3D-bioprinted placenta-on-a-chip platform for modeling the human maternal–fetal barrier

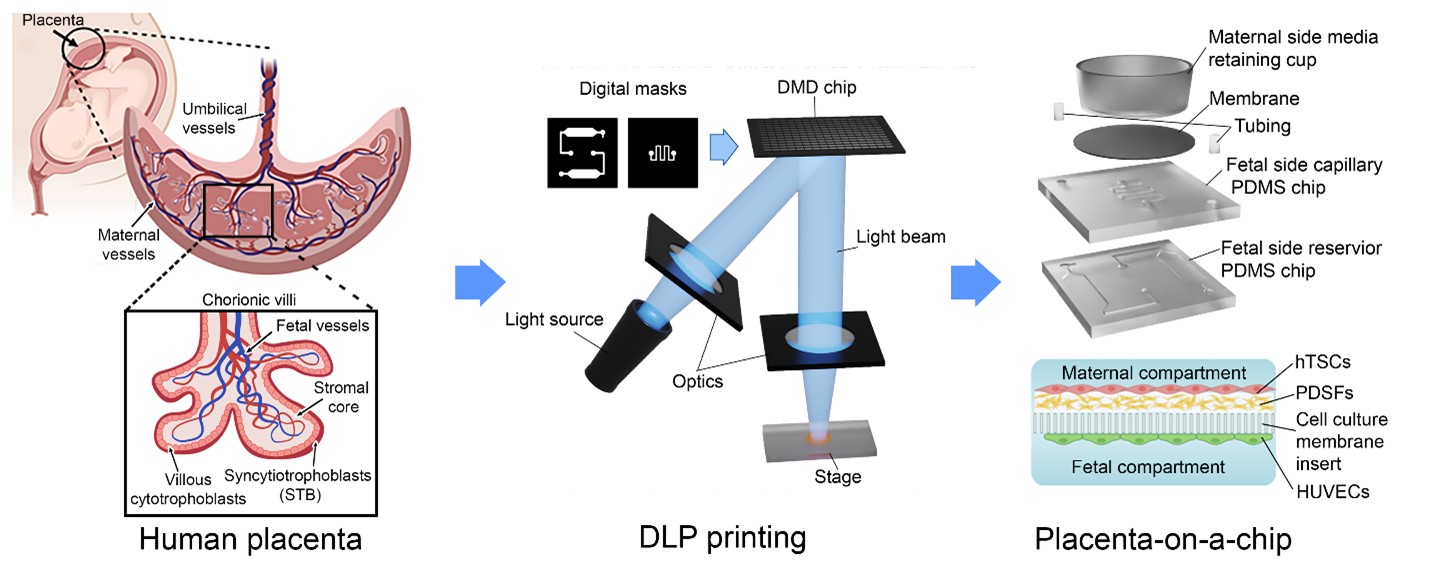
The placenta plays a vital role in pregnancy by regulating selective exchange between the maternal and fetal circulations and producing essential hormonal signals. In this study, we present an in vitro placenta-on-a-chip platform that leverages 3D bioprinting to replicate the structural and functional features of the human placental barrier. This microengineered system utilizes digital light processing-based 3D bioprinting to fabricate the microfluidic mold and construct 3D encapsulated cell cultures within a biomimetic hydrogel scaffold, enabling co-culture of three human cell types, including two derived from primary placental tissue. The system demonstrated excellent cell viability, high metabolic activity, placental hormone secretion, and native-like selective barrier transport properties. This system offers a versatile platform for experimental perturbations to explore mechanisms of normal placental function and identify contributors to placental dysfunction.
- Mitchell AA, Gilboa SM, Werler MM, Kelley KE, Louik C, Hernández-Díaz S. Medication use during pregnancy, with particular focus on prescription drugs: 1976–2008. Am J Obstetr Gynecol. 2011;205(1):51.e1-51.e8. doi: 10.1016/j.ajog.2011.02.029
- Wesley BD, Sewell CA, Chang CY, Hatfield KP, Nguyen CP. Prescription medications for use in pregnancy–perspective from the US Food and Drug Administration. Am J Obstetr Gynecol. 2021;225(1):21-32. doi: 10.1016/j.ajog.2021.02.032
- Zubizarreta ME, Xiao S. Bioengineering models of female reproduction. Bio Des Manuf. 2020;3(3):237. doi: 10.1007/s42242-020-00082-8
- Zabel RR, Favaro RR, Groten T, Brownbill P, Jones S. Ex vivo perfusion of the human placenta to investigate pregnancy pathologies. Placenta. 2022;130:1-8. doi: 10.1016/j.placenta.2022.10.006
- Glättli SC, Elzinga FA, van der Bijl W, et al. Variability in perfusion conditions and set-up parameters used in ex vivo human placenta models: a literature review. Placenta. 2024;157:37-49. doi: 10.1016/j.placenta.2024.03.007
- Carter AM. Animal models of human placentation – a review. Placenta. 2007;28:S41-S47. doi: 10.1016/j.placenta.2006.11.002
- Carter AM. Evolution of placental hormones: implications for animal models. Front Endocrinol. 2022;13:891927. doi: 10.3389/fendo.2022.891927
- Sood A, Kumar A, Gupta VK, Kim CM, Han SS. Translational nanomedicines across human reproductive organs modeling on microfluidic chips: state-of-the-art and future prospects. ACS Biomater Sci Eng. 2023;9(1):62-84. doi: 10.1021/acsbiomaterials.2c01080
- Pu Y, Gingrich J, Veiga-Lopez A. A 3-dimensional microfluidic platform for modeling human extravillous trophoblast invasion and toxicological screening. Lab Chip. 2021;21(3):546-557. doi: 10.1039/D0LC01013H
- Heaton SJ, Eady JJ, Parker ML, et al. The use of BeWo cells as an in vitro model for placental iron transport. Am J Physiol Cell Physiol. 2008;295(5):C1445-C1453. doi: 10.1152/ajpcell.00286.2008
- Park JY, Mani S, Clair G, et al. A microphysiological model of human trophoblast invasion during implantation. Nat Commun. 2022;13(1):1252. doi: 10.1038/s41467-022-28663-4
- Hori T, Okae H, Shibata S, et al. Trophoblast stem cell-based organoid models of the human placental barrier. Nat Commun. 2024;15(1):962. doi: 10.1038/s41467-024-45279-y
- Kallol S, Moser-Haessig R, Ontsouka CE, Albrecht C. Comparative expression patterns of selected membrane transporters in differentiated BeWo and human primary trophoblast cells. Placenta. 2018;72-73:48-52. doi: 10.1016/j.placenta.2018.10.008
- Cao R, Wang Y, Liu J, Rong L, Qin J. Self-assembled human placental model from trophoblast stem cells in a dynamic organ-on-a-chip system. Cell Prolif. 2023;56(5):e13469. doi: 10.1111/cpr.13469
- Cao R, Guo Y, Liu J, et al. Assessment of nanotoxicity in a human placenta-on-a-chip from trophoblast stem cells. Ecotoxicol Environ Saf. 2024;285:117051. doi: 10.1016/j.ecoenv.2024.117051
- Lermant A, Rabussier G, Davidson L, Lanz HL, Murdoch CE. Protocol for a placenta-on-a-chip model using trophoblasts differentiated from human induced pluripotent stem cells. STAR Protoc. 2024;5(1):102879. doi: 10.1016/j.xpro.2024.102879
- Wang Y, Guo Y, Wang P, et al. An engineered human placental organoid microphysiological system in a vascular niche to model viral infection. Commun Biol. 2025;8(1):669. doi: 10.1038/s42003-025-08057-0
- Pemathilaka RL, Caplin JD, Aykar SS, et al. Placenta-on-a- Chip: In Vitro Study of Caffeine Transport across Placental Barrier Using Liquid Chromatography Mass Spectrometry. Global Challenges. 2019;3(3):1800112. doi: 10.1002/gch2.201800112
- Ma Z, Sagrillo-Fagundes L, Mok S, Vaillancourt C, Moraes C. Mechanobiological regulation of placental trophoblast fusion and function through extracellular matrix rigidity. Sci Rep. 2020;10(1):5837. doi: 10.1038/s41598-020-62659-8
- Lee JS, Romero R, Han YM, et al. Placenta-on-a-chip: a novel platform to study the biology of the human placenta. J Matern Fetal Neonatal Med. 2016;29(7):1046-1054. doi: 10.3109/14767058.2015.1038518
- Ma X, Yu C, Wang P, et al. Rapid 3D bioprinting of decellularized extracellular matrix with regionally varied mechanical properties and biomimetic microarchitecture. Biomaterials. 2018;185:310-321. doi: 10.1016/j.biomaterials.2018.09.026
- Pyo SH, Wang P, Hwang HH, Zhu W, Warner J, Chen S. Continuous optical 3D printing of green aliphatic polyurethanes. ACS Appl Mater Interfaces. 2017;9(1):836-844. doi: 10.1021/acsami.6b12500
- Igura K, Zhang X, Takahashi K, Mitsuru A, Yamaguchi S, Takahashi TA. Isolation and characterization of mesenchymal progenitor cells from chorionic villi of human placenta. Cytotherapy. 2004;6(6):543-553. doi: 10.1080/14653240410005366-1
- Okae H, Toh H, Sato T, et al. Derivation of human trophoblast stem cells. Cell Stem Cell. 2018;22(1):50-63.e6. doi: 10.1016/j.stem.2017.11.004
- Bai T, Peng CY, Aneas I, et al. Establishment of human induced trophoblast stem-like cells from term villous cytotrophoblasts. Stem Cell Res. 2021;56:102507. doi: 10.1016/j.scr.2021.102507
- Zhu W, Qu X, Zhu J, et al. Direct 3D bioprinting of prevascularized tissue constructs with complex microarchitecture. Biomaterials. 2017;124:106-115. doi: 10.1016/j.biomaterials.2017.01.042
- Teasdale F. Gestational changes in the functional structure of the human placenta in relation to fetal growth: a morphometric study. Am J Obstetr Gynecol. 1980;137(5):560-568. doi: 10.1016/0002-9378(80)90696-1
- Blundell C, Yi YS, Ma L, et al. Placental drug transport-on-a-chip: a microengineered in vitro model of transporter-mediated drug efflux in the human placental barrier. Adv Healthc Mater. 2018;7(2):1700786. doi: 10.1002/adhm.201700786
- Wice B, Menton D, Geuze H, Schwartz AL. Modulators of cyclic AMP metabolism induce syncytiotrophoblast formation in vitro. Exp Cell Res. 1990;186(2):306-316. doi: 10.1016/0014-4827(90)90310-7
- Das S, Jegadeesan JT, Basu B. Gelatin methacryloyl (GelMA)-based biomaterial inks: process science for 3D/4D printing and current status. Biomacromolecules. 2024;25(4):2156-2221. doi: 10.1021/acs.biomac.3c01271.
- Tang C, Jin M, Ma B, et al. RGS2 promotes estradiol biosynthesis by trophoblasts during human pregnancy. Exp Mol Med. 2023;55(1):240-252. doi: 10.1038/s12276-023-00927-z
- Shutt DA, Smith ID, Shearman RP. Oestrone, oestradiol- 17beta and oestriol levels in human foetal plasma during gestation and at term. J Endocrinol. 1974;60(2):333-341. doi: 10.1677/joe.0.0600333
- Johnson MS, Jackson DL, Schust DJ. Endocrinology of pregnancy. In: Skinner MK, ed. Encyclopedia of Reproduction (Second Edition). Academic Press, Cambridge, MA, USA.; 2018:469-476. doi: 10.1016/B978-0-12-801238-3.64672-X
- Malek A, Sager R, Schneider H. Transport of proteins across the human placenta. Am J Reprod Immunol. 1998;40(5):347-351. doi: 10.1111/j.1600-0897.1998.tb00064.x
- Tal R, Taylor HS. Endocrinology of pregnancy. In: Feingold KR, Ahmed SF, Anawalt B, Blackman MR, Boyce A, Chrousos G, et al., eds. Endotext. MDText.com, Inc., South Dartmouth, MA, USA.; 2000. http://www.ncbi.nlm.nih.gov/books/NBK278962/. Accessed May 15, 2025.
- Kozler P, Pokorný J. Altered blood-brain barrier permeability and its effect on the distribution of Evans blue and sodium fluorescein in the rat brain applied by intracarotid injection. Physiol Res. 2003;52(5):607-14. doi: 10.33549/physiolres.930289
- Wevers NR, Nair AL, Fowke TM, et al. Modeling ischemic stroke in a triculture neurovascular unit on-a-chip. Fluids Barriers CNS. 2021;18(1):59. doi: 10.1186/s12987-021-00294-9
- Schaffenrath J, Huang SF, Wyss T, Delorenzi M, Keller A. Characterization of the blood–brain barrier in genetically diverse laboratory mouse strains. Fluids Barriers CNS. 2021;18(1):34. doi: 10.1186/s12987-021-00269-w
- Zambuto SG, Clancy KBH, Harley BAC. A gelatin hydrogel to study endometrial angiogenesis and trophoblast invasion. Interface Focus. 2019;9(5):20190016. doi: 10.1098/rsfs.2019.0016
- Kılıç F, Kayadibi Y, Yüksel MA, et al. Shear wave elastography of placenta: in vivo quantitation of placental elasticity in preeclampsia. Diagn Interv Radiol. 2015; 21(3):202-207. doi: 10.5152/dir.2014.14338
- Tarbell JM. Shear stress and the endothelial transport barrier. Cardiovasc Res. 2010;87(2):320-330. doi: 10.1093/cvr/cvq146
- Blundell C, Tess ER, Schanzer ASR, et al. A microphysiological model of the human placental barrier. Lab Chip. 2016;16(16):3065-3073. doi: 10.1039/C6LC00259E
- Boos JA, Misun PM, Brunoldi G, et al. Microfluidic co-culture platform to recapitulate the maternal–placental– embryonic axis. Adv Biol. 2021;5(8):2100609. doi: 10.1002/adbi.202100609
- Bhide A, Aboo A, Sawant M, Majumder A, Paul D, Modi D. Placenta on chip: a modern approach to probe feto-maternal interface. In: Mohanan PV, ed. Microfluidics and Multi Organs on Chip. Springer Nature, Singapore.; 2022:359-380. doi: 10.1007/978-981-19-1379-2_16
- Cole LA. Biological functions of hCG and hCG-related molecules. Reprod Biol Endocrinol. 2010;8(1):102. doi: 10.1186/1477-7827-8-102
- Brandes JM, Tavoloni N, Potter BJ, Sarkozi L, Shepard MD, Berk PD. A new recycling technique for human placental cotyledon perfusion: Application to studies of the fetomaternal transfer of glucose, inulin, and antipyrine. Am J Obstetr Gynecol. 1983;146(7):800-806. doi: 10.1016/0002-9378(83)91081-5
- Hahn T, Barth S, Weiss U, Mosgoeller W, Desoye G. Sustained hyperglycemia in vitro down-regulates the GLUT1 glucose transport system of cultured human term placental trophoblast: a mechanism to protect fetal development? FASEB J. 1998;12(12):1221-1231. doi: 10.1096/fasebj.12.12.1221
- Brownbill P, Sebire N, McGillick EV, Ellery S, Murthi P. Ex vivo dual perfusion of the human placenta: disease simulation, therapeutic pharmacokinetics and analysis of off-target effects. In: Murthi P, Vaillancourt C, eds. Preeclampsia: Methods and Protocols. Springer, New York, NY, USA.; 2018:173-189. doi: 10.1007/978-1-4939-7498-6_14
- Kouthouridis S, Saha P, Ludlow M, N. Truong BY, Zhang B. Late-stage placental barrier model for transport studies of prescription drugs during pregnancy. Lab Chip. 2025;25(13):3168-3184. doi: 10.1039/D5LC00075K
- Rabussier G, Bünter I, Bouwhuis J, et al. Healthy and diseased placental barrier on-a-chip models suitable for standardized studies. Acta Biomater. 2023;164:363-376. doi: 10.1016/j.actbio.2023.04.033
- Palmeira P, Quinello C, Silveira-Lessa AL, Zago CA, Carneiro-Sampaio M. IgG placental transfer in healthy and pathological pregnancies. J Immunol Res. 2012;2012(1):985646. doi: 10.1155/2012/985646
- Karakis V, Britt JW, Jabeen M, San Miguel A, Rao BM. Derivation of human trophoblast stem cells from placentas at birth. J Biol Chem. 2025;301(6):108505. doi: 10.1016/j.jbc.2025.108505

