatt A. Skin substitutes: from conventional to 3D bioprinting. J Artif Organs. 2025;28(2):154-170. doi: 10.1007/s10047-024-01481-9
- Farahani M, Shafiee A. Wound Healing: From Passive to Smart Dressings. Adv Healthc Mater. 2021;10(16):2100477. doi: 10.1002/adhm.202100477
- Peña OA, Martin P. Cellular and molecular mechanisms of skin wound healing. Nat Rev Mol Cell Biol. 2024;25(8):599-616.
doi: 10.1038/s41580-024-00715-1
- Kolimi P, Narala S, Nyavanandi D, Youssef AAA, Dudhipala N. Innovative Treatment Strategies to Accelerate Wound Healing: Trajectory and Recent Advancements. Cells. 2022;11(15):2439. doi: 10.3390/cells11152439
- Aoyu C, Yibing W. Advances in the Study of the Etiology and Pathogenesis of Chronic Wounds. ACM. 2023;13(03):2958- 2966. [In Chinese] doi: 10.12677/ACM.2023.133419.
- Pradhan L, Nabzdyk C, Andersen ND, LoGerfo FW, Veves A. Inflammation and neuropeptides: the connection in diabetic wound healing. Expert Rev Mol Med. 2009;11:e2. doi: 10.1017/S1462399409000945
- Sen CK. Human Wound and Its Burden: Updated 2020 Compendium of Estimates. Adv Wound Care. 2021;10(5):281-292. doi: 10.1089/wound.2021.0026
- Zhou X, Yu X, You T, et al. 3D Printing‐Based Hydrogel Dressings for Wound Healing. Adv Sci. 2024;11(47):2404580. doi: 10.1002/advs.202404580
- Liang Y, He J, Guo B. Functional Hydrogels as Wound Dressing to Enhance Wound Healing. ACS Nano. 2021;15(8):12687-12722. doi: 10.1021/acsnano.1c04206
- Chylińska N, Maciejczyk M. Hyaluronic Acid and Skin: Its Role in Aging and Wound-Healing Processes. Gels. 2025;11(4):281. doi: 10.3390/gels11040281
- Shang L, Li M, Xu A, Zhuo F. Recent applications and molecular mechanisms of hyaluronic acid in skin aging and wound healing. Med Nov Technol. Devices. 2024;23:100320. doi: 10.1016/j.medntd.2024.100320
- Antoszewska M, Sokolewicz EM, Barańska-Rybak W. Wide Use of Hyaluronic Acid in the Process of Wound Healing—A Rapid Review. Sci Pharm. 2024;92(2):23. doi: 10.3390/scipharm92020023
- Iaconisi GN, Lunetti P, Gallo N, et al. Hyaluronic Acid: A Powerful Biomolecule with Wide-Ranging Applications—A Comprehensive Review. Int J Mol Sci. 2023;24(12):10296. doi: 10.3390/Int J Mol Sci241210296
- Salih ARC, Farooqi HMU, Amin H, Karn PR, Meghani N, Nagendran S. Hyaluronic acid: comprehensive review of a multifunctional biopolymer. Futur J Pharm Sci. 2024;10(1):63. doi: 10.1186/s43094-024-00636-y
- Graça MFP, Miguel SP, Cabral CSD, Correia IJ. Hyaluronic acid—Based wound dressings: A review. Carbohydr Polym. 2020;241(8):116364. doi: 10.1016/j.carbpol.2020.116364
- Dereure O, Mikosinki J, Zegota Z, Allaert FA. RCT to evaluate a hyaluronic acid containing gauze pad in leg ulcers of venous or mixed aetiology. J Wound Care. 2012;21(11):539-547. doi: 10.12968/jowc.2012.21.11.539
- Humbert P, Mikosinki J, Benchikhi H, Allaert F. Efficacy and safety of a gauze pad containing hyaluronic acid in treatment of leg ulcers of venous or mixed origin: a double‐blind, randomised, controlled trial. Int Wound J. 2013;10(2):159- 166. doi: 10.1111/j.1742-481X.2012.00957.x
- Price RD, Berry MG, Navsaria HA. Hyaluronic acid: the scientific and clinical evidence. J Plast Reconstr Aesthetic Surg. 2007;60(10):1110-1119. doi: 10.1016/j.bjps.2007.03.005
- Simińska‐Stanny J, Podstawczyk D, Delporte C, Nie L, Shavandi A. Hyaluronic Acid Role in Biomaterials Prevascularization. Adv Healthcare Mater. 2024;13(30):2402045. doi: 10.1002/adhm.202402045
- Sekar MP, Suresh S, Zennifer A, Sethuraman S, Sundaramurthi D. Hyaluronic Acid as Bioink and Hydrogel Scaffolds for Tissue Engineering Applications. ACS Biomater Sci Eng. 2023;9(6):3134-3159. doi: 10.1021/acsbiomaterials.3c00299
- Luo Z, Wang Y, Xu Y, Wang J, Yu Y. Modification and crosslinking strategies for hyaluronic acid‐based hydrogel biomaterials. Smart Med. 2023;2(4):e20230029. doi: 10.1002/SMMD.20230029
- Ye H, Zhang R, Zhang C, Xia Y, Jin L. Advances in hyaluronic acid: Bioactivity, complexed biomaterials and biological application: A review. Asian J Surg. 2025;48(1):49-61. doi: 10.1016/j.asjsur.2024.08.100
- Gómez-Guillén MC. Functional and bioactive properties of collagen and gelatin from alternative sources: A review. Food Hydrocoll. 2011;25:1813-1827. doi: 10.1016/j.foodhyd.2011.02.007
- Kamoun EA, Kenawy ERS, Chen X. A review on polymeric hydrogel membranes for wound dressing applications: PVA-based hydrogel dressings. J Adv Res. 2017;8(3):217-233. doi: 10.1016/j.jare.2017.01.005
- Altunbek M, Afghah F, Caliskan OS, Yoo JJ, Koc B. Design and bioprinting for tissue interfaces. Biofabrication. 2023;15(2):022002. doi: 10.1088/1758-5090/acb73d
- Daly AC, Prendergast ME, Hughes AJ, Burdick JA. Bioprinting for the Biologist. Cell. 2021;184(1):18-32. doi: 10.1016/j.cell.2020.12.002
- Li X, Liu B, Pei B, et al. Inkjet Bioprinting of Biomaterials. Chem Rev. 2020;120(19):10793-10833. doi: 10.1021/acs.chemrev.0c00008
- Cheng J, Yu S, Wang R, Ge Q. Digital light processing based multimaterial 3D printing: challenges, solutions and perspectives. Int J Extrem Manuf. 2024;6(4):042006. doi: 10.1088/2631-7990/ad4a2c
- Askari M, Afzali Naniz M, Kouhi M, Saberi A, Zolfagharian A, Bodaghi M. Recent progress in extrusion 3D bioprinting of hydrogel biomaterials for tissue regeneration: a comprehensive review with focus on advanced fabrication techniques. Biomater Sci. 2021;9(3):535-573. doi: 10.1039/D0BM00973C
- Fraser JRE, Laurent TC, Laurent UBG. Hyaluronan: its nature, distribution, functions and turnover. J Intern Med. 1997;242(1):27-33. doi: 10.1046/j.1365-2796.1997.00170.x
- Tammi MI, Day AJ, Turley EA. Hyaluronan and Homeostasis: A Balancing Act. J Biol Chem. 2002;277(7):4581-4584. doi: 10.1074/jbc.R100037200
- Weigel PH, DeAngelis PL. Hyaluronan Synthases: A Decade-plus of Novel Glycosyltransferases. J Biol Chem. 2007;282(51):36777-36781. doi: 10.1074/jbc.R700036200
- Itano N, Sawai T, Yoshida M, et al. Three Isoforms of Mammalian Hyaluronan Synthases Have Distinct Enzymatic Properties. J Biol Chem. 1999;274(35):25085-25092. doi: 10.1074/jbc.274.35.25085
- Vigetti D, Karousou E, Viola M, Deleonibus S, De Luca G, Passi A. Hyaluronan: Biosynthesis and signaling. Biochim Biophys Acta (BBA)-Gen Subj. 2014;1840(8):2452-2459. doi: 10.1016/j.bbagen.2014.02.001
- Tammi RH, Kultti A, Kosma VM, Pirinen R, Auvinen P, Tammi MI. Hyaluronan in human tumors: Pathobiological and prognostic messages from cell-associated and stromal hyaluronan. Semin Cancer Biol. 2008;18(4):288-295. doi: 10.1016/j.semcancer.2008.03.005
- Stern R, Jedrzejas MJ. Hyaluronidases: Their Genomics, Structures, and Mechanisms of Action. Chem Rev. 2006;106(3):818-839. doi: 10.1021/cr050247k
- Chen L, Bourguignon LYW. Hyaluronan-CD44 interaction promotes c-Jun signaling and miRNA21 expression leading to Bcl-2 expression and chemoresistance in breast cancer cells. Mol Cancer. 2014;13(1):52. doi: 10.1186/1476-4598-13-52
- Csoka AB, Frost GI, Stern R. The six hyaluronidase-like genes in the human and mouse genomes. Matrix Biol. 2001;20(8):499-508. doi: 10.1016/S0945-053X(01)00172-X
- Harada H, Takahashi M. CD44-dependent Intracellular and Extracellular Catabolism of Hyaluronic Acid by Hyaluronidase-1 and -2. J Biol Chem. 2007;282(8):5597- 5607. doi: 10.1074/jbc.M608358200
- Monzon ME, Fregien N, Schmid N, et al. Reactive Oxygen Species and Hyaluronidase 2 Regulate Airway Epithelial Hyaluronan Fragmentation. J Biol Chem. 2010;285(34):26126-26134. doi: 10.1074/jbc.M110.135194
- Jiang D, Liang J, Noble PW. Hyaluronan as an Immune Regulator in Human Diseases. Physiol Rev. 2011;91(1):221- 264. doi: 10.1152/physrev.00052.2009
- Tavianatou AG, Caon I, Franchi M, Piperigkou Z, Galesso D, Karamanos NK. Hyaluronan: molecular size‐dependent signaling and biological functions in inflammation and cancer. FEBS J. 2019;286(15):2883-2908. doi: 10.1111/febs.14777
- Snetkov P, Zakharova K, Morozkina S, Olekhnovich R, Uspenskaya M. Hyaluronic Acid: The Influence of Molecular Weight on Structural, Physical, Physico-Chemical, and Degradable Properties of Biopolymer. Polymers. 2020;12(8):1800. doi: 10.3390/polym12081800
- Fallacara A, Baldini E, Manfredini S, Vertuani S. Hyaluronic Acid in the Third Millennium. Polymers. 2018;10(7):701. doi: 10.3390/polym10070701
- Stern R, Asari AA, Sugahara KN. Hyaluronan fragments: An information-rich system. Eur J Cell Biol. 2006;85(8):699- 715. doi: 10.1016/j.ejcb.2006.05.009
- Cowman MK, Schmidt TA, Raghavan P, Stecco A. Viscoelastic Properties of Hyaluronan in Physiological Conditions. F1000Res. 2015;4:622. doi: 10.12688/f1000research.6885.1
- Chellaiah MA. CD44: A Multifunctional Cell Surface Adhesion Receptor Is a Regulator of Progression and Metastasis of Cancer Cells. Front Cell Dev Biol. 2017;5. doi: 10.3389/fcell.2017.00018
- Mattheolabakis G, Milane L, Singh A, Amiji MM. Hyaluronic acid targeting of CD44 for cancer therapy: from receptor biology to nanomedicine. J Drug Target. 2015;23(7-8):605- 618. doi: 10.3109/1061186X.2015.1052072
- Lee BM, Park SJ, Noh I, Kim CH. The effects of the molecular weights of hyaluronic acid on the immune responses. Biomater Res. 2021;25(1):27. doi: 10.1186/s40824-021-00228-4
- Jou I, Wu T, Hsu C, et al. High molecular weight form of hyaluronic acid reduces neuroinflammatory response in injured sciatic nerve via the intracellular domain of CD44. J Biomed Mater Res. 2021;109(5):673-680. doi: 10.1002/jbm.b.34731
- Slevin M, Krupinski J, Gaffney J, et al. Hyaluronan-mediated angiogenesis in vascular disease: Uncovering RHAMM and CD44 receptor signaling pathways. Matrix Biol. 2007;26(1):58-68. doi: 10.1016/j.matbio.2006.08.261
- Yang B, Guo X, Zang H, Liu J. Determination of modification degree in BDDE-modified hyaluronic acid hydrogel by SEC/ MS. Carbohydr Polym. 2015;131:233-239. doi: 10.1016/j.carbpol.2015.05.050
- Šoltés L, Mendichi R, Kogan G, Schiller J, Stankovská M, Arnhold J. Degradative Action of Reactive Oxygen Species on Hyaluronan. Biomacromolecules. 2006;7(3):659-668. doi: 10.1021/bm050867v
- Bourguignon LYW, Wong G, Earle CA, Xia W. Interaction of low molecular weight hyaluronan with CD44 and toll‐like receptors promotes the actin filament‐associated protein 110‐actin binding and MyD88‐NFκB signaling leading to proinflammatory cytokine/chemokine production and breast tumor invasion. Cytoskeleton. 2011;68(12):671-693. doi: 10.1002/cm.20544
- Gao F, Yang CX, Mo W, Liu YW, He YQ. Hyaluronan oligosaccharides are potential stimulators to angiogenesis via RHAMM mediated signal pathway in wound healing. Clin Investig Med. 2008;31(3):E106-E116. doi: 10.25011/cim.v31i3.3467
- Kouvidi K, Berdiaki A, Nikitovic D, et al. Role of Receptor for Hyaluronic Acid-mediated Motility (RHAMM) in Low Molecular Weight Hyaluronan (LMWHA)- mediated Fibrosarcoma Cell Adhesion. J Biol Chem. 2011;286(44):38509-38520. doi: 10.1074/jbc.M111.275875
- Webber J, Jenkins RH, Meran S, Phillips A, Steadman R. Modulation of TGFβ1-Dependent Myofibroblast Differentiation by Hyaluronan. Am J Pathol. 2009;175(1):148- 160. doi: 10.2353/ajpath.2009.080837
- Frenkel JS. The role of hyaluronan in wound healing. Int Wound J. 2014;11(2):159-163. doi: 10.1111/j.1742-481X.2012.01057.x
- Dicker KT, Gurski LA, Pradhan-Bhatt S, Witt RL, Farach- Carson MC, Jia X. Hyaluronan: A simple polysaccharide with diverse biological functions. Acta Biomater. 2014;10(4):1558-1570. doi: 10.1016/j.actbio.2013.12.019
- Aya KL, Stern R. Hyaluronan in wound healing: Rediscovering a major player. Wound Repair Regen. 2014;22(5):579-593. doi: 10.1111/wrr.12214
- Kawano Y, Patrulea V, Sublet E, et al. Wound Healing Promotion by Hyaluronic Acid: Effect of Molecular Weight on Gene Expression and In Vivo Wound Closure. Pharmaceuticals. 2021;14(4):301. doi: 10.3390/ph14040301
- Gallo N, Nasser H, Salvatore L, et al. Hyaluronic acid for advanced therapies: Promises and challenges. Eur Polym J. 2019;117:134-147. doi: 10.1016/j.eurpolymj.2019.05.007
- McKeown-Longo PJ, Higgins PJ. Hyaluronan, Transforming Growth Factor β, and Extra Domain A-Fibronectin: A Fibrotic Triad. Adv Wound Care. 2021;10(3):137-152. doi: 10.1089/wound.2020.1192
- Papakonstantinou E, Roth M, Karakiulakis G. Hyaluronic acid: A key molecule in skin aging. Derm.-Endocrinol. 2012;4(3):253-258. doi: 10.4161/derm.21923
- Schneider LA, Korber A, Grabbe S, Dissemond J. Influence of pH on wound-healing: a new perspective for wound-therapy? Arch Dermatol Res. 2007;298(9):413-420. doi: 10.1007/s00403-006-0713-x
- Eke G, Mangir N, Hasirci N, MacNeil S, Hasirci V. Development of a UV crosslinked biodegradable hydrogel containing adipose derived stem cells to promote vascularization for skin wounds and tissue engineering. Biomaterials. 2017;129:188-198. doi: 10.1016/j.biomaterials.2017.03.021
- Lee HJ, Fernandes-Cunha GM, Myung D. In situ-forming hyaluronic acid hydrogel through visible light-induced thiol-ene reaction. React Funct Polym. 2018;131:29-35. doi: 10.1016/j.reactfunctpolym.2018.06.010
- Hwang HS, Lee CS. Recent Progress in Hyaluronic-Acid- Based Hydrogels for Bone Tissue Engineering. Gels. 2023;9(7):588. doi: 10.3390/gels9070588
- Maloney FP, Kuklewicz J, Corey RA, et al. Structure, substrate recognition and initiation of hyaluronan synthase. Nature. 2022;604(7904):195-201. doi: 10.1038/s41586-022-04534-2
- Bencherif SA, Srinivasan A, Horkay F, Hollinger JO, Matyjaszewski K, Washburn NR. Influence of the degree of methacrylation on hyaluronic acid hydrogels properties. Biomaterials. 2008;29(12):1739-1749. doi: 10.1016/j.biomaterials.2007.11.047
- Stern R, Maibach HI. Hyaluronan in skin: aspects of aging and its pharmacologic modulation. Clin Dermatol. 2008;26(2):106-122. doi: 10.1016/j.clindermatol.2007.09.013
- Girish KS, Kemparaju K. The magic glue hyaluronan and its eraser hyaluronidase: A biological overview. Life Sci. 2007;80(21):1921-1943. doi: 10.1016/j.lfs.2007.02.037
- Zhao R, Liang H, Clarke E, Jackson C, Xue M. Inflammation in Chronic Wounds. Int J Mol Sci. 2016;17(12):2085. doi: 10.3390/Int J Mol Sci17122085
- Dovedytis M, Liu ZJ, Bartlett S. Hyaluronic acid and its biomedical applications: A review. Eng Regen. 2020;1:102- 113. doi: 10.1016/j.engreg.2020.10.001
- Khunmanee S, Jeong Y, Park H. Crosslinking method of hyaluronic-based hydrogel for biomedical applications. J Tissue Eng. 2017;8:2041731417726464. doi: 10.1177/2041731417726464
- Hou X, Zhong D, Chen H, et al. Recent advances in hyaluronic acid-based nanomedicines: Preparation and application in cancer therapy. Carbohydr Polym. 2022;292:119662. doi: 10.1016/j.carbpol.2022.119662
- Burdick JA, Prestwich GD. Hyaluronic Acid Hydrogels for Biomedical Applications. Adv Mater. 2011;23(12). doi: 10.1002/adma.201003963
- Xu Q, Torres JE, Hakim M, et al. Collagen- and hyaluronic acid-based hydrogels and their biomedical applications. Mater Sci Eng R Rep. 2021;146:100641. doi: 10.1016/j.mser.2021.100641
- Bermejo-Velasco D, Azémar A, Oommen OP, Hilborn J, Varghese OP. Modulating Thiol p Ka Promotes Disulfide Formation at Physiological pH: An Elegant Strategy To Design Disulfide Cross-Linked Hyaluronic Acid Hydrogels. Biomacromolecules. 2019;20(3):1412-1420. doi: 10.1021/acs.biomac.8b01830
- Dheer D, Gupta R, Singh D, et al. Hyaluronic Acid- Tacrolimus Bioconjugate: Synthesis, Characterization, and Pharmacokinetic Investigation of an Acid-Responsive Macromolecular Prodrug. ACS Appl Bio Mater. 2019;2(11):4728-4736. doi: 10.1021/acsabm.9b00423
- Crescenzi V, Cornelio L, Di Meo C, Nardecchia S, Lamanna R. Novel Hydrogels via Click Chemistry: Synthesis and Potential Biomedical Applications. Biomacromolecules. 2007;8(6):1844-1850. doi: 10.1021/bm0700800
- Fu S, Dong H, Deng X, Zhuo R, Zhong Z. Injectable hyaluronic acid/poly(ethylene glycol) hydrogels crosslinked via strain-promoted azide-alkyne cycloaddition click reaction. Carbohydr Polym. 2017;169:332-340. doi: 10.1016/j.carbpol.2017.04.028
- Zhang W, Mu H, Dong D, Wang D, Zhang A, Duan J. Alteration in immune responses toward N-deacetylation of hyaluronic acid. Glycobiology. 2014;24(12):1334-1342. doi: 10.1093/glycob/cwu079
- Sedláček J, Hermannová M, Šatínský D, Velebný V. Current analytical methods for the characterization of N-deacetylated hyaluronan: A critical review. Carbohydr Polym. 2020;249:116720. doi: 10.1016/j.carbpol.2020.116720
- Luo Y, Tan J, Zhou Y, et al. From crosslinking strategies to biomedical applications of hyaluronic acid-based hydrogels: A review. Int J Biol Macromol. 2023;231:123308. doi: 10.1016/j.ijbiomac.2023.123308
- Huang Q, Zou Y, Arno MC, et al. Hydrogel scaffolds for differentiation of adipose-derived stem cells. Chem Soc Rev. 2017;46(20):6255-6275. doi: 10.1039/C6CS00052E
- Lu HD, Charati MB, Kim IL, Burdick JA. Injectable shear-thinning hydrogels engineered with a self-assembling Dock-and-Lock mechanism. Biomaterials. 2012;33(7):2145-2153. doi: 10.1016/j.biomaterials.2011.11.076
- Jung Y seok, Park W, Park H, Lee DK, Na K. Thermo-sensitive injectable hydrogel based on the physical mixing of hyaluronic acid and Pluronic F-127 for sustained NSAID delivery. Carbohydr Polym. 2017;156:403-408. doi: 10.1016/j.carbpol.2016.08.068
- Miller B, Hansrisuk A, Highley CB, Caliari SR. Guest– Host Supramolecular Assembly of Injectable Hydrogel Nanofibers for Cell Encapsulation. ACS Biomater Sci Eng. 2021;7(9):4164-4174. doi: 10.1021/acsbiomaterials.1c00275
- Rodell CB, Kaminski AL, Burdick JA. Rational Design of Network Properties in Guest–Host Assembled and Shear- Thinning Hyaluronic Acid Hydrogels. Biomacromolecules. 2013;14(11):4125-4134. doi: 10.1021/bm401280z
- Kim J, Lee C, Ryu JH. Adhesive Catechol-Conjugated Hyaluronic Acid for Biomedical Applications: A Mini Review. Appl Sci. 2020;11(1):21. doi: 10.3390/app11010021
- An S, Choi S, Min S, Cho SW. Hyaluronic Acid-based Biomimetic Hydrogels for Tissue Engineering and Medical Applications. Biotechnol Bioproc E. 2021;26(4):503-516. doi: 10.1007/s12257-020-0343-8
- Chaudhary R, Fabbri P, Leoni E, Mazzanti F, Akbari R, Antonini C. Additive manufacturing by digital light processing: a review. Prog Addit Manuf. 2023;8(2):331-351. doi: 10.1007/s40964-022-00336-0
- Pérez LA, Hernández R, Alonso JM, Pérez-González R, Sáez-Martínez V. Hyaluronic Acid Hydrogels Crosslinked in Physiological Conditions: Synthesis and Biomedical Applications. Biomedicines. 2021;9(9):1113. doi: 10.3390/biomedicines9091113
- Xu Q, A S, Gao Y, et al. A hybrid injectable hydrogel from hyperbranched PEG macromer as a stem cell delivery and retention platform for diabetic wound healing. Acta Biomater. 2018;75:63-74. doi: 10.1016/j.actbio.2018.05.039
- Kurisawa M, Lee F, Wang LS, Chung JE. Injectable enzymatically crosslinked hydrogel system with independent tuning of mechanical strength and gelation rate for drug delivery and tissue engineering. J Mater Chem. 2010;20(26):5371. doi: 10.1039/b926456f
- Rizwan M, Baker AEG, Shoichet MS. Designing Hydrogels for 3D Cell Culture Using Dynamic Covalent Crosslinking. Adv Healthcare Mater. 2021;10(12):2100234. doi: 10.1002/adhm.202100234
- Shi W, Hass B, Kuss MA, et al. Fabrication of versatile dynamic hyaluronic acid-based hydrogels. Carbohydr Polym. 2020;233:115803. doi: 10.1016/j.carbpol.2019.115803
- Kim DY, Park H, Kim SW, Lee JW, Lee KY. Injectable hydrogels prepared from partially oxidized hyaluronate and glycol chitosan for chondrocyte encapsulation. Carbohydr Polym. 2017;157:1281-1287. doi: 10.1016/j.carbpol.2016.11.002
- Yang X, Wang B, Peng D, et al. Hyaluronic Acid‐Based Injectable Hydrogels for Wound Dressing and Localized Tumor Therapy: A Review. Adv NanoBiomed Res. 2022;2(12):2200124. doi: 10.1002/anbr.202200124
- Nimmo CM, Owen SC, Shoichet MS. Diels−Alder Click Cross-Linked Hyaluronic Acid Hydrogels for Tissue Engineering. Biomacromolecules. 2011;12(3):824-830. doi: 10.1021/bm101446k
- Wang G, Cao X, Dong H, Zeng L, Yu C, Chen X. A Hyaluronic Acid Based Injectable Hydrogel Formed via Photo-Crosslinking Reaction and Thermal-Induced Diels- Alder Reaction for Cartilage Tissue Engineering. Polymers. 2018;10(9):949. doi: 10.3390/polym10090949
- Famili A, Rajagopal K. Bio-Orthogonal Cross-Linking Chemistry Enables In Situ Protein Encapsulation and Provides Sustained Release from Hyaluronic Acid Based Hydrogels. Mol Pharm. 2017;14(6):1961-1968. doi: 10.1021/acs.molpharmaceut.7b00067
- Tarus D, Hachet E, Messager L, Catargi B, Ravaine V, Auzély‐Velty R. Readily Prepared Dynamic Hydrogels by Combining Phenyl Boronic Acid‐ and Maltose‐Modified Anionic Polysaccharides at Neutral pH. Macromol Rapid Commun. 2014;35(24):2089-2095. doi: 10.1002/marc.201400477
- Jin Y, Yu C, Denman RJ, Zhang W. Recent advances in dynamic covalent chemistry. Chem Soc Rev. 2013;42(16):6634. doi: 10.1039/c3cs60044k
- Liu J, Willför S, Xu C. A review of bioactive plant polysaccharides: Biological activities, functionalization, and biomedical applications. Bioact Carbohydr Diet Fibre. 2015;5(1):31-61. doi: 10.1016/j.bcdf.2014.12.001
- Zamboni F, Wong CK, Collins MN. Hyaluronic acid association with bacterial, fungal and viral infections: Can hyaluronic acid be used as an antimicrobial polymer for biomedical and pharmaceutical applications? Bioact Mater. 2023;19:458-473. doi: 10.1016/j.bioactmat.2022.04.023
- Conrozier T. Impact of the Skin Disinfection with Quaternary Ammonium Salts on the Rheological Properties of Hyaluronic Acid Viscosupplements. BJSTR. 2021;35(4). doi: 10.26717/BJSTR.2021.35.005728
- Arpicco S, Milla P, Stella B, Dosio F. Hyaluronic Acid Conjugates as Vectors for the Active Targeting of Drugs, Genes and Nanocomposites in Cancer Treatment. Molecules. 2014;19(3):3193-3230. doi: 10.3390/molecules19033193
- Zamboni F, Okoroafor C, Ryan MP, et al. On the bacteriostatic activity of hyaluronic acid composite films. Carbohydr Polym. 2021;260:117803. doi: 10.1016/j.carbpol.2021.117803
- Kaplan JB, Sukhishvili SA, Sailer M, Kridin K, Ramasubbu N. Aggregatibacter actinomycetemcomitans Dispersin B: The Quintessential Antibiofilm Enzyme. Pathogens. 2024;13(8):668. doi: 10.3390/pathogens13080668
- Hunt M, Torres M, Bachar-Wikstrom E, Wikstrom JD. Cellular and molecular roles of reactive oxygen species in wound healing. Commun Biol. 2024;7(1):1534. doi: 10.1038/s42003-024-07219-w
- Wong RSY, Tan T, Pang ASR, Srinivasan DK. The role of cytokines in wound healing: from mechanistic insights to therapeutic applications. Explor Immunol. 2025;5:1003183. doi: 10.37349/ei.2025.1003183
- Lu Y, Kang W, Yu Y, et al. A synergistically antimicrobial and antioxidant hyaluronic acid hydrogel for infected wounds. Int J Biol Macromol. 2024;269:131795. doi: 10.1016/j.ijbiomac.2024.131795
- Park HJ, Jin Y, Shin J, et al. Catechol-Functionalized Hyaluronic Acid Hydrogels Enhance Angiogenesis and Osteogenesis of Human Adipose-Derived Stem Cells in Critical Tissue Defects. Biomacromolecules. 2016;17(6):1939- 1948. doi: 10.1021/acs.biomac.5b01670
- Senobari F, Abolmaali SS, Farahavr G, Tamaddon AM. Targeting inflammation with hyaluronic acid-based micro-and nanotechnology: A disease-oriented review. Int J Biol Macromol. 2024;280:135923. doi: 10.1016/j.ijbiomac.2024.135923
- Xiao L, Tong Z, Chen Y, Pochan DJ, Jia X. Hyaluronic Acid-Based Hydrogels Containing Covalently Integrated Drug Depots: Implication for Controlling Inflammation in Mechanically Stressed Tissues. Biomacromolecules. 2014;14(11). doi: 10.1021/bm4011276
- Gallagher LB, Dolan EB, O’Sullivan J, et al. Pre-culture of mesenchymal stem cells within RGD-modified hyaluronic acid hydrogel improves their resilience to ischaemic conditions. Acta Biomater. 2020;107:78-90. doi: 10.1016/j.actbio.2020.02.043
- Shu XZ, Ghosh K, Liu Y, et al. Attachment and spreading of fibroblasts on an RGD peptide–modified injectable hyaluronan hydrogel. J Biomed Mater Res. 2004;68A(2):365- 375. doi: 10.1002/jbm.a.20002
- Lu Q, Tang X, Tao B, et al. Multifunctional hyaluronic acid microneedle patch enhances diabetic wound healing in diabetic infections. Int J Biol Macromol. 2025;296:139685. doi: 10.1016/j.ijbiomac.2025.139685
- Perera TH, Lu X, Smith Callahan LA. Effect of Laminin Derived Peptides IKVAV and LRE Tethered to Hyaluronic Acid on hiPSC Derived Neural Stem Cell Morphology, Attachment and Neurite Extension. J Funct Biomater. 2020;11(1):15. doi: 10.3390/jfb11010015
- Lu D, Cai K, Zeng Z, et al. VEGF loading heparinized hyaluronic acid macroporous hydrogels for enhanced 3D endothelial cell migration and vascularization. Biomater Adv. 2025;167:214094.doi: 10.1016/j.bioadv.2024.214094
- Qin X, Yuan H, Zhang M, et al. Controlled platelet-derived growth factor delivery by hyaluronic acid hydrogels with tunable crosslinking for accelerated chronic wound healing. Int J Biol Macromol. 2025;304:140710. doi: 10.1016/j.ijbiomac.2025.140710
- Lee SY, Park Y, Hwang SJ. Effect of bFGF and fibroblasts combined with hyaluronic acid-based hydrogels on soft tissue augmentation: an experimental study in rats. Maxillofac Plast Reconstr Surg. 2019;41(1):47. doi: 10.1186/s40902-019-0234-0
- Ostróżka-Cieślik A, Tanwar A, Michalak M. Hyaluronan-based hybrid systems as growth factor carriers in the treatment of chronic wounds. Int J Mol Sci. 2025;26(22):10871. doi: 10.3390/Int J Mol Sci262210871
- Martin AR, Patel JM, Locke RC, et al. Nanofibrous hyaluronic acid scaffolds delivering TGF-β3 and SDF-1α for articular cartilage repair in a large animal model. Acta Biomater. 2021;126:170-182. doi: 10.1016/j.actbio.2021.03.013
- Kim J, Kim IS, Cho TH, et al. Bone regeneration using hyaluronic acid-based hydrogel with bone morphogenic protein-2 and human mesenchymal stem cells. Biomaterials. 2007;28(10):1830-1837. doi: 10.1016/j.biomaterials.2006.11.050
- Wang J, Liu M, Yang C, et al. Photocrosslinked gelatin methacryloyl/hyaluronic acid methacryloyl composite hydrogels loaded with bone morphogenetic protein 2-black phosphorus nanosheets for bone regeneration. J Biomater Sci Polym Ed. 2025;36(14):1962-1984. doi: 10.1080/09205063.2025.2489846
- Valachová K, El Meligy MA, Šoltés L. Hyaluronic acid and chitosan-based electrospun wound dressings: Problems and solutions. Int J Biol Macromol. 2022;206:74-91. doi: 10.1016/j.ijbiomac.2022.02.117
- Orellana SL, Giacaman A, Pavicic F, Vidal A, Moreno‐ Villoslada I, Concha M. Relevance of charge balance and hyaluronic acid on alginate‐chitosan sponge microstructure and its influence on fibroblast growth. J Biomed Mater Res. 2016;104(10):2537-2543. doi: 10.1002/jbm.a.35797
- Anisha BS, Biswas R, Chennazhi KP, Jayakumar R. Chitosan–hyaluronic acid/nano silver composite sponges for drug resistant bacteria infected diabetic wounds. Int J Biol Macromol. 2013;62:310-320. doi: 10.1016/j.ijbiomac.2013.09.011
- Saravanakumar K, Park S, Santosh SS, et al. Application of hyaluronic acid in tissue engineering, regenerative medicine, and nanomedicine: A review. Int J Biol Macromol. 2022;222:2744-2760. doi: 10.1016/j.ijbiomac.2022.10.055
- Bai Q, Gao Q, Hu F, et al. Chitosan and hyaluronic-based hydrogels could promote the infected wound healing. Int J Biol Macromol. 2023;232:123271. doi: 10.1016/j.ijbiomac.2023.123271
- Yuan N, Shao K, Huang S, Chen C. Chitosan, alginate, hyaluronic acid and other novel multifunctional hydrogel dressings for wound healing: A review. Int J Biol Macromol. 2023;240:124321. doi: 10.1016/j.ijbiomac.2023.124321
- Wu S, Deng L, Hsia H, et al. Evaluation of gelatin-hyaluronic acid composite hydrogels for accelerating wound healing. J Biomater Appl. 2017;31(10):1380-1390. doi: 10.1177/0885328217702526
- Hwang J, Kiick KL, Sullivan MO. VEGF-Encoding, Gene- Activated Collagen-Based Matrices Promote Blood Vessel Formation and Improved Wound Repair. ACS Appl Mater Interfaces. 2023;15(13):16434-16447. doi: 10.1021/acsami.2c23022
- Ying H, Zhou J, Wang M, et al. In situ formed collagen-hyaluronic acid hydrogel as biomimetic dressing for promoting spontaneous wound healing. Mater Sci Eng C. 2019;101:487-498. doi: 10.1016/j.msec.2019.03.093
- Wang Y, Zhang Y, Yang YP, et al. Versatile dopamine-functionalized hyaluronic acid-recombinant human collagen hydrogel promoting diabetic wound healing via inflammation control and vascularization tissue regeneration. Bioact Mater. 2024;35:330-345. doi: 10.1016/j.bioactmat.2024.02.010
- Yang C, Zhang Y, Zhang X, et al. An injectable, self-healing, and antioxidant collagen- and hyaluronic acid-based hydrogel mediated with gallic acid and dopamine for wound repair. Carbohydr Polym. 2023;320:121231. doi: 10.1016/j.carbpol.2023.121231
- Catanzano O, D’Esposito V, Formisano P, Boateng JS, Quaglia F. Composite Alginate-Hyaluronan Sponges for the Delivery of Tranexamic Acid in Postextractive Alveolar Wounds. J Pharm Sci. 2018;107(2):654-661. doi: 10.1016/j.xphs.2017.09.026
- Zhou J, Zhang B, Liu X, et al. Facile method to prepare silk fibroin/hyaluronic acid films for vascular endothelial growth factor release. Carbohydr Polym. 2016;143:301-309. doi: 10.1016/j.carbpol.2016.01.023
- Guan Y, You H, Cai J, Zhang Q, Yan S, You R. Physically crosslinked silk fibroin/hyaluronic acid scaffolds. Carbohydr Polym. 2020;239:116232. doi: 10.1016/j.carbpol.2020.116232
- Eskandarinia A, Kefayat A, Rafienia M, Agheb M, Navid S, Ebrahimpour K. Cornstarch-based wound dressing incorporated with hyaluronic acid and propolis: In vitro and in vivo studies. Carbohydr Polym. 2019;216:25-35. doi: 10.1016/j.carbpol.2019.03.091
- Satchanska G, Davidova S, Petrov PD. Natural and Synthetic Polymers for Biomedical and Environmental Applications. Polymers. 2024;16(8):1159. doi: 10.3390/polym16081159
- Ibrahim M, Ramadan E, Elsadek NE, et al. Polyethylene glycol (PEG): The nature, immunogenicity, and role in the hypersensitivity of PEGylated products. J Control Release. 2022;351:215-230. doi: 10.1016/j.jconrel.2022.09.031
- Gao Y, Vogus D, Zhao Z, et al. Injectable hyaluronic acid hydrogels encapsulating drug nanocrystals for long‐term treatment of inflammatory arthritis. Bioeng Transl Med. 2022;7(1):e10245. doi: 10.1002/btm2.10245
- Yang R, Chen M, Yang X, et al. Modified poloxamer 407 and hyaluronic acid thermosensitive hydrogel-encapsulated keratinocyte growth factor 2 improves knee osteoarthritis in rats. Mater Des. 2021;210:110086. doi: 10.1016/j.matdes.2021.110086
- Silva R, Medeiros M, Paula CTB, et al. Light-Mediated 3D-Printed Wound Dressings Based on Natural Polymers with Improved Adhesion and Antioxidant Properties. Polymers. 2025;17(8):1114. doi: 10.3390/polym17081114
- Zamboni F, Keays M, Hayes S, et al. Enhanced cell viability in hyaluronic acid coated poly(lactic-co-glycolic acid) porous scaffolds within microfluidic channels. Int J Pharm. 2017;532(1):595-602. doi: 10.1016/j.ijpharm.2017.09.053
- Bhattacharya S, Singh D, Aich J, Ajazuddin, Shete MB. Development and characterization of hyaluronic acid surface scaffolds Encorafenib loaded polymeric nanoparticles for colorectal cancer targeting. Mater Today Commun. 2022;31:103757. doi: 10.1016/j.mtcomm.2022.103757
- Wang Z, Qian Y, Li L, et al. Evaluation of emulsion electrospun polycaprolactone/hyaluronan/epidermal growth factor nanofibrous scaffolds for wound healing. J Biomater Appl. 2016;30(6):686-698. doi: 10.1177/0885328215586907
- Niu Y, Stadler FJ, Fang J, Galluzzi M. Hyaluronic acid-functionalized poly-lactic acid (PLA) microfibers regulate vascular endothelial cell proliferation and phenotypic shape expression. Colloids Surf B Biointerfaces. 2021;206:111970. doi: 10.1016/j.colsurfb.2021.111970
- Zhang X, Wan H, Lan W, et al. Fabrication of adhesive hydrogels based on poly (acrylic acid) and modified hyaluronic acid. J Mech Behav Biomed Mater. 2022;126:105044. doi: 10.1016/j.jmbbm.2021.105044
- Lewandowska K. Miscibility Studies of Hyaluronic Acid and Poly(Vinyl Alcohol) Blends in Various Solvents. Materials. 2020;13(21):4750. doi: 10.3390/ma13214750
- Ren Y, Ma S, Zhang D, et al. Functionalized injectable hyaluronic acid hydrogel with antioxidative and photothermal antibacterial activity for infected wound healing. Int J Biol Macromol. 2022;210:218-232. doi: 10.1016/j.ijbiomac.2022.05.024
- Yu S, Wang S, Xie Z, et al. Hyaluronic acid coating on the surface of curcumin-loaded ZIF-8 nanoparticles for improved breast cancer therapy: An in vitro and in vivo study. Colloids Surf B Biointerfaces. 2021;203:111759. doi: 10.1016/j.colsurfb.2021.111759
- Da Silva LP, Kundu SC, Reis RL, Correlo VM. Electric phenomenon: a disregarded tool in tissue engineering and regenerative medicine. Trends Biotechnol. 2020;38(1):24-49. doi: 10.1016/j.tibtech.2019.07.002
- Hayes AJ, Melrose J. Electro‐stimulation, a promising therapeutic treatment modality for tissue repair: emerging roles of sulfated glycosaminoglycans as electro‐regulatory mediators of intrinsic repair processes. Adv Ther. 2020;3(11):2000151. doi: 10.1002/adtp.202000151
- Thakral G, LaFontaine J, Najafi B, Talal TK, Kim P, Lavery LA. Electrical stimulation to accelerate wound healing. Diabet Foot Ankle. 2013;4(1):22081. doi: 10.3402/dfa.v4i0.22081
- Castrejón-Comas V, Alemán C, Pérez-Madrigal MM. Multifunctional conductive hyaluronic acid hydrogels for wound care and skin regeneration. Biomater Sci. 2023;11(7):2266-2276. doi: 10.1039/D2BM02057B
- Lu Y, Wang Y, Zhang J, et al. In-situ doping of a conductive hydrogel with low protein absorption and bacterial adhesion for electrical stimulation of chronic wounds. Acta Biomater. 2019;89:217-226. doi: 10.1016/j.actbio.2019.03.018
- Fu F, Wang J, Zeng H, Yu J. Functional Conductive Hydrogels for Bioelectronics. ACS Mater Lett. 2020;2(10):1287-1301. doi: 10.1021/acsmaterialslett.0c00309
- Min JH, Patel M, Koh WG. Incorporation of Conductive Materials into Hydrogels for Tissue Engineering Applications. Polymers. 2018;10(10):1078. doi: 10.3390/polym10101078
- Shin M, Song KH, Burrell JC, Cullen DK, Burdick JA. Injectable and Conductive Granular Hydrogels for 3D Printing and Electroactive Tissue Support. Adv Sci. 2019;6(20):1901229. doi: 10.1002/advs.201901229
- Chen X, Zhang H, Yang X, et al. Preparation and Application of Quaternized Chitosan- and AgNPs-Base Synergistic Antibacterial Hydrogel for Burn Wound Healing. Molecules. 2021;26(13):4037. doi: 10.3390/molecules26134037
- Deng M, Wu Y, Ren Y, et al. Clickable and smart drug delivery vehicles accelerate the healing of infected diabetic wounds. J Control. Release. 2022;350:613-629. doi: 10.1016/j.jconrel.2022.08.053
- El-Aassar MR, El-Beheri NG, Agwa MM, et al. Antibiotic-free combinational hyaluronic acid blend nanofibers for wound healing enhancement. Int J Biol Macromol. 2021;167:1552-1563. doi: 10.1016/j.ijbiomac.2020.11.109
- Zhang B, Lv Y, Yu C, et al. Au–Pt nanozyme-based multifunctional hydrogel dressing for diabetic wound healing. Biomater Adv. 2022;137:212869. doi: 10.1016/j.bioadv.2022.212869
- Cho KW, Sunwoo SH, Hong YJ, et al. Soft Bioelectronics Based on Nanomaterials. Chem Rev. 2022;122(5):5068- 5143. doi: 10.1021/acs.chemrev.1c00531
- Liang Y, Zhao X, Hu T, et al. Adhesive Hemostatic Conducting Injectable Composite Hydrogels with Sustained Drug Release and Photothermal Antibacterial Activity to Promote Full‐Thickness Skin Regeneration During Wound Healing. Small. 2019;15(12):1900046. doi: 10.1002/smll.201900046
- Li J, Ma J, Sun H, et al. Transformation of arginine into zero-dimensional nanomaterial endows the material with antibacterial and osteoinductive activity. Sci Adv. 2023;9(21):eadf8645. doi: 10.1126/sciadv.adf8645
- Rastin H, Zhang B, Mazinani A, et al. 3D bioprinting of cell-laden electroconductive MXene nanocomposite bioinks. Nanoscale. 2020;12(30):16069-16080. doi: 10.1039/D0NR02581J
- Talikowska M, Fu X, Lisak G. Application of conducting polymers to wound care and skin tissue engineering: A review. Biosens Bioelectron. 2019;135:50-63. doi: 10.1016/j.bios.2019.04.001
- Yang Y, Xu H, Li M, et al. Antibacterial Conductive UV-Blocking Adhesion Hydrogel Dressing with Mild On-Demand Removability Accelerated Drug-Resistant Bacteria-Infected Wound Healing. ACS Appl Mater Interfaces. 2022;14(37):41726-41741. doi: 10.1021/acsami.2c10490
- Qu J, Zhao X, Liang Y, Xu Y, Ma PX, Guo B. Degradable conductive injectable hydrogels as novel antibacterial, anti-oxidant wound dressings for wound healing. Chem Eng J. 2019;362:548-560. doi: 10.1016/j.cej.2019.01.028
- Wu C, Shen L, Lu Y, et al. Intrinsic Antibacterial and Conductive Hydrogels Based on the Distinct Bactericidal Effect of Polyaniline for Infected Chronic Wound Healing. ACS Appl Mater Interfaces. 2021;13(44):52308-52320. doi: 10.1021/acsami.1c14088
- Jin X, Shang Y, Zou Y, et al. Injectable Hypoxia-Induced Conductive Hydrogel to Promote Diabetic Wound Healing. ACS Appl Mater Interfaces. 2020;12(51):56681-56691. doi: 10.1021/acsami.0c13197
- Wu C, Long L, Zhang Y, et al. Injectable conductive and angiogenic hydrogels for chronic diabetic wound treatment. J Control Release. 2022;344:249-260. doi: 10.1016/j.jconrel.2022.03.014
- Xu Y, Patsis PA, Hauser S, et al. Cytocompatible, Injectable, and Electroconductive Soft Adhesives with Hybrid Covalent/Noncovalent Dynamic Network. Adv Sci. 2019;6(15):1802077. doi: 10.1002/advs.201802077
- Long L, Liu W, Hu C, Yang L, Wang Y. Construction of multifunctional wound dressings with their application in chronic wound treatment. Biomater Sci. 2022;10(15):4058- 4076. doi: 10.1039/D2BM00620K
- Lv R, Bei Z, Huang Y, et al. Mussel‐Inspired Flexible, Wearable, and Self‐Adhesive Conductive Hydrogels for Strain Sensors. Macromol Rapid Commun. 2020;41(2):1900450. doi: 10.1002/marc.201900450
- Lei L, Zhu Y, Qin X, et al. Magnetic biohybrid microspheres for protein purification and chronic wound healing in diabetic mice. Chem Eng J. 2021;425:130671. doi: 10.1016/j.cej.2021.130671
- Jin L, Guo X, Gao D, et al. An NIR photothermal-responsive hybrid hydrogel for enhanced wound healing. Bioact Mater. 2022;16:162-172. doi: 10.1016/j.bioactmat.2022.03.006
- Hu B, Gao M, Boakye-Yiadom KO, et al. An intrinsically bioactive hydrogel with on-demand drug release behaviors for diabetic wound healing. Bioact Mater. 2021;6(12):4592- 4606. doi: 10.1016/j.bioactmat.2021.04.040
- Fiorica C, Palumbo FS, Pitarresi G, Allegra M, Puleio R, Giammona G. Hyaluronic acid and α-elastin based hydrogel for three dimensional culture of vascular endothelial cells. J Drug Deliv Sci Technol. 2018;46:28-33. doi: 10.1016/j.jddst.2018.04.017
- Yang AL, Sun SB, Qu LY, et al. Polysaccharide hydrogel containing silver nanoparticle@catechol microspheres with photothermal, antibacterial and anti-inflammatory activities for infected-wounds repair. Int J Biol Macromol. 2024;265:130898. doi: 10.1016/j.ijbiomac.2024.130898
- Chen RF, Wang CT, Chen YH, et al. Hyaluronic Acid– Povidone-Iodine Compound Facilitates Diabetic Wound Healing in a Streptozotocin-Induced Diabetes Rodent Model. Plast Reconstr Surg. 2019;143(5):1371-1382. doi: 10.1097/PRS.0000000000005504
- Mahedia M, Shah N, Amirlak B. Clinical Evaluation of Hyaluronic Acid Sponge with Zinc versus Placebo for Scar Reduction after Breast Surgery. Plast Reconstr Surg. 2016;4(7):e791. doi: 10.1097/GOX.0000000000000747
- Zhu J, Tang X, Jia Y, Ho CT, Huang Q. Applications and delivery mechanisms of hyaluronic acid used for topical/transdermal delivery–A review. Int J Pharm. 2020;578:119127. doi: 10.1016/j.ijpharm.2020.119127
- Çetinkalp Ş, Gökçe EH, Şimşir I, et al. Comparative Evaluation of Clinical Efficacy and Safety of Collagen Laminin–Based Dermal Matrix Combined With Resveratrol Microparticles (Dermalix) and Standard Wound Care for Diabetic Foot Ulcers. Int J Low Extrem Wounds. 2021;20(3):217-226. doi: 10.1177/1534734620907773
- De Caridi G, Massara M, Acri I, et al. Trophic effects of polynucleotides and hyaluronic acid in the healing of venous ulcers of the lower limbs: a clinical study. Int Wound J. 2016;13(5):754-758. doi: 10.1111/iwj.12368
- Gong M, Yan F, Yu L, Li F. A dopamine-methacrylated hyaluronic acid hydrogel as an effective carrier for stem cells in skin regeneration therapy. Cell Death Dis. 2022;13(8):738. doi: 10.1038/s41419-022-05060-9
- Ferroni L, D’Amora U, Gardin C, et al. Stem cell-derived small extracellular vesicles embedded into methacrylated hyaluronic acid wound dressings accelerate wound repair in a pressure model of diabetic ulcer. J Nanobiotechnol. 2023;21(1):469. doi: 10.1186/s12951-023-02202-9
- Ni C, Zhang Z, Wang Y, Zhang Z, Guo X, Lv H. Hyaluronic acid and HA-modified cationic liposomes for promoting skin penetration and retention. J Control Release. 2023;357:432-443. doi: 10.1016/j.jconrel.2023.03.049
- Sharma T, Thakur S, Kaur M, Singh A, Jain SK. Novel Hyaluronic Acid ethosomes based gel formulation for topical use with reduced toxicity, better skin permeation, deposition, and improved pharmacodynamics. J Liposome Res. 2023;33(2):129-143. doi: 10.1080/08982104.2022.2087675
- Mansoori B, Mohammadi A, Abedi‐Gaballu F, et al. Hyaluronic acid‐decorated liposomal nanoparticles for targeted delivery of 5‐fluorouracil into HT‐29 colorectal cancer cells. J Cell Physiol. 2020;235(10):6817-6830. doi: 10.1002/jcp.29576
- Šmejkalová D, Muthný T, Nešporová K, et al. Hyaluronan polymeric micelles for topical drug delivery. Carbohydr Polym. 2017;156:86-96. doi: 10.1016/j.carbpol.2016.09.013
- Yue Y, Zhao D, Yin Q. Hyaluronic acid modified nanostructured lipid carriers for transdermal bupivacaine delivery: In vitro and in vivo anesthesia evaluation. Biomed Pharmacother. 2018;98:813-820. doi: 10.1016/j.biopha.2017.12.103
- Paghetti A, Bellingeri A, Pomponio G, Sansoni J, Paladino D. [Topic efficacy of ialuronic acid associated with argentic sulphadiazine (Connettivina Plus) in the treatment of pressure sores: a prospective observational cohort study]. Prof Inferm. 2009;62(2):67-77. [In Italian]
- Myers SR, Partha VN, Soranzo C, Price RD, Navsaria HA. Hyalomatrix: A Temporary Epidermal Barrier, Hyaluronan Delivery, and Neodermis Induction System for Keratinocyte Stem Cell Therapy. Tissue Eng. 2007;13(11):2733-2741. doi: 10.1089/ten.2007.0109
- Khelfi A. Therapeutic Enzymes Used for the Treatment of Non-Deficiency Diseases. In: Research Advancements in Pharmaceutical, Nutritional, and Industrial Enzymology. IGI Global Scientific Publishing; 2018:25. doi: 10.4018/978-1-5225-5237-6.ch003
- Colletta V, Dioguardi D, Di Lonardo A, Maggio G, Torasso F. A trial to assess the efficacy and tolerability of Hyalofill-F in non-healing venous leg ulcers. J Wound Care. 2003;12(9):357-361. doi: 10.12968/jowc.2003.12.9.26530
- Taddeucci P, Pianigiani E, Colletta V, Torasso F, Andreassi L, Andreass A. An evaluation of Hyalofill-F plus compression bandaging in the treatment of chronic venous ulcers. J Wound Care. 2004;13(5):202-204. doi: 10.12968/jowc.2004.13.5.26613
- Zerbinati N, Esposito C, Cipolla G, et al. Chemical and mechanical characterization of hyaluronic acid hydrogel cross‐linked with polyethylen glycol and its use in dermatology. Dermatol Ther. 2020;33(4). doi: 10.1111/dth.13747
- Simões D, Miguel SP, Ribeiro MP, Coutinho P, Mendonça AG, Correia IJ. Recent advances on antimicrobial wound dressing: A review. Eur J Pharm Biopharm. 2018;127:130- 141. doi: 10.1016/j.ejpb.2018.02.022
- Longinotti C. The use of hyaluronic acid based dressings to treat burns: A review. Burn Trauma. 2014;2(4):162. doi: 10.4103/2321-3868.142398
- Uccioli L, Giurato L, Ruotolo V, et al. Two-Step Autologous Grafting Using HYAFF Scaffolds in Treating Difficult Diabetic Foot Ulcers: Results of A Multicenter, Randomized Controlled Clinical Trial With Long-Term Follow-up. Int J Low Extrem Wounds. 2011;10(2):80-85. doi: 10.1177/1534734611409371
- Huerta-Ángeles G, Nešporová K, Ambrožová G, Kubala L, Velebný V. An Effective Translation: The Development of Hyaluronan-Based Medical Products From the Physicochemical, and Preclinical Aspects. Front Bioeng Biotechnol. 2018;6:62. doi: 10.3389/fbioe.2018.00062
- Gudapati H, Dey M, Ozbolat I. A comprehensive review on droplet-based bioprinting: Past, present and future. Biomaterials. 2016;102:20-42. doi: 10.1016/j.biomaterials.2016.06.012
- Xu H, Zhang S, Song K, Yang H, Yin J, Huang Y. Droplet-based 3D bioprinting for drug delivery and screening. Adv Drug Deliv Rev. 2025;217:115486. doi: 10.1016/j.addr.2024.115486
- Ng WL, Shkolnikov V. Jetting-based bioprinting: process, dispense physics, and applications. Bio-des Manuf. 2024;7(5):771-799. doi: 10.1007/s42242-024-00285-3
- Chivate A, Zhou C. Additive manufacturing of micropatterned functional surfaces: a review. Int J Extrem Manuf. 2024;6(4):042004. doi: 10.1088/2631-7990/ad4240
- Christensen K, Xu C, Chai W, Zhang Z, Fu J, Huang Y. Freeform inkjet printing of cellular structures with bifurcations. Biotech Bioeng. 2015;112(5):1047-1055. doi: 10.1002/bit.25501
- Yoon S, Park JA, Lee H, Yoon WH, Hwang DS, Jung S. Inkjet–Spray Hybrid Printing for 3D Freeform Fabrication of Multilayered Hydrogel Structures. Adv Healthc Mater. 2018;7(14):1800050. doi: 10.1002/adhm.201800050
- Serra P, Piqué A. Laser‐Induced Forward Transfer: Fundamentals and Applications. Adv Mater Technol. 2019;4(1):1800099. doi: 10.1002/admt.201800099
- Sopeña P, Fernández-Pradas JM, Serra P. Laser-induced forward transfer of conductive screen-printing inks. Appl Surf Sci. 2020;507:145047. doi: 10.1016/j.apsusc.2019.145047
- Munoz-Martin D, Brasz CF, Chen Y, Morales M, Arnold CB, Molpeceres C. Laser-induced forward transfer of high-viscosity silver pastes. Appl Surf Sci. 2016;366:389-396. doi: 10.1016/j.apsusc.2016.01.029
- Ding YW, Zhang XW, Mi CH, Qi XY, Zhou J, Wei DX. Recent advances in hyaluronic acid-based hydrogels for 3D bioprinting in tissue engineering applications. Smart Mater Med. 2023;4:59-68. doi: 10.1016/j.smaim.2022.07.003
- Michael S, Sorg H, Peck CT, et al. Tissue Engineered Skin Substitutes Created by Laser-Assisted Bioprinting Form Skin-Like Structures in the Dorsal Skin Fold Chamber in Mice. PLoS ONE. 2013;8(3):e57741. doi: 10.1371/journal.pone.0057741
- Murphy SV, Atala A. 3D bioprinting of tissues and organs. Nat Biotechnol. 2014;32(8):773-785. doi: 10.1038/nbt.2958
- Chen DXB. Extrusion Bioprinting of Scaffolds for Tissue Engineering. Springer International Publishing; 2025. doi: 10.1007/978-3-031-72471-8
- Mandrycky C, Wang Z, Kim K, Kim DH. 3D bioprinting for engineering complex tissues. Biotechnol Adv. 2016;34(4):422- 434. doi: 10.1016/j.biotechadv.2015.12.011
- Sarker Md, Chen XB. Modeling the Flow Behavior and Flow Rate of Medium Viscosity Alginate for Scaffold Fabrication With a Three-Dimensional Bioplotter. J Manuf Sci Eng. 2017;139(8):081002. doi: 10.1115/1.4036226
- Jeschke MG, Van Baar ME, Choudhry MA, Chung KK, Gibran NS, Logsetty S. Burn injury. Nat Rev Dis Primers. 2020;6(1):11. doi: 10.1038/s41572-020-0145-5
- Fu Z, Naghieh S, Xu C, Wang C, Sun W, Chen X. Printability in extrusion bioprinting. Biofabrication. 2021;13(3):033001. doi: 10.1088/1758-5090/abe7ab
- Lu T, Li Y, Chen T. Techniques for fabrication and construction of three-dimensional scaffolds for tissue engineering. Int J Nanomed. 2013:337. doi: 10.2147/IJN.S38635
- Zhang YS, Haghiashtiani G, Hübscher T, et al. 3D extrusion bioprinting. Nat Rev Methods Primers. 2021;1(1):75. doi: 10.1038/s43586-021-00073-8
- Narayanan LK, Huebner P, Fisher MB, Spang JT, Starly B, Shirwaiker RA. 3D-Bioprinting of Polylactic Acid (PLA) Nanofiber–Alginate Hydrogel Bioink Containing Human Adipose-Derived Stem Cells. ACS Biomater Sci Eng. 2016;2(10):1732-1742. doi: 10.1021/acsbiomaterials.6b00196
- Barros NR, Kim HJ, Gouidie MJ, et al. Biofabrication of endothelial cell, dermal fibroblast, and multilayered keratinocyte layers for skin tissue engineering. Biofabrication. 2021;13(3):035030. doi: 10.1088/1758-5090/aba503
- Wang Z, Liang X, Wang G, Wang X, Chen Y. Emerging Bioprinting for Wound Healing. Carbohydr Polym. 2023;37(31):2304738. doi: 10.1002/adma.202304738
- Kjar A, McFarland B, Mecham K, Harward N, Huang Y. Engineering of tissue constructs using coaxial bioprinting. Bioact Mater. 2021;6(2):460-471. doi: 10.1016/j.bioactmat.2020.08.020
- Brunel LG, Hull SM, Heilshorn SC. Engineered assistive materials for 3D bioprinting: support baths and sacrificial inks. Biofabrication. 2022;14(3):032001. doi: 10.1088/1758-5090/ac6bbe
- He C, Yu B, Lv Y, et al. Biomimetic Asymmetric Composite Dressing by Electrospinning with Aligned Nanofibrous and Micropatterned Structures for Severe Burn Wound Healing. ACS Appl Mater Interfaces. 2022;14(29):32799-32812. doi: 10.1021/acsami.2c04323
- Keirouz A, Chung M, Kwon J, Fortunato G, Radacsi N. 2D and 3D electrospinning technologies for the fabrication of nanofibrous scaffolds for skin tissue engineering: A review. WIREs Nanomed Nanobiotechnol. 2020;12(4):e1626. doi: 10.1002/wnan.1626
- Rahmati M, Mills DK, Urbanska AM, et al. Electrospinning for tissue engineering applications. Prog Mater Sci. 2021;117:100721. doi: 10.1016/j.pmatsci.2020.100721
- Al-Abduljabbar A, Farooq I. Electrospun Polymer Nanofibers: Processing, Properties, and Applications. Polymers. 2022;15(1):65. doi: 10.3390/polym15010065
- Ahmadi S, Shafiei SS, Sabouni F. Electrospun Nanofibrous Scaffolds of Polycaprolactone/Gelatin Reinforced with Layered Double Hydroxide Nanoclay for Nerve Tissue Engineering Applications. ACS Omega. 2022;7(32):28351- 28360. doi: 10.1021/acsomega.2c02863
- Khil M, Cha D, Kim H, Kim I, Bhattarai N. Electrospun nanofibrous polyurethane membrane as wound dressing. J Biomed Mater Res. 2003;67B(2):675-679. doi: 10.1002/jbm.b.10058
- Hadisi Z, Nourmohammadi J, Nassiri SM. The antibacterial and anti-inflammatory investigation of Lawsonia Inermis-gelatin-starch nano-fibrous dressing in burn wound. Int J Biol Macromol. 2018;107:2008-2019. doi: 10.1016/j.ijbiomac.2017.10.061
- Adeli-Sardou M, Yaghoobi MM, Torkzadeh-Mahani M, Dodel M. Controlled release of lawsone from polycaprolactone/gelatin electrospun nano fibers for skin tissue regeneration. Int J Biol Macromol. 2019;124:478-491. doi: 10.1016/j.ijbiomac.2018.11.237
- Huang B, Aslan E, Jiang Z, et al. Engineered dual-scale poly (ε-caprolactone) scaffolds using 3D printing and rotational electrospinning for bone tissue regeneration. Addit Manuf. 2020;36:101452. doi: 10.1016/j.addma.2020.101452
- Choi KY, Ajiteru O, Hong H, Suh YJ. A digital light processing 3D-printed artificial skin model and full-thickness wound models using silk fibroin bioink. Acta Biomater. 2023;164:159-174. doi: 10.1016/j.actbio.2023.04.034
- Lu Z, Cui J, Liu F, et al. A 4D Printed Adhesive, Thermo‐ Contractile, and Degradable Hydrogel for Diabetic Wound Healing. Adv Healthc Mater. 2024;13(10):2303499. doi: 10.1002/adhm.202303499
- Monk DW, Gale RO. The Digital Micromirror Device for Projection Display. Microelectron Eng. 1995;27:489-493. doi: 10.1016/0167-9317(94)00151-J
- Janusziewicz R, Tumbleston JR, Quintanilla AL, Mecham SJ, DeSimone JM. Layerless fabrication with continuous liquid interface production. Proc Natl Acad Sci USA. 2016;113(42):11703-11708. doi: 10.1073/pnas.1605271113
- Li S, Huang C, Liu H, et al. A Silk Fibroin Methacryloyl- Modified Hydrogel Promoting Cell Adhesion for Customized 3D Cell-Laden Structures. ACS Appl Polym Mater. 2022;4(10):7014-7024. doi: 10.1021/acsapm.2c00952
- Wang M, Li W, Hao J, et al. Molecularly cleavable bioinks facilitate high-performance digital light processing-based bioprinting of functional volumetric soft tissues. Nat Commun. 2022;13(1):3317. doi: 10.1038/s41467-022-31002-2
- Liu D, Jiang P, Wang Y, et al. Engineering Tridimensional Hydrogel Tissue and Organ Phantoms with Tunable Springiness. Adv Funct Mater. 2023;33(17):2214885. doi: 10.1002/adfm.202214885
- Peng X, Wu S, Sun X, et al. 4D Printing of Freestanding Liquid Crystal Elastomers via Hybrid Additive Manufacturing. Carbohydr Polym. 2022;34(39):2204890. doi: 10.1002/adma.202204890
- Zhou F, Hong Y, Liang R, et al. Rapid printing of bio-inspired 3D tissue constructs for skin regeneration. Biomaterials. 2020;258:120287. doi: 10.1016/j.biomaterials.2020.120287
- Foroozandeh A, Shakiba M, Zamani A, et al. Electrospun nylon 6/hyaluronic acid/chitosan bioactive nanofibrous composite as a potential antibacterial wound dressing. J Biomed Mater Res. 2024;112(1):e35370. doi: 10.1002/jbm.b.35370
- De Castro KC, Silva EK, Campos MGN, Mei LHI. Hyaluronic Acid/Polyvinyl Alcohol Electrospun Nanofiber Membranes Loaded with Plantago Major Extract for Smart Wound Dressings. ACS Appl Nano Mater. 2022;5(9):12616- 12625. doi: 10.1021/acsanm.2c02402
- Ilomuanya MO, Bassey PO, Cardoso‐Daodu IM, et al. Evaluation of Far‐Field Electrospun Polyvinyl Alcohol/ Hyaluronic Acid Nanofibrous Membranes for Skin Tissue Engineering Applications. Macro Mater Eng. 2024;309(3):2300299. doi: 10.1002/mame.202300299
- Su S, Bedir T, Kalkandelen C, et al. A drug-eluting nanofibrous hyaluronic acid-keratin mat for diabetic wound dressing. Emergent Mater. 2022;5(6):1617-1627. doi: 10.1007/s42247-022-00418-3
- Yang Q, Xie Z, Hu J, Liu Y. Hyaluronic acid nanofiber mats loaded with antimicrobial peptide towards wound dressing applications. Mater Sci Eng C. 2021;128:112319. doi: 10.1016/j.msec.2021.112319
- Gruppuso M, Turco G, Marsich E, Porrelli D. Antibacterial and bioactive multilayer electrospun wound dressings based on hyaluronic acid and lactose-modified chitosan. Biomater Adv. 2023;154:213613. doi: 10.1016/j.bioadv.2023.213613
- Xue F, Zhang H, Hu J, Liu Y. Hyaluronic acid nanofibers crosslinked with a nontoxic reagent. Carbohydr Polym. 2021;259:117757. doi: 10.1016/j.carbpol.2021.117757
- Chen C, He J, Huang J, et al. Fetal dermis inspired parallel PCL fibers layered PCL/COL/HA scaffold for dermal regeneration. React Funct Polym. 2022;170:105146. doi: 10.1016/j.reactfunctpolym.2021.105146
- Puertas-Bartolomé M, Włodarczyk-Biegun MK, Del Campo A, Vázquez-Lasa B, San Román J. Development of bioactive catechol functionalized nanoparticles applicable for 3D bioprinting. Mater Sci Eng C. 2021;131:112515. doi: 10.1016/j.msec.2021.112515
- Wang S, Xiong Y, Chen J, et al. Three Dimensional Printing Bilayer Membrane Scaffold Promotes Wound Healing. Front Bioeng Biotechnol. 2019;7:348. doi: 10.3389/fbioe.2019.00348
- Si H, Xing T, Ding Y, Zhang H, Yin R, Zhang W. 3D Bioprinting of the Sustained Drug Release Wound Dressing with Double-Crosslinked Hyaluronic-Acid-Based Hydrogels. Polymers. 2019;11(10):1584. doi: 10.3390/polym11101584
- Liang M, Dong L, Guo Z, et al. Collagen–Hyaluronic Acid Composite Hydrogels with Applications for Chronic Diabetic Wound Repair. ACS Biomater Sci Eng. 2023;9(9):5376-5388. doi: 10.1021/acsbiomaterials.3c00695
- Zhao H, Xu J, Yuan H, et al. 3D printing of artificial skin patches with bioactive and optically active polymer materials for anti-infection and augmenting wound repair. Mater Horiz. 2022;9(1):342-349. doi: 10.1039/D1MH00508A
- Ferroni L, Gardin C, D’Amora U, et al. Exosomes of mesenchymal stem cells delivered from methacrylated hyaluronic acid patch improve the regenerative properties of endothelial and dermal cells. Biomater Adv. 2022;139:213000. doi: 10.1016/j.bioadv.2022.213000
- Guan G, Qizhuang Lv, Liu S, Jiang Z, Zhou C, Liao W. 3D-bioprinted peptide coupling patches for wound healing. Mater Today Bio. 2022;13:100188. doi: 10.1016/j.mtbio.2021.100188
- Rimal R, Muduli S, Desai P, et al. Vascularized 3D Human Skin Models in the Forefront of Dermatological Research. Adv Healthc Mater. 2024;13(9):2303351. doi: 10.1002/adhm.202303351
- Kang MS, Jang J, Jo HJ, et al. Advances and Innovations of 3D Bioprinting Skin. Biomolecules. 2022;13(1):55. doi: 10.3390/biom13010055
- Wang Y, Gao C, Cheng S, et al. 3D Bioprinting of Double‐ Layer Conductive Skin for Wound Healing. Adv Healthc Mater. 2025;14(9):2404388. doi: 10.1002/adhm.202404388
- Bettendorf E, Schmid R, E. Horch R, et al. Bioprinted keratinocyte and stem cell laden constructs for skin tissue engineering. Int J Nanomed. 2024;10(6):3925. doi: 10.36922/ijb.3925
- Admane P, Gupta AC, Jois P, et al. Direct 3D bioprinted full-thickness skin constructs recapitulate regulatory signaling pathways and physiology of human skin. Bioprinting. 2019;15:e00051. doi: 10.1016/j.bprint.2019.e00051
- Ma X, Zhu X, Lv S, et al. 3D bioprinting of prefabricated artificial skin with multicomponent hydrogel for skin and hair follicle regeneration. Theranostics. 2025;15(7):2933- 2950. doi: 10.7150/thno.104854
- Kang MS, Kwon M, Lee SH, et al. 3D Printing of Skin Equivalents with Hair Follicle Structures and Epidermal‐ Papillary‐Dermal Layers Using Gelatin/Hyaluronic Acid Hydrogels. Chem Asian J. 2022;17(18):e202200620. doi: 10.1002/asia.202200620
- Zhang G, Zhang Z, Cao G, et al. Engineered dermis loaded with confining forces promotes full-thickness wound healing by enhancing vascularisation and epithelialisation. Acta Biomater. 2023;170:464-478. doi: 10.1016/j.actbio.2023.08.049
- Zhou Y, Fan Y, Chen Z, Yue Z, Wallace G. Catechol functionalized ink system and thrombin-free fibrin gel for fabricating cellular constructs with mechanical support and inner micro channels. Biofabrication. 2022;14(1):015004. doi: 10.1088/1758-5090/ac2ef8
- Hao L, Tao X, Feng M, et al. Stepwise Multi-Cross-Linking Bioink for 3D Embedded Bioprinting to Promote Full- Thickness Wound Healing. ACS Appl Mater Interfaces. 2023;15(20):24034-24046. doi: 10.1021/acsami.3c00688
- Hakimi N, Cheng R, Leng L, et al. Handheld skin printer: in situ formation of planar biomaterials and tissues. Lab Chip. 2018;18(10):1440-1451. doi: 10.1039/C7LC01236E
- Amirsadeghi A, Jafari A, Eggermont LJ, Hashemi SS, Bencherif SA, Khorram M. Vascularization strategies for skin tissue engineering. Biomater Sci. 2020;8(15):4073-4094. doi: 10.1039/D0BM00266F
- Budharaju H, Sundaramurthi D, Sethuraman S. Embedded 3D bioprinting–An emerging strategy to fabricate biomimetic & large vascularized tissue constructs. Bioact Mater. 2024;32:356-384. doi: 10.1016/j.bioactmat.2023.10.012
- Lee HR. 3D microextrusion-inkjet hybrid printing of structured human skin equivalents. Bioprinting. 2021;22:e00143. doi: 10.1016/j.bprint.2021.e00143
- Pontiggia L, Hengel IAV, Klar A, et al. Bioprinting and plastic compression of large pigmented and vascularized human dermo-epidermal skin substitutes by means of a new robotic platform. J Tissue Eng. 2022;13:1-20. doi: 10.1177/20417314221088513
- Blaeser A, Duarte Campos DF, Puster U, Richtering W, Stevens MM, Fischer H. Controlling Shear Stress in 3D Bioprinting is a Key Factor to Balance Printing Resolution and Stem Cell Integrity. Adv Healthc Mater. 2016;5(3):326- 333. doi: 10.1002/adhm.201500677
- Zhu H, Li R, Li S, et al. Multi-physical field control piezoelectric inkjet bioprinting for 3D tissue-like structure manufacturing. Int J Nanomed. 2024;10(3):2120. doi: 10.36922/ijb.2120
- Alblawi A, Ranjani AS, Yasmin H, Gupta S, Bit A, Rahimi- Gorji M. Scaffold-free: A developing technique in field of tissue engineering. Comput Methods and Programs Biomed. 2020;185:105148. doi: 10.1016/j.cmpb.2019.105148
- Chen K, Jiang E, Wei X, et al. The acoustic droplet printing of functional tumor microenvironments. Lab Chip. 2021;21(8):1604-1612. doi: 10.1039/D1LC00003A
- Vítková L, Kazantseva N, Musilová L, et al. Magneto-responsive hyaluronan hydrogel for hyperthermia and bioprinting: Magnetic, rheological properties and biocompatibility. APL Bioeng. 2023;7(3):036113. doi: 10.1063/5.0147181
- Dabas M, Schwartz D, Beeckman D, Gefen A. Application of Artificial Intelligence Methodologies to Chronic Wound Care and Management: A Scoping Review. Adv Wound Care. 2023;12(4):205-240. doi: 10.1089/wound.2021.0144
- Sarah R, Schimmelpfennig K, Rohauer R, Lewis CL, Limon SM, Habib A. Characterization and Machine Learning- Driven Property Prediction of a Novel Hybrid Hydrogel Bioink Considering Extrusion-Based 3D Bioprinting. Gels. 2025;11(1):45. doi: 10.3390/gels11010045
- Freeman S, Calabro S, Williams R, Jin S, Ye K. Bioink Formulation and Machine Learning-Empowered Bioprinting Optimization. Front Bioeng Biotechnol. 2022;10:913579. doi: 10.3389/fbioe.2022.913579
- Ning H, Zhou T, Joo SW. Machine learning boosts three-dimensional bioprinting. Int J Nanomed. 2023;9(4):739. doi: 10.18063/ijb.739
- Squiers JJ, Thatcher JE, Bastawros DS, et al. Machine learning analysis of multispectral imaging and clinical risk factors to predict amputation wound healing. J Vasc Surg. 2022;75(1):279-285. doi: 10.1016/j.jvs.2021.06.478
- Lemonnier D, Sumpio BJ, Crouse M, et al. Quantitative ultrasound augmented with machine learning to assess tissue microstructure during wound healing. Biomed Signal Process Control. 2025;103:107420. doi: 10.1016/j.bspc.2024.107420
- Xiao J, Zhou Z, Zhong G, Xu T, Zhang X. Self‐Sterilizing Microneedle Sensing Patches for Machine Learning‐ Enabled Wound pH Visual Monitoring. Adv Funct Mater. 2024;34(22):2315067. doi: 10.1002/adfm.202315067
- Deng D, Liang L, Su K, et al. Smart hydrogel dressing for machine learning-enabled visual monitoring and promote diabetic wound healing. Nano Today. 2025;60:102559. doi: 10.1016/j.nantod.2024.102559
- Lalman C, Stabler KR, Yang Y, Walker JL. Supervised Machine-Based Learning and Computational Analysis to Reveal Unique Molecular Signatures Associated with Wound Healing and Fibrotic Outcomes to Lens Injury. Int J Mol Sci. 2025;26(15):7422. doi: 10.3390/Int J Mol Sci26157422
- Veličković VM, Spelman T, Clark M, Probst S, Armstrong DG, Steyerberg E. Individualized Risk Prediction for Improved Chronic Wound Management. Adv Wound Care. 2023;12(7):387-398. doi: 10.1089/wound.2022.0017
- Najjar R. Redefining Radiology: A Review of Artificial Intelligence Integration in Medical Imaging. Diagnostics. 2023;13(17):2760. doi: 10.3390/diagnostics13172760
- Manickam P, Mariappan SA, Murugesan SM, et al. Artificial Intelligence (AI) and Internet of Medical Things (IoMT) Assisted Biomedical Systems for Intelligent Healthcare. Biosensors. 2022;12(8):562. doi: 10.3390/bios12080562
- Feng J, Phillips RV, Malenica I, et al. Clinical artificial intelligence quality improvement: towards continual monitoring and updating of AI algorithms in healthcare. npj Digit Med. 2022;5(1):66. doi: 10.1038/s41746-022-00611-y
- Chen RJ, Wang JJ, Williamson DFK, et al. Algorithmic fairness in artificial intelligence for medicine and healthcare. Nat Biomed Eng. 2023;7(6):719-742. doi: 10.1038/s41551-023-01056-8
- Hosseini SA, Noruzi S, Kesharwani P, Sahebkar A. Hydrogel-based dressing for wound healing: A systematic review of clinical trials. Int J Biol Macromol. 2025;308:142322. doi: 10.1016/j.ijbiomac.2025.142322
- Shi C, Wang C, Liu H, et al. Selection of Appropriate Wound Dressing for Various Wounds. Front Bioeng Biotechnol. 2020;8:182. doi: 10.3389/fbioe.2020.00182
- Bari E, Di Silvestre D, Mastracci L, et al. GMP-compliant sponge-like dressing containing MSC lyo-secretome: Proteomic network of healing in a murine wound model. Eur J Pharm Biopharm. 2020;155:37-48. doi: 10.1016/j.ejpb.2020.08.003
- Weller CD, Team V, Sussman G. First-Line Interactive Wound Dressing Update: A Comprehensive Review of the Evidence. Front Pharmacol. 2020;11:155. doi: 10.3389/fphar.2020.00155
- Abdi F, Michel R, Poirot R, et al. Dynamic Covalent Chemistry Enables Reconfigurable All‐Polysaccharide Nanogels. Macromol Rapid Commun. 2020;41(15):2000213. doi: 10.1002/marc.202000213
- Simińska-Stanny J, Hachemi F, Dodi G, et al. Optimizing phenol-modified hyaluronic acid for designing shape-maintaining biofabricated hydrogel scaffolds in soft tissue engineering. Int J Biol Macromol. 2023;244:125201. doi: 10.1016/j.ijbiomac.2023.125201
- Nishiyama Y, Nakamura M, Henmi C, et al. Development of a Three-Dimensional Bioprinter: Construction of Cell Supporting Structures Using Hydrogel and State-Of-The- Art Inkjet Technology. J Biomech Eng. 2008;131(3):035001. doi: 10.1115/1.3002759
- Liu M, Wang J, He M, et al. Inkjet Printing Controllable Footprint Lines by Regulating the Dynamic Wettability of Coalescing Ink Droplets. ACS Appl Mater Interfaces. 2014;6(16):13344-13348. doi: 10.1021/am5042548
- Teo MY, Kee S, RaviChandran N, Stuart L, Aw KC, Stringer J. Enabling Free-Standing 3D Hydrogel Microstructures with Microreactive Inkjet Printing. ACS Appl Mater Interfaces. 2020;12(1):1832-1839. doi: 10.1021/acsami.9b17192
- Malekpour A, Chen X. Printability and Cell Viability in Extrusion-Based Bioprinting from Experimental, Computational, and Machine Learning Views. J Funct Biomater. 2022;13(2):40. doi: 10.3390/jfb13020040
- Wang X, Yang C, Yu Y, Zhao Y. In Situ 3D Bioprinting Living Photosynthetic Scaffolds for Autotrophic Wound Healing. Research. 2022;2022. doi: 10.34133/2022/9794745
- Duan N, Geng X, Ye L, et al. A vascular tissue engineering scaffold with core–shell structured nano-fibers formed by coaxial electrospinning and its biocompatibility evaluation. Biomed Mater. 2016;11(3):035007. doi: 10.1088/1748-6041/11/3/035007
- Caprioli M, Roppolo I, Chiappone A, Larush L, Pirri CF, Magdassi S. 3D-printed self-healing hydrogels via Digital Light Processing. Nat Commun. 2021;12(1):2462. doi: 10.1038/s41467-021-22802-z
313. Hong H, Seo YB, Kim DY, et al. Digital light processing 3D printed silk fibroin hydrogel for cartilage tissue engineering. Biomaterials. 2020;232:119679. doi: 10.1016/j.biomaterials.2019.119679

