Inkjet bioprinting of NE-4C neural progenitor cells with enhanced neuronal differentiation via retinoic acid treatment

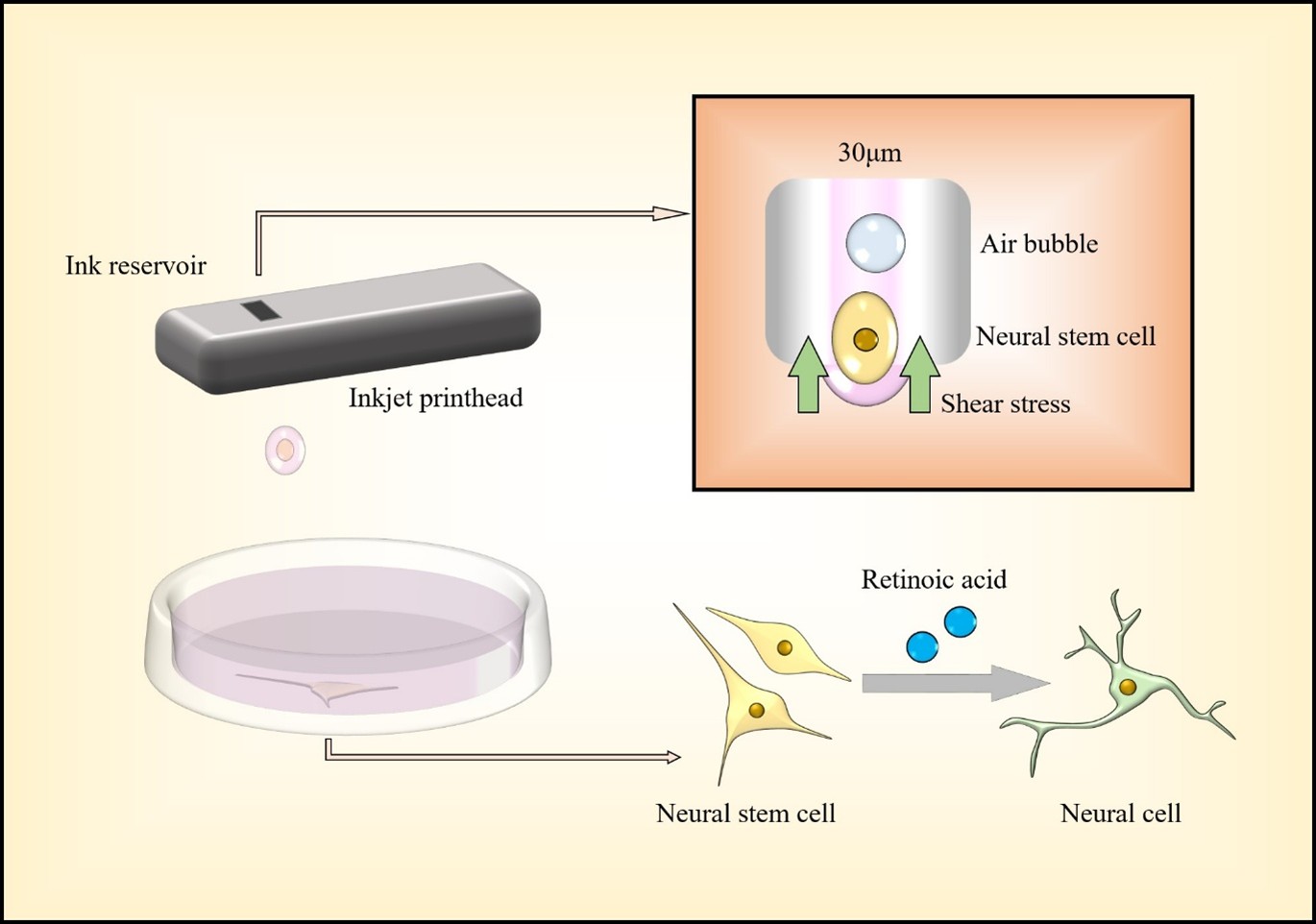
During inkjet bioprinting, cells are subjected to direct shear stress as they pass through the nozzles, causing reversible deformation of the cell membranes and potentially triggering subcellular changes, such as activation of molecular pathways, leading to beneficial o utcomes. I n t his s tudy, n eural p rogenitor NE-4C c ells were printed through 30μm thermal inkjet nozzles. Compared to manually pipetted cells (control group), bioprinted cells (inkjet group) exhibited several distinct changes, such as reduced cell proliferation during the first four days after bioprinting, increased tolerance to high-concentration retinoic acid, and significantly elevated expression of the early neuronal marker class III β-tubulin, indicating enhanced neuronal differentiation. Furthermore, RNA sequencing and enrichment analysis further revealed upregulation of cell metabolism pathways in the bioprinted group. Collectively, these findings suggest that inkjet bioprinting may be a promising strategy to accelerate neural tissue formation, warranting further studies.
- Saunders RE, Derby B. Inkjet printing biomaterials for tissue engineering: bioprinting. Int Mater Rev. 2014;59(8):430-448. doi: 10.1179/1743280414Y.0000000040.
- Gudapati H, Dey M, Ozbolat I. A comprehensive review on droplet-based bioprinting: Past, present and future. Biomaterials. 2016;102:20-42. doi: 10.1016/j.biomaterials.2016.06.012.
- Yeong WY, Chua C.K, Leong KF et al. Indirect fabrication of collagen scaffold based on inkjet printing technique. Rapid Prototyping J. 2006;12(4):229-237. doi: 10.1108/13552540610682741.
- Zhang C, Wen X, Vyavahare NR, Boland T. Synthesis and characterization of biodegradable elastomeric polyurethane scaffolds fabricated by the inkjet technique. Biomaterials. 2008;29(28):3781-3791. doi: 10.1016/j.biomaterials.2008.06.009.
- Inzana JA, Olvera D, Fuller SM, et al. 3D printing of composite calcium phosphate and collagen scaffolds for bone regeneration. Biomaterials. 2014;35(13): 4026-4034. doi: 10.1016/j.biomaterials.2014.01.064.
- Campbell PG, Miller ED, Fisher GW, Walker LM, Weiss LE. Engineered spatial patterns of FGF-2 immobilized on fibrin direct cell organization. Biomaterials. 2005;26(33): 6762-6770. doi: 10.1016/j.biomaterials.2005.04.032.
- Ilkhanizadeh S, Teixeira AI, Hermanson O, et al. Inkjet printing of macromolecules on hydrogels to steer neural stem cell differentiation. Biomaterials. 2007;28(27):3936-3943. doi: 10.1016/j.biomaterials.2007.05.018.
- Miller ED, Li K, Kanade T Weiss LE, Walker LM, Campbell PG. Spatially directed guidance of stem cell population migration by immobilized patterns of growth factors. Biomaterials. 2011;32(11):2775-2785. doi: 10.1016/j.biomaterials.2010.12.005.
- Xu T, Petridou S, Lee EH, et al. Construction of high-density bacterial colony arrays and patterns by the ink-jet method. Biotechnol Bioeng. 2004;85(1):29-33. doi: 10.1002/bit.10768.
- Merrin J, Leibler S, Chuang JS. Printing multistrain bacterial patterns with a piezoelectric inkjet printer. PLoS One. 2007;2(7):e663. doi: 10.1371/journal.pone.0000663.
- Yamazoe H, Tanabe T. Cell micropatterning on an albumin-based substrate using an inkjet printing technique. J Biomed Mater Res A. 2009;91(4):1202-1209. doi: 10.1002/jbm.a.32312.
- Arai K, Iwanaga S, Toda H, Genci C, Nishiyama Y, Nakamura M. Three-dimensional inkjet biofabrication based on designed images. Biofabrication. 2011;3(3):034113. doi: 10.1088/1758-5082/3/3/034113.
- Xu C, Chai W, Huang Y, Markwald RR. Scaffold-free inkjet printing of three-dimensional zigzag cellular tubes. Biotechnol Bioeng. 2012;109(12):3152-3160. doi: 10.1002/bit.24591.
- Xu T, Zhao W, Zhu JM, Albanna MZ, Yoo JJ, Atala, A. Complex heterogeneous tissue constructs containing multiple cell types prepared by inkjet printing technology. Biomaterials. 2013;34(1):130-139. doi: 10.1016/j.biomaterials.2012.09.035.
- Christensen K, Xu C, Chai W, Zhang Z, Fu J, Huang, Y. Freeform inkjet printing of cellular structures with bifurcations. Biotechnol Bioeng. 2015;112(5):1047-1055. doi: 10.1002/bit.25501.
- Li X, Liu B, Pei B, et al. Inkjet Bioprinting of Biomaterials. Chem Rev. 2020 14;120(19):10793-10833. doi: 10.1021/acs.chem rev.0c00008.
- Murphy SV, Atala A. 3D bioprinting of tissues and organs. Nat Biotechnol. 2014;32(8):773-785. doi: 10.1038/nbt.2958.
- Xu T, Jin J, Gregory C, Hickman JJ, Boland T. Inkjet printing of viable mammalian cells. Biomaterials. 2005;26(1): 93-99. doi: 10.1016/j.biomaterials.2004.04.011.
- Saunders RE, Gough JE, Derby B. Delivery of human fibroblast cells by piezoelectric drop-on-demand inkjet printing. Biomaterials. 2008;29(2):193-203. doi: 10.1016/j.biomaterials.2007.09.032.
- Nakamura M, Kobayashi A, Takagi F, et al. Biocompatible inkjet printing technique for designed seeding of individual living cells. Tissue Eng. 2005;11(11-12):1658-1666. doi: 10.1089/ten.2005.11.1658.
- Xu T, Kincaid H, Atala A, Yoo JJ. High-throughput production of single-cell microparticles using an inkjet printing technology. J Manuf Sci E. 2008;130(2):021017. doi: 10.1115/1.2903064.
- Kim YK, Park JA, Yoon WH, Kim J, Jung, S. Drop-on-demand inkjet-based cell printing with 30-μm nozzle diameter for cell-level accuracy. Biomicrofluidics. 2016 30;10(6):064110. doi: 10.1063/1.4968845.
- Stumpf F, Schoendube J, Gross A, et al. Single-cell PCR of genomic DNA enabled by automated single-cell printing for cell isolation. Biosens Bioelectron. 2015;69:301-306. doi: 10.1016/j.bios.2015.03.008.
- Chen F, Lin L, Zhang J, He Z, Uchiyama K, Lin, J.M. Single- Cell Analysis Using Drop-on-Demand Inkjet Printing and Probe Electrospray Ionization Mass Spectrometry. Anal Chem. 2016;88(8):4354-4360. doi: 10.1021/acs.analchem.5b04749.
- Dou R, Saunders RE, Mohamet L, Ward CM, Derby, B. High throughput cryopreservation of cells by rapid freezing of sub-μl drops using inkjet printing--cryoprinting. Lab Chip. 2015;15(17):3503-3513. doi: 10.1039/c5lc00674k.
- Akiyama Y, Shinose M, Watanabe H, Yamada S, Kanda Y. Cryoprotectant-free cryopreservation of mammalian cells by superflash freezing. Proc Natl Acad Sci USA. 2019;116(16):7738-7743. doi: 10.1073/pnas.1808645116.
- Xu T, Rohozinski J, Zhao W, Moorefield EC, Atala A, Yoo JJ. Inkjet-mediated gene transfection into living cells combined with targeted delivery. Tissue Eng Part A. 2009;15(1):95-101. doi: 10.1089/ten.tea.2008.0095.
- Cui X, Dean D, Ruggeri ZM, Boland T. Cell damage evaluation of thermal inkjet printed Chinese hamster ovary cells. Biotechnol Bioeng. 2010;106(6):963-969. doi: 10.1002/bit.22762.
- Sohrabi S, Liu Y. Modeling thermal inkjet and cell printing process using modified pseudopotential and thermal lattice Boltzmann methods. Phys Rev E. 2018;97(3-1):033105. doi: 10.1103/PhysRevE.97.033105.
- Das SR, Uz M, Ding S, et al. Electrical differentiation of mesenchymal stem cells into Schwann-Cell-Like phenotypes using inkjet-printed graphene circuits. Adv Healthc Mater. 2017;6(7):1601087. doi: 10.1002/adhm.201601087.
- Zips S, Huang B, Hotte S, et al. Aerosol Jet-Printed High- Aspect ratio Micro-Needle electrode arrays applied for human cerebral organoids and 3D neurospheroid networks. ACS Appl Mater Interfaces. 2023;15(30):35950-35961. doi: 10.1021/acsami.3c06210.
- Tse C, Whiteley R, Yu T, et al. Inkjet printing Schwann cells and neuronal analogue NG108-15 cells. Biofabrication. 2016;8(1):015017. doi: 10.1088/1758-5090/8/1/015017.
- Lorber B, Hsiao WK, Hutchings, IM, Martin KR. Adult rat retinal ganglion cells and glia can be printed by piezoelectric inkjet printing. Biofabrication. 2014;6(1):015001. doi: 10.1088/1758-5082/6/1/015001.
- Solis LH, Ayala Y, Portillo S, Varela-Ramirez A, Aguilera R, Boland, T. Thermal inkjet bioprinting triggers the activation of the VEGF pathway in human microvascular endothelial cells in vitro. Biofabrication. 2019;11(4):045005. doi: 10.1088/1758-5090/ab25f9.
- Yumoto M, Hemmi N, Sato N, et al. Evaluation of the effects of cell-dispensing using an inkjet-based bioprinter on cell integrity by RNA-seq analysis. Sci Rep. 2020;10(1):7158. doi: 10.1038/s41598-020-64193-z.
- Schlett K, Herberth B, Madarász E. In vitro pattern formation during neurogenesis in neuroectodermal progenitor cells immortalized by p53-deficiency. Int J Dev Neurosci. 1997;15(6):795-804. doi: 10.1016/s0736-5748(97)00015-4.
- Schlett K, Madarász E. Retinoic acid induced neural differentiation in a neuroectodermal cell line immortalized by p53 deficiency. J Neurosci Res. 1997;47(4):405-415.
- Strickland S, Mahdavi V. The induction of differentiation in teratocarcinoma stem cells by retinoic acid. Cell. 1978;15(2):393-403. doi: 10.1016/0092-8674(78)90008-9.
- Maden, M. Retinoic acid in the development, regeneration and maintenance of the nervous system. Nat Rev Neurosci. 2007;8(10):755-765. doi: 10.1038/nrn2212.
- Napoli JL. Interactions of retinoid binding proteins and enzymes in retinoid metabolism. Biochim Biophys Acta. 1999;1440(2-3):139-162. doi: 10.1016/s1388-1981(99)00117-1.
- Ables JL, Decarolis NA, Johnson MA, et al. Notch1 is required for maintenance of the reservoir of adult hippocampal stem cells. J Neurosci. 2010;30(31):10484-10492. doi: 10.1523/JNEUROSCI.4721-09.2010.
- Park JA, Yoon S, Kwon J, et al. Freeform micropatterning of living cells into cell culture medium using direct inkjet printing. Sci Rep. 2017;7(1):14610. doi: 10.1038/s41598-017-14726-w.
- Barui S, Saunders RE, Naskar S, Basu B, Derby B. Acoustic poration and dynamic healing of mammalian cell membranes during inkjet printing. ACS Biomater Sci Eng. 2020;6(1):749-757. doi: 10.1021/acsbiomaterials.9b01635.

