Macro–micro analysis of the physical and biological properties of 3D-printed hydroxyapatite/β-tricalcium phosphate scaffolds with varying polyvinyl alcohol concentrations

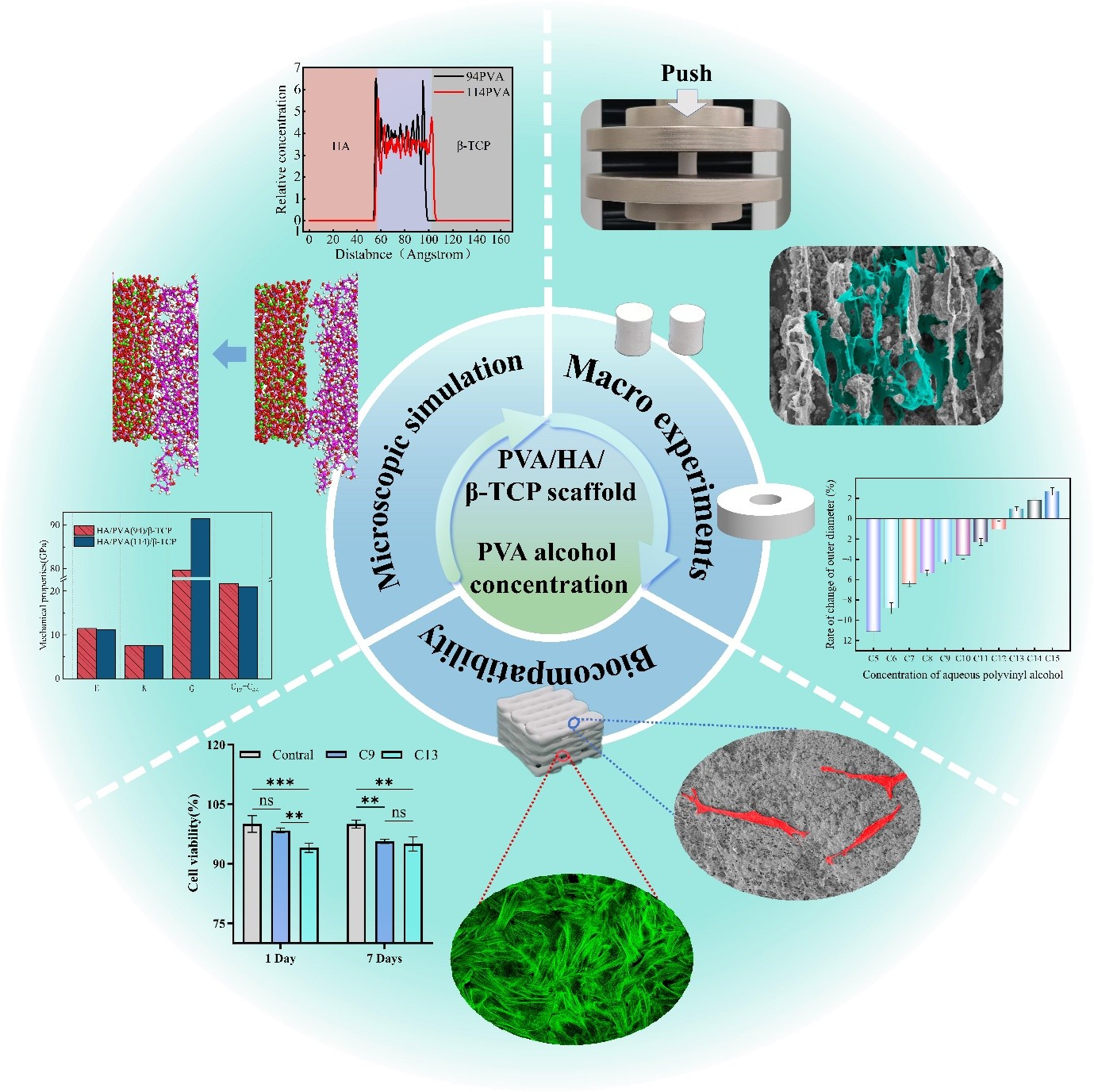
The concentration of the binder is a key factor affecting the quality of 3D-printed bone scaffolds. In this study, a macro–micro analysis was conducted to evaluate the effects of varying concentrations of polyvinyl alcohol (PVA) aqueous solution on the physical and biological properties of hydroxyapatite/β-tricalcium phosphate bone scaffolds. Both molecular dynamics (MD) simulations and experimental approaches were employed. The MD simulations analyzed microscopic interactions between PVA and ceramic powders by assessing changes in chain length at different concentrations. Experimentally, slurries containing 5–15% wt% PVA were characterized in terms of solid content, zeta potential, and extrusion rheology. Bone scaffolds were fabricated via 3D printing followed by freeze-drying, and their porosity, mechanical properties, dimensional shrinkage, and swelling behavior were examined. In vitro tests were conducted to assess biological performance. The results indicated that hydrogen and ionic bonding between PVA and ceramic powders were the primary mechanisms of adhesion. Increased chain length led to higher Cauchy pressure, thereby enhancing the mechanical properties of the material. Higher PVA concentrations produced slurries with increased solid content and shear-thinning capabilities, improving printability. The resulting bone scaffolds exhibited higher mechanical properties and shrinkage during drying but showed reduced porosity and swelling capability. In vitro experiments revealed that increasing PVA concentration decreased both the porosity and ion concentration of the bone scaffolds, thereby reducing their bioactivity. These findings provide a theoretical basis for optimizing binder concentration in 3D-printed bone scaffolds by linking slurry characteristics to scaffold performance.
- Thurzo A, Gálfiová P, Nováková Z, et al. Fabrication and in vitro characterization of novel hydroxyapatite scaffolds 3‑D printed using polyvinyl alcohol as a thermoplastic binder. Int J Mol Sci. 2022;23(23):14870. doi: 10.3390/ijms232314870
- Wascher DC, Bulthuis L. Extremity trauma: field management of sports injuries. Curr Rev Musculoskelet Med. 2014;7(4):387-393. doi: 10.1007/s12178-014-9242-y
- Xu C, Kang Y, Guan S, Dong X, Jiang D, Qi M. Iron-based metal–organic framework as a dual cooperative release system for enhanced vascularization and bone regeneration. Chinese Chem Lett. 2023;34:107825. doi: 10.1016/j.cclet.2022.107825
- Huang K, Liu G, Gu Z, Wu J. Tofu as excellent scaffolds for potential bone regeneration. Chinese Chem Lett. 2020;31:3190-3194. doi: 10.1016/j.cclet.2020.07.002
- Shi W, Jiang Y, Wu T, Zhang Y, Li T. Advancements in drug‑loaded hydrogel systems for bone defect repair. Regen Ther. 2024;25:174-185. doi: 10.1016/j.reth.2023.12.010
- Mauffrey C, Barlow BT, Smith W. Management of segmental bone defects. J Am Acad Orthop Surg. 2015;23(3): 143-153. doi: 10.5435/JAAOS-D-14-00018
- Faour O, Dimitriou R, Cousins CA, Giannoudis PV. The use of bone graft substitutes in large cancellous voids: any specific needs? Injury. 2011;42(Suppl 2):S87-S90. doi: 10.1016/j.injury.2011.06.020
- Yang DH, Nah H, Lee D, et al. A review on gold nanoparticles as an innovative therapeutic cue in bone tissue engineering: prospects and future clinical applications. Mater Today Bio. 2024;26:101016. doi: 10.1016/j.mtbio.2024.101016
- Kelly CN, Miller AT, Hollister SJ, Guldberg RE, Gall K. Design and structure–function characterization of 3‑D printed synthetic porous biomaterials for tissue engineering. Adv Healthcare Mater. 2018;7(1):1701095. doi: 10.1002/adhm.201701095
- Baniasadi H, Abidnejad R, Fazeli M, et al. Innovations in hydrogel‑based manufacturing: a comprehensive review of direct ink writing technique for biomedical applications. Adv Colloid Interface Sci. 2024;324:103095. doi: 10.1016/j.cis.2024.103095
- Murab S, Gupta A, Włodarczyk‑Biegun MK, et al. Alginate‑based hydrogel inks for 3‑D bioprinting of engineered orthopedic tissues. Carbohydr Polym. 2022;296:119964. doi: 10.1016/j.carbpol.2022.119964
- Li L, Lin Q, Tang M, Duncan AJ, Ke C. Advanced polymer designs for direct‑ink‑write 3‑D printing. Chem – Eur J. 2019;25(47):10768-10781. doi: 10.1002/chem.201900975
- Kumar A, Matari IAI, Han SS. 3‑D printable carboxylated cellulose nanocrystal‑reinforced hydrogel inks for tissue engineering. Biofabrication. 2020;12(2):025029. doi: 10.1088/1758-5090/ab736e
- Haring AP, Thompson EG, Tong Y, et al. Process‑and bio‑inspired hydrogels for 3‑D bioprinting of soft free‑standing neural and glial tissues. Biofabrication. 2019;11(2):025009. doi: 10.1088/1758-5090/ab02c9
- Fu Q, Saiz E, Tomsia AP. Direct ink writing of highly porous and strong glass scaffolds for load‑bearing bone defects repair and regeneration. Acta Biomater. 2011;7(10):3547-3554. doi: 10.1016/j.actbio.2011.06.030
- Jiang P, Yan C, Guo Y, et al. Direct ink writing with high‑strength and swelling‑resistant biocompatible physically crosslinked hydrogels. Biomater Sci. 2019;7(5):1805-1814. doi: 10.1039/C9BM00081J
- Ingwersen L‑C, Ahlhelm M, Schwarzer‑Fischer E, et al. Characterization of the osteogenic differentiation capacity of human bone cells on hybrid β‑TCP/ZrO₂ structures. Mater Design. 2024;238:112687. doi: 10.1016/j.matdes.2024.112687
- Agrawal V, Sinha M. A review on carrier systems for bone morphogenetic protein‑2. J Biomed Mater Res B Appl Biomater. 2017;105(4):904-925. doi: 10.1002/jbm.b.33599
- Lima DB, de Souza MAA, de Lima GG, et al. Injectable bone substitute based on chitosan with polyethylene glycol polymeric solution and biphasic calcium phosphate microspheres. Carbohydr Polym. 2020;245:116575. doi: 10.1016/j.carbpol.2020.116575
- Shakir M, Jolly R, Khan AA, et al. Resole‑based chitosan/ nano‑hydroxyapatite nanoensemble for effective bone tissue engineering. Carbohydr Polym. 2018;179:317-327. doi: 10.1016/j.carbpol.2017.09.103
- Yang Y, Yang S, Wang Y, et al. Anti‑infective efficacy, cytocompatibility and biocompatibility of a 3‑D‑printed osteoconductive composite scaffold functionalized with quaternized chitosan. Acta Biomater. 2016;46:112-128. doi: 10.1016/j.actbio.2016.09.035
- Anjaneyulu U, Priyadarshini B, Nirmala Grace A, Vijayalakshmi U. Fabrication and characterization of Ag doped hydroxyapatite–polyvinyl alcohol composite nanofibers and its in vitro biological evaluations for bone tissue engineering applications. J Sol‑Gel Sci Technol. 2017;81(3):750-761. doi: 10.1007/s10971-016-4243-5
- Nie L, Chen D, Suo J, et al. Physicochemical characterization and biocompatibility in vitro of biphasic calcium phosphate/ polyvinyl alcohol scaffolds prepared by freeze‑drying method for bone tissue engineering applications. Colloids Surf B Biointerfaces. 2012;100:169-176. doi: 10.1016/j.colsurfb.2012.04.046
- Bendtsen ST, Quinnell SP, Wei M. Development of a novel alginate‑polyvinyl alcohol‑hydroxyapatite hydrogel for 3‑D bioprinting bone tissue engineered scaffolds. J Biomed Mater Res A. 2017;105(6):1457-1468. doi: 10.1002/jbm.a.36036
- Ba Linh NT, Lee KH, Lee BT. Functional nanofiber mat of polyvinyl alcohol/gelatin containing nanoparticles of biphasic calcium phosphate for bone regeneration in rat calvaria defects. J Biomed Mater Res A. 2013;101(9):2412-2423. doi: 10.1002/jbm.a.34533
- Poursamar SA, Azami M, Mozafari M. Controllable synthesis and characterization of porous polyvinyl alcohol/hydroxyapatite nanocomposite scaffolds via an in situ colloidal technique. Colloids Surf B Biointerfaces. 2011;84(2):310-316. doi: 10.1016/j.colsurfb.2011.01.015
- Nie L, Deng Y, Li P, Hou R, Shavandi A, Yang S. Hydroxyethyl chitosan‑reinforced polyvinyl alcohol/biphasic calcium phosphate hydrogels for bone regeneration. ACS Omega. 2020;5(18):10948-10957. doi: 10.1021/acsomega.0c00727
- Wei Q, Wang Y, Li X, Yang M, Chai W, Wang K. Study the bonding mechanism of binders on hydroxyapatite surface and mechanical properties for 3‑DP fabrication bone scaffolds. J Mech Behav Biomed Mater. 2016;57:190-200. doi: 10.1016/j.jmbbm.2015.12.007
- Li Z, Lu H, Deng Y, Rao S, Huang X, Zhang Q. Advances in molecular dynamics simulation on heterogeneous nucleation of gas hydrate. Funct Mater Lett. 2021;14(1):2130010. doi: 10.1142/S1793604721300103
- Zhang L. Understanding the radiation resistance mechanisms of nanocrystalline metals from atomistic simulation. Metals. 2021;11(12):1875. doi: 10.3390/met11111875
- Róg T, Girych M, Bunker A. Mechanistic understanding from molecular dynamics in pharmaceutical research 2: lipid membrane in drug design. Pharmaceuticals. 2021;14(10):1062. doi: 10.3390/ph14101062
- Wei Q, Zhang Y, Wang Y, et al. Study of the effects of water content and temperature on polyacrylamide/polyvinyl alcohol interpenetrating network hydrogel performance by a molecular dynamics method. e‑Polymers. 2015;15(1):301- 309. doi: 10.1515/epoly-2015-0087

