Advances in biomanufacturing and medical applications of three-dimensional-printed organoids: A review

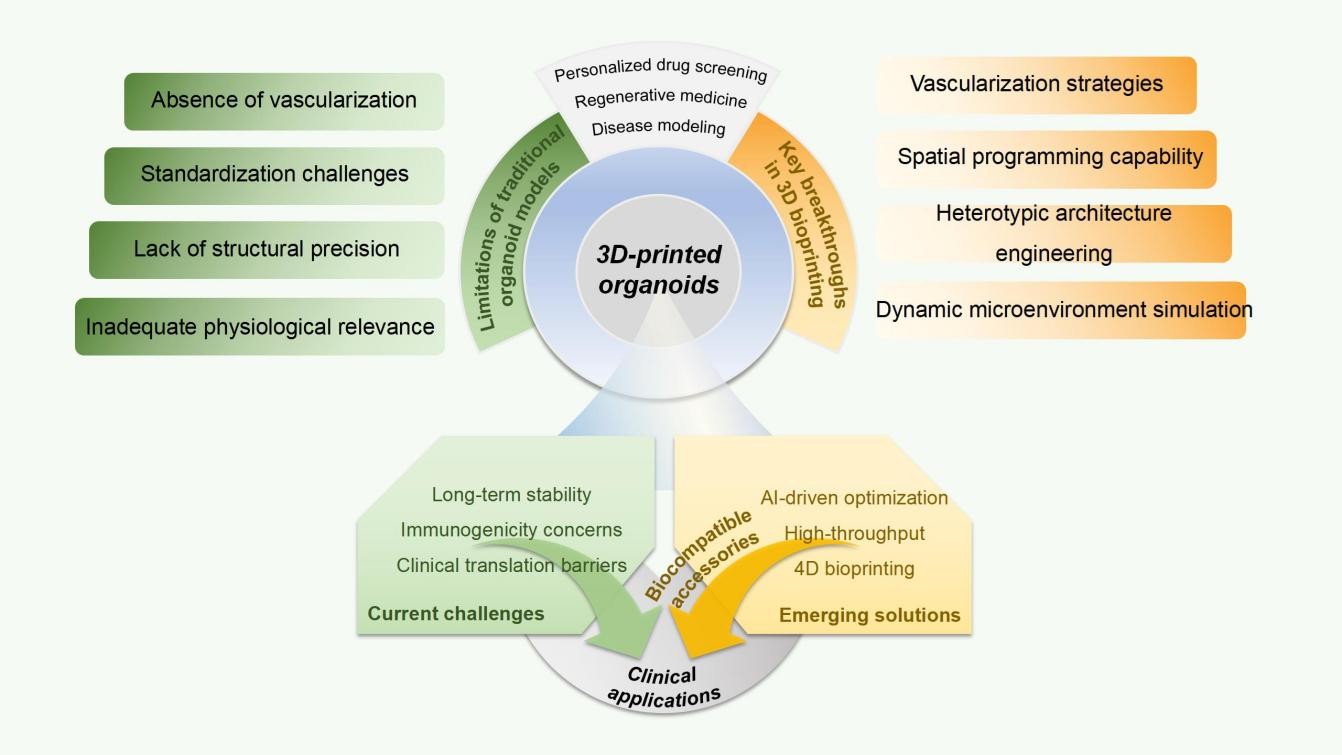
The emergence of organoid technology has bridged critical gaps between conventional two-dimensional cell cultures and in vivo systems by offering self-organized three-dimensional (3D) microtissues that recapitulate organ-specific architecture, cellular heterogeneity, and functional dynamics. However, traditional organoid models face inherent limitations in structural precision, scalability, and physiological relevance, particularly in replicating vascular networks, mechanical microenvironments, and multicellular interactions. Recent advancements in 3D bioprinting have enabled unprecedented spatial control over cellular and extracellular matrix organization, unlocking new frontiers in engineering organoids with enhanced biomimicry and functionality. This review systematically examines the integration of bioprinting technologies with organoid science, spanning biomaterial innovations, vascularization strategies, and dynamic microenvironmental cues that drive functional maturation. By synthesizing interdisciplinary advances in stem cell biology, materials science, and computational modeling, the work highlights applications across regenerative medicine, disease pathophysiology, and personalized drug screening. Key challenges, including immunogenicity, long-term stability, and clinical scalability, are critically evaluated alongside emerging solutions such as four-dimensional bioprinting, organ-on-chip integration, and artificial intelligence-driven bioink optimization. Through a comprehensive analysis of bioprinted organoids for physiology and 3D disease modeling, this review aims to establish a translational roadmap for leveraging spatially programmed organoids to address unmet clinical needs, revolutionize therapeutic development, and advance precision medicine.
- Zhao Z, Chen X, Dowbaj AM, et al. Organoids. Nat Rev Methods Primers. 2022;2(1):94. doi: 10.1038/s43586-022-00174-y
- Geng Y, Chen Z, Luo T, et al. Innovative construction and application of bile duct organoids: unraveling the complexity of bile duct diseases and potential therapeutic strategies. Cancer Lett. 2025;618:217619. doi: 10.1016/j.canlet.2025.217619
- Hermans F, Hasevoets S, Vankelecom H, Bronckaers A, Lambrichts I. From pluripotent stem cells to organoids and bioprinting: recent advances in dental epithelium and ameloblast models to study tooth biology and regeneration. Stem Cell Rev Rep. 2024;20(5):1184-1199. doi: 10.1007/s12015-024-10702-w
- Mallya D, Gadre MA, Varadharajan S, Vasanthan KS. 3D bioprinting for the construction of drug testing models-development strategies and regulatory concerns. Front Bioeng Biotechnol. 2025;13:1457872. doi: 10.3389/fbioe.2025.1457872
- Nejati B, Shahhosseini R, Hajiabbasi M, et al. Cancer-on-chip: a breakthrough organ-on-a-chip technology in cancer cell modeling. Med Biol Eng Comput. 2025;63(2):321-337. doi: 10.1007/s11517-024-03199-5
- Hu Y, Zhu T, Cui H, Cui H. Integrating 3D bioprinting and organoids to better recapitulate the complexity of cellular microenvironments for tissue engineering. Adv Healthc Mater. 2025;14(3):e2403762. doi: 10.1002/adhm.202403762
- Huang MS, Christakopoulos F, Roth JG, Heilshorn SC. Organoid bioprinting: from cells to functional tissues. Review. Nat Rev Bioeng. 2025;3(2):126-142. doi: 10.1038/s44222-024-00268-0
- Ju M, Jin Z, Yu X, et al. Gastric cancer models developed via GelMA 3D bioprinting accurately mimic cancer hallmarks, tumor microenvironment features, and drug responses. Small. 2025;21(8):e2409321. doi: 10.1002/smll.202409321
- Ding Z, Huang J, Ren Y, et al. 3D bioprinted advanced cartilage organoids with engineered magnetic nanoparticles polarized-BMSCs/alginate/gelatin for cartilage tissue regeneration. Article. Nano Res. 2025;18(2)94907084. doi: 10.26599/nr.2025.94907084
- Shyu J-F, Chu T-H, Lo Y-C, et al. Fabrication of 3D bioprinting vascularized bone organoid under compressive stimulation for study of osteogenesis and angiogenesis. Meeting Abstract. J Bone Miner Res. 2023;38:304-304.
- Abaci A, Camci-Unal G, Guvendiren M. Three-dimensional bioprinting for medical applications. Article. MRS Bulletin. 2023;48(6):624-631. doi: 10.1557/s43577-023-00546-z
- Shen N, Li Z, Yang P, et al. Designing methacrylic anhydride-based hydrogels for 3D bioprinting. IJB. 2024;11(1):84-138. doi: 10.36922/ijb.4650
- Chen Z, Zhang H, Huang J, et al. DNA-encoded dynamic hydrogels for 3D bioprinted cartilage organoids. Mater Today Bio. 2025;31:101509. doi: 10.1016/j.mtbio.2025.101509
- Shen C, Wang J, Li G, et al. Boosting cartilage repair with silk fibroin-DNA hydrogel-based cartilage organoid precursor. Article. Bioact Mater. 2024;35:429-444. doi: 10.1016/j.bioactmat.2024.02.016
- Ali EAM, Smaida R, Meyer M, et al. iPSCs chondrogenic differentiation for personalized regenerative medicine: a literature review. Stem Cell Res Ther. 2024;15(1):185. doi: 10.1186/s13287-024-03794-1
- Lawlor KT, Vanslambrouck JM, Higgins JW, et al. Cellular extrusion bioprinting improves kidney organoid reproducibility and conformation. Nat Mater. 2021;20(2):260-271. doi: 10.1038/s41563-020-00853-9
- Long B, Mengmeng L, Jiacan S. A perspective on light-based bioprinting of DNA hydrogels for advanced bone regeneration: implication for bone organoids. Int J Bioprint. 2023;9(2):688. doi: 10.18063/ijb.688
- Wang Z, Wang X, Huang Y, et al. Cav3.3-mediated endochondral ossification in a three-dimensional bioprinted GelMA hydrogel. Article. Bio-Des Manuf. 2024;7(6):983-999. doi: 10.1007/s42242-024-00287-1
- Wang J. Engineering large-scale self‐mineralizing bone organoids with bone matrix‐inspired hydroxyapatite hybrid bioinks. Adv Mater (Weinheim). 2024;36(30):e2309875. doi: 10.1002/adma.202309875
- Ren X, Wang J, Wu Y, et al. One-pot synthesis of hydroxyapatite hybrid bioinks for digital light processing 3D printing in bone regeneration. J Mater Sci Technol. 2024;188:84-97. doi: 10.1016/j.jmst.2024.01.001
- Wang J, Zhou D, Li R, et al. Protocol for engineering bone organoids from mesenchymal stem cells. Bioact Mater. 2025;45:388-400. doi: 10.1016/j.bioactmat.2024.11.017
- Li H, Chen H, Du C, et al. Effect of hydroxyapatite nanowires on formation and bioactivity of osteoblastic cell spheroid. ACS Biomater Sci Eng. 2024;10(12):7413-7428. doi: 10.1021/acsbiomaterials.4c01159
- Fang Y, Ji M, Wu B, et al. Engineering highly vascularized bone tissues by 3d bioprinting of granular prevascularized spheroids. ACS Appl Mater Interfaces. 2023;15(37):43492-43502. doi: 10.1021/acsami.3c08550
- Loukelis K, Koutsomarkos N, Mikos AG, Chatzinikolaidou M. Advances in 3D bioprinting for regenerative medicine applications. Regen Biomater. 2024;11:rbae033. doi: 10.1093/rb/rbae033
- Bernal PN, Bouwmeester M, Madrid-Wolff J, et al. Volumetric bioprinting of organoids and optically tuned hydrogels to build liver-like metabolic biofactories. Adv Mater (Deerfield Beach, Fla). 2022;34(15):e2110054. doi: 10.1002/adma.202110054
- Luo Y, Xu R, Hu Z, et al. Gel-based suspension medium used in 3D bioprinting for constructing tissue/organ analogs. Gels. 2024;10(10):644. doi: 10.3390/gels10100644
- De Leeuw A, Graf R, Zhang J, et al. Increased cell density increases mineral formation rates and stiffness in 3D bioprinted patient-derived bone organoids using dynamic loading. Meeting Abstract. Tissue Eng Part A. 2023;29(11-12):582-583.
- Wang J, Chen X, Li R, et al. Standardization and consensus in the development and application of bone organoids. Theranostics. 2025;15(2):682-706. doi: 10.7150/thno.105840
- Park S, Cho SW. Bioengineering toolkits for potentiating organoid therapeutics. Adv Drug Deliv Rev. 2024;208:115238. doi: 10.1016/j.addr.2024.115238
- Frenz-Wiessner S, Fairley SD, Buser M, et al. Generation of complex bone marrow organoids from human induced pluripotent stem cells. Article. Nat Methods. 2024;21(5). doi: 10.1038/s41592-024-02172-2
- De Leeuw A, Schadli GN, Steffi C, et al. A novel 3D-bioprinted patient-specific biomimetic bone organoid to model osteogenesis imperfecta. Meeting Abstract. Tissue Eng Part A. 2023;29(13-14):582-583.
- Abbott A. Cell culture: biology’s new dimension. Nature. 2003;424(6951):870-872. doi: 10.1038/424870a
- Li J, Han S, Yu F, Li T, Liao B, Liu F. Mapping the landscape of PSC-CM research through bibliometric analysis. Front Cardiovasc Med. 2024;11:1435874. doi: 10.3389/fcvm.2024.1435874
- Wang Y, Hou Y, Hao T, et al. Model construction and clinical therapeutic potential of engineered cardiac organoids for cardiovascular diseases. Biomater Transl. 2024;5(4):337-354. doi: 10.12336/biomatertransl.2024.04.002
- Nwokoye PN, Abilez OJ. Blood vessels in a dish: the evolution, challenges, and potential of vascularized tissues and organoids. Front Cardiovasc Med. 2024;11:1336910. doi: 10.3389/fcvm.2024.1336910
- Khoury RE, Nagiah N, Mudloff JA, Thakur V, Chattopadhyay M, Joddar B. 3D bioprinted spheroidal droplets for engineering the heterocellular coupling between cardiomyocytes and cardiac fibroblasts. Cyborg Bionic Syst. 2021;2021:9864212. doi: 10.34133/2021/9864212
- Mohr E, Thum T, Bär C. Accelerating cardiovascular research: recent advances in translational 2D and 3D heart models. Eur J Heart Fail. Oct 2022;24(10):1778-1791. doi: 10.1002/ejhf.2631
- Zhang W, Chen Y, Li M, et al. A PDA-functionalized 3d lung scaffold bioplatform to construct complicated breast tumor microenvironment for anticancer drug screening and immunotherapy. Adv Sci (Weinh). 2023;10(26):e2302855. doi: 10.1002/advs.202302855
- Li S, Li J, Xu J, et al. Removal-free and multicellular suspension bath-based 3D bioprinting. Adv Mater (Deerfield Beach, Fla). 2024;36(48):e2406891. doi: 10.1002/adma.202406891
- Hoang P, Sun S, Tarris BA, Ma Z. Controlling morphology and functions of cardiac organoids by two-dimensional geometrical templates. Cells Tissues Organs. 2023;212(1):64-73. doi: 10.1159/000521787
- Noël ES. Cardiac construction—recent advances in morphological and transcriptional modeling of early heart development. Curr Top Dev Biol. 2024;156:121-156. doi: 10.1016/bs.ctdb.2024.02.005
- Fang Y, Guo Y, Wu B, et al. Expanding embedded 3D bioprinting capability for engineering complex organs with freeform vascular networks. Adv Mater (Deerfield Beach, Fla). 2023;35(22):e2205082. doi: 10.1002/adma.202205082
- Cui H, Liu C, Esworthy T, et al. 4D physiologically adaptable cardiac patch: A 4-month in vivo study for the treatment of myocardial infarction. Sci Adv. 2020;6(26):eabb5067. doi: 10.1126/sciadv.abb5067
- Zhang Z, Wu C, Dai C, et al. A multi-axis robot-based bioprinting system supporting natural cell function preservation and cardiac tissue fabrication. Bioact Mater. 2022;18:138-150. doi: 10.1016/j.bioactmat.2022.02.009
- Fareez UNM, Naqvi SAA, Mahmud M, Temirel M. Computational fluid dynamics (CFD) analysis of bioprinting. Adv Healthc Mater. 2024;13(20):e2400643. doi: 10.1002/adhm.202400643
- Abolhassani S, Fattahi R, Safshekan F, Saremi J, Hasanzadeh E. Advances in 4D bioprinting: the next frontier in regenerative medicine and tissue engineering applications. Adv Healthc Mater. 2025;14(4):e2403065. doi: 10.1002/adhm.202403065
- Wu J, Fu J. Toward developing human organs via embryo models and chimeras. Cell. 2024;187(13):3194-3219. doi: 10.1016/j.cell.2024.05.027
- Wu Y, Qin M, Yang X. Organ bioprinting: progress, challenges and outlook. J Mater Chem B. 2023;11(43):10263-10287. doi: 10.1039/d3tb01630g
- Roza Vaez G, Ileana LC, Matthew CM, Vikramaditya GY. Brain organoids: a new, transformative investigational tool for neuroscience research. Adv Biosyst. 2018;3(1):174. doi: 10.1002/adbi.201800174
- Renjitha G, Rakhi P. Bioengineering of brain organoids: advancements and challenges. Tissue Eng. 2022:399-414. doi: 10.1016/b978-0-12-824064-9.00002-2
- Madeline AL, Magdalena R, Carol-Anne M, et al. Cerebral organoids model human brain development and microcephaly. Nature. 2013;501(7467):373-379. doi: 10.1038/nature12517
- Jeong E, Choi S, Cho SW. Recent advances in brain organoid technology for human brain research. ACS Appl Mater Interfaces. 2023;15(1):200-219. doi: 10.1021/acsami.2c17467
- Cadena MA, Sing A, Taylor K, et al. A 3D bioprinted cortical organoid platform for modeling human brain development. Adv Healthc Mater. 2024;13(27):e2401603. doi: 10.1002/adhm.202401603
- Jihoon K, Sujin H, Sunghun C, Yoojin C, Noo Li J. Revealing the clinical potential of high-resolution organoids. Adv Drug Deliv Rev. 2024;207:115202. doi: 10.1016/j.addr.2024.115202
- Natan Roberto de B, Canran W, Surjendu M, et al. Engineered organoids for biomedical applications. Adv Drug Deliv Rev. 2023;203:115142. doi: 10.1016/j.addr.2023.115142
- Zhe L, Weizi G, Fukang L, et al. Vat photopolymerization based digital light processing 3D printing hydrogels in biomedical fields: key parameters and perspective. Addit Manuf. 2024;94:104443. doi: 10.1016/j.addma.2024.104443
- Jennifer Sally S, Anuradha R, Venkatachalam Deepa P. Development of midbrain dopaminergic neurons and the advantage of using hiPSCs as a model system to study Parkinson’s disease. Neuroscience. 2024;546:1-19. doi: 10.1016/j.neuroscience.2024.03.025
- Yan Y, Li X, Gao Y, et al. 3D bioprinting of human neural tissues with functional connectivity. Cell Stem Cell. 2024;31(2):260-274.e7. doi: 10.1016/j.stem.2023.12.009
- Layrolle P, Payoux P, Chavanas S. Message in a scaffold: natural biomaterials for three-dimensional (3D) bioprinting of human brain organoids. Biomolecules. 2022;13(1):25. doi: 10.3390/biom13010025
- Gabriel E, Albanna W, Pasquini G, et al. Human brain organoids assemble functionally integrated bilateral optic vesicles. Cell Stem Cell. 2021;28(10):1740-1757.e8. doi: 10.1016/j.stem.2021.07.010
- Jing G, Jiahui K, Minghui L, Xiao L, Jun Y, Haiwei X. Applications of neural organoids in neurodevelopment and regenerative medicine. Biomed Eng. 2022. doi: 10.5772/intechopen.104044
- Tariku Sinshaw T, Frehiwot Bayelign T, Xijin H, et al. A review of advances in 3D and 4D bioprinting: toward mass individualization paradigm. J Intell Manuf. 2024. doi: 10.1007/s10845-024-02529-6
- Wang X, Yang X, Liu Z, et al. 3D bioprinting of an in vitro hepatoma microenvironment model: establishment, evaluation, and anticancer drug testing. Acta Biomater. 2024;185:173-189. doi: 10.1016/j.actbio.2024.07.019
- Zhuang X, Deng G, Wu X, et al. Recent advances of three-dimensional bioprinting technology in hepato-pancreato-biliary cancer models. Front Oncol. 2023;13:1143600. doi: 10.3389/fonc.2023.1143600
- Kim MH, Singh YP, Celik N, et al. High-throughput bioprinting of spheroids for scalable tissue fabrication. Nat Commun. 2024;15(1):10083. doi: 10.1038/s41467-024-54504-7
- Lekkala VKR, Shrestha S, Al Qaryoute A, et al. Enhanced maturity and functionality of vascular human liver organoids through 3D bioprinting and pillar plate culture. ACS Biomater Sci Eng. 2025;11(1):506-517. doi: 10.1021/acsbiomaterials.4c01658
- Kang SY, Kimura M, Shrestha S, et al. A pillar and perfusion plate platform for robust human organoid culture and analysis. Adv Healthc Mater. 2024;13(21):e2302502. doi: 10.1002/adhm.202302502
- Shrestha S, Lekkala VKR, Acharya P, Kang SY, Vanga MG, Lee MY. Reproducible generation of human liver organoids (HLOs) on a pillar plate platform via microarray 3D bioprinting. Lab Chip. 2024;24(10):2747-2761. doi: 10.1039/d4lc00149d
- Gao Z, Liu X, Zhao H, et al. Synthesis of easily-processable collagen bio-inks using ionic liquid for 3D bioprinted liver tissue models with branched vascular networks. Sci China Chem. 2023;66(5):1489-1499. doi: 10.1007/s11426-022-1472-6
- Yan J, Ye Z, Lu Y, et al. 3D bioprinting lobule-like hepatorganoids with induced vascularization for orthotopic implantation. Mater Today Bio. 2025;31:101515. doi: 10.1016/j.mtbio.2025.101515
- Brumberg VA, Bikmulina PY, Pozdnyakov AA, et al. Scaling liver bioprinting: a guide for usage of the hepatic extracellular matrix as a bioink. Review. Int J Bioprint. 2025;11(1):57-83. doi: 10.36922/ijb.4343
- Zhang Y, Li L, Dong L, et al. Hydrogel-based strategies for liver tissue engineering. Chem Bio Eng. 2024;1(11):887-915. doi: 10.1021/cbe.4c00079
- Cross-Najafi AA, Farag K, Chen AM, et al. The long road to develop custom-built livers: current status of 3D liver bioprinting. Transplantation. 2024;108(2):357-368. doi: 10.1097/tp.0000000000004668
- Li W, Liu Z, Tang F, et al. Application of 3D bioprinting in liver diseases. Micromachines (Basel). 2023;14(8):1648. doi: 10.3390/mi14081648
- Willemse J, van der Laan LJW, de Jonge J, Verstegen MMA. Design by nature: emerging applications of native liver extracellular matrix for cholangiocyte organoid-based regenerative medicine. Bioengineering (Basel). 2022;9(3):110. doi: 10.3390/bioengineering9030110
- Shi W, Zhang Z, Wang X. The prospect of hepatic decellularized extracellular matrix as a bioink for liver 3D bioprinting. Biomolecules. 2024;14(8):1019. doi: 10.3390/biom14081019
- Kim Y, Kang M, Mamo MG, Adisasmita M, Huch M, Choi D. Liver organoids: current advances and future applications for hepatology. Clin Mol Hepatol. 2025;31(Suppl):S327-S348. doi: 10.3350/cmh.2024.1040
- Li G, He J, Shi J, et al. Bioprinting functional hepatocyte organoids derived from human chemically induced pluripotent stem cells to treat liver failure. Gut. 2025; 74(7):1150-1164. doi: 10.1136/gutjnl-2024-333885
- Sun H, Sun L, Ke X, et al. Prediction of clinical precision chemotherapy by patient-derived 3D bioprinting models of colorectal cancer and its liver metastases. Adv Sci (Weinh). 2024;11(2):e2304460. doi: 10.1002/advs.202304460
- Chen F, Wei X, Chen K, Wang L, Xu M. Massive fabrication of functional hepatic cancer spheroids by micropatterned GelMA hydrogel chip for drug screening. Colloids Surf B Biointerfaces. 2024;244:114171. doi: 10.1016/j.colsurfb.2024.114171
- Yeo M, Sarkar A, Singh YP, Derman ID, Datta P, Ozbolat IT. Synergistic coupling between 3D bioprinting and vascularization strategies. Biofabrication. 2023;16(1):e2302506. doi: 10.1088/1758-5090/ad0b3f
- Reza HA, Santangelo C, Iwasawa K, et al. Multi-zonal liver organoids from human pluripotent stem cells. Nature. 2025; 641(8065):1258-1267. doi: 10.1038/s41586-025-08850-1
- Falandt M, Bernal PN, Longoni A, et al. Hybrid supramolecular-covalent bioresin promotes cell migration and self-assembly in light-based volumetric bioprinted constructs. preprint. bioRxiv. 2025. doi: 10.1101/2025.01.06.631505
- Skylar-Scott MA, Huang JY, Lu A, et al. Orthogonally induced differentiation of stem cells for the programmatic patterning of vascularized organoids and bioprinted tissues. Nature Biomed Eng. 2022;6(4):449-462. doi: 10.1038/s41551-022-00856-8
- Urkasemsin G, Rungarunlert S, Ferreira JN. Bioprinting strategies for secretory epithelial organoids. Methods Mol Biol. 2020;2140:243-249. doi: 10.1007/978-1-0716-0520-2_16
- Klangprapan J, Souza GR, Ferreira JN. Bioprinting salivary gland models and their regenerative applications. BDJ Open. 2024;10(1):39. doi: 10.1038/s41405-024-00219-2
- Liu N, Huang S, Yao B, Xie J, Wu X, Fu X. 3D bioprinting matrices with controlled pore structure and release function guide in vitro self-organization of sweat gland. Sci Rep. 2016;6:34410. doi: 10.1038/srep34410
- Dai R, Chen W, Chen Y, et al. 3D bioprinting platform development for high-throughput cancer organoid models construction and drug evaluation. Biofabrication. 2024;16(3):34410. doi: 10.1088/1758-5090/ad51a6
- Shiwarski DJ, Hudson AR, Tashman JW, et al. 3D bioprinting of collagen-based microfluidics for engineering fully-biologic tissue systems. bioRxiv. 2024. doi: 10.1101/2024.01.26.577422
- Maciel BR, Grimm A, Oelschlaeger C, Schepers U, Willenbacher N. Targeted micro-heterogeneity in bioinks allows for 3D printing of complex constructs with improved resolution and cell viability. Biofabrication. 2023;15(4):042004. doi: 10.1088/1758-5090/acee22
- Markstedt K, Mantas A, Tournier I, Martínez Ávila H, Hägg D, Gatenholm P. 3D bioprinting human chondrocytes with nanocellulose-alginate bioink for cartilage tissue engineering applications. Biomacromolecules. 2015;16(5):1489-96. doi: 10.1021/acs.biomac.5b00188
- Maharjan S, Ma C, Singh B, et al. Advanced 3D imaging and organoid bioprinting for biomedical research and therapeutic applications. Adv Drug Deliv Rev. 2024;208:115237. doi: 10.1016/j.addr.2024.115237
- Gugulothu SB, Chatterjee K. Visible light-based 4D-bioprinted tissue scaffold. ACS Macro Lett. 2023;12(4):494-502. doi: 10.1021/acsmacrolett.3c00036
- Wang D, Guo Y, Zhu J, et al. Hyaluronic acid methacrylate/ pancreatic extracellular matrix as a potential 3D printing bioink for constructing islet organoids. Acta Biomater. 2023;165:86-101. doi: 10.1016/j.actbio.2022.06.036
- Kim M, Cho S, Hwang DG, et al. Bioprinting of bespoke islet-specific niches to promote maturation of stem cell-derived islets. Nat Commun. 2025;16(1):1430. doi: 10.1038/s41467-025-56665-5
- Reid JA, Mollica PA, Bruno RD, Sachs PC. Consistent and reproducible cultures of large-scale 3D mammary epithelial structures using an accessible bioprinting platform. Breast Cancer Res. 2018;20(1):122. doi: 10.1186/s13058-018-1045-4
- Shi W, Mirza S, Kuss M, et al. Embedded bioprinting of breast tumor cells and organoids using low-concentration collagen-based bioinks. Adv Healthc Mater. 2023;12(26):e2300905. doi: 10.1002/adhm.202300905
- Zhang Y, Li G, Wang J, Zhou F, Ren X, Su J. Small joint organoids 3D bioprinting: construction strategy and application. Small. 2024;20(8):e2302506. doi: 10.1002/smll.202302506
- Bertassoni LE. Bioprinting of complex multicellular organs with advanced functionality-recent progress and challenges ahead. Adv Mater (Deerfield Beach, Fla). 2022;34(3):e2101321. doi: 10.1002/adma.202101321
- Freedman BS, Brooks CR, Lam AQ, et al. Modelling kidney disease with CRISPR-mutant kidney organoids derived from human pluripotent epiblast spheroids. Nat Commun. 2015;6:8715. doi: 10.1038/ncomms9715
- Votanopoulos KI, Forsythe S, Sivakumar H, et al. Model of patient-specific immune-enhanced organoids for immunotherapy screening: feasibility study. Ann Surg Oncol. 2020;27(6):1956-1967. doi: 10.1245/s10434-019-08143-8
- Childs CJ, Poling HM, Chen K, et al. Coordinated differentiation of human intestinal organoids with functional enteric neurons and vasculature. Cell Stem Cell. 2025;32(4):640-651.e9. doi: 10.1016/j.stem.2025.02.007
- Park HS, Park JH, Oh M-K, Yu K-R. Advancements in 3D bioprinting for precision medicine: enhancing patient-derived organoids and extracellular vesicle applications in inflammatory diseases. Article. Int J Bioprint. 2024;10(5)4054. doi: 10.36922/ijb.4054
- Datta P, Dey M, Ataie Z, Unutmaz D, Ozbolat IT. 3D bioprinting for reconstituting the cancer microenvironment. NPJ Precis Oncol. 2020;4:18. doi: 10.1038/s41698-020-0121-2
- Augustine R, Kalva SN, Ahmad R, et al. 3D bioprinted cancer models: revolutionizing personalized cancer therapy. Transl Oncol. 2021;14(4):101015. doi: 10.1016/j.tranon.2021.101015
- Qazi TH, Blatchley MR, Davidson MD, et al. Programming hydrogels to probe spatiotemporal cell biology. Cell Stem Cell. 2022;29(5):678-691. doi: 10.1016/j.stem.2022.03.013
- Gopalakrishnan S, Bakke I, Hansen MD, et al. Comprehensive protocols for culturing and molecular biological analysis of IBD patient-derived colon epithelial organoids. Front Immunol. 2023;14:1097383. doi: 10.3389/fimmu.2023.1097383
- Caire R, Audoux E, Courbon G, et al. YAP/TAZ: key players for rheumatoid arthritis severity by driving fibroblast like synoviocytes phenotype and fibro-inflammatory response. Front Immunol. 2021;12:791907. doi: 10.3389/fimmu.2021.791907
- Günther C, Winner B, Neurath MF, Stappenbeck TS. Organoids in gastrointestinal diseases: from experimental models to clinical translation. Gut. 2022;71(9): 1892-1908. doi: 10.1136/gutjnl-2021-326560
- Sachs N, Tsukamoto Y, Kujala P, Peters PJ, Clevers H. Intestinal epithelial organoids fuse to form self-organizing tubes in floating collagen gels. Development. 2017;144(6):1107-1112. doi: 10.1242/dev.143933
- Niklinska-Schirtz BJ, Venkateswaran S, Anbazhagan M, et al. Ileal derived organoids from Crohn’s disease patients show unique transcriptomic and secretomic signatures. Cell Mol Gastroenterol Hepatol. 2021;12(4):1267-1280. doi: 10.1016/j.jcmgh.2021.06.018
- Taebnia N, Zhang R, Kromann EB, Dolatshahi-Pirouz A, Andresen TL, Larsen NB. Dual-material 3D-printed intestinal model devices with integrated villi-like scaffolds. ACS Appl Mater Interfaces. 2021;13(49): 8434-58446. doi: 10.1021/acsami.1c22185
- Brassard JA, Nikolaev M, Hübscher T, Hofer M, Lutolf MP. Recapitulating macro-scale tissue self-organization through organoid bioprinting. Nat Mater. 2021;20(1):22-29. doi: 10.1038/s41563-020-00803-5
- Carvalho MR, Yan L-P, Li B, et al. Gastrointestinal organs and organoids-on-a-chip: advances and translation into the clinics. Review. Biofabrication. 2023;15(4):042004. doi: 10.1088/1758-5090/acf8fb
- Xiaoshuai L, Qiushi W, Rui W. Advantages of CRISPR-Cas9 combined organoid model in the study of congenital nervous system malformations. Front Bioeng Biotechnol. 2022;10:932936. doi: 10.3389/fbioe.2022.932936
- Gopal S, Rodrigues AL, Dordick JS. Exploiting CRISPR Cas9 in three-dimensional stem cell cultures to model disease. Front Bioeng Biotechnol. 2020;8:692. doi: 10.3389/fbioe.2020.00692
- Schene IF, Joore IP, Oka R, et al. Prime editing for functional repair in patient-derived disease models. Nat Commun. 2020;11(1):5352. doi: 10.1038/s41467-020-19136-7
- Inak G, Rybak-Wolf A, Lisowski P, et al. Defective metabolic programming impairs early neuronal morphogenesis in neural cultures and an organoid model of Leigh syndrome. Nat Commun. 2021;12(1):1929. doi: 10.1038/s41467-021-22117-z
- Zhang W, Ma L, Yang M, et al. Cerebral organoid and mouse models reveal a RAB39b-PI3K-mTOR pathway-dependent dysregulation of cortical development leading to macrocephaly/autism phenotypes. Genes Dev. 2020;34(7-8):580-597. doi: 10.1101/gad.332494.119
- An HL, Kuo HC, Tang TK. Modeling human primary microcephaly with hiPSC-derived brain organoids carrying CPAP-E1235V disease-associated mutant protein. Front Cell Dev Biol. 2022;10:830432. doi: 10.3389/fcell.2022.830432
- Cidonio G, Glinka M, Dawson JI, Oreffo ROC. The cell in the ink: Improving biofabrication by printing stem cells for skeletal regenerative medicine. Biomater. 2019;209:10-24. https://doi.org/10.1016/j.biomaterials.2019.04.009
- Ahn CB, Lee J-H, Kim JH, et al. Development of a 3D subcutaneous construct containing insulin-producing beta cells using bioprinting. Bio-Des Manuf. 2022;5(2): 265-276. doi: 10.1007/s42242-021-00178-9
- Enrico A, Voulgaris D, Ostmans R, et al. 3D microvascularized tissue models by laser-based cavitation molding of collagen. Adv Mater (Deerfield Beach, Fla). 2022;34(11):e2109823. doi: 10.1002/adma.202109823
- Bai L, Zhou D, Li G, Liu J, Chen X, Su J. Engineering bone/ cartilage organoids: strategy, progress, and application. Bone Res. 2024;12(1):66. doi: 10.1038/s41413-024-00376-y
- O’Connor C, Brady E, Zheng Y, Moore E, Stevens KR. Engineering the multiscale complexity of vascular networks. Nat Rev Mater. 2022;7(9):702-716. doi: 10.1038/s41578-022-00447-8
- Michael S, Sorg H, Peck CT, et al. Tissue engineered skin substitutes created by laser-assisted bioprinting form skin-like structures in the dorsal skin fold chamber in mice. PLoS One. 2013;8(3):e57741. doi: 10.1371/journal.pone.0057741
- Rioux G, Simard M, Morin S, Lorthois I, Guérin SL, Pouliot R. Development of a 3D psoriatic skin model optimized for infiltration of IL-17A producing T cells: focus on the crosstalk between T cells and psoriatic keratinocytes. Acta Biomater. 2021;136:210-222. doi: 10.1016/j.actbio.2021.09.018
- Shin JU, Abaci HE, Herron L, et al. Recapitulating T cell infiltration in 3D psoriatic skin models for patient-specific drug testing. Sci Rep. 2020;10(1):4123. doi: 10.1038/s41598-020-60275-0
- Lorthois I, Simard M, Morin S, Pouliot R. Infiltration of T cells into a three-dimensional psoriatic skin model mimics pathological key features. Int J Mol Sci. 2019;20(7):1670. doi: 10.3390/ijms20071670
- Gong L, Li J, Zhang J, et al. An interleukin-4-loaded bi-layer 3D printed scaffold promotes osteochondral regeneration. Acta Biomater. 2020;117:246-260. doi: 10.1016/j.actbio.2020.09.039
- Derman ID, Rivera T, Garriga Cerda L, et al. Advancements in 3D skin bioprinting: processes, bioinks, applications and sensor integration. Int J Extrem Manuf. 2025;7(1):012009. doi: 10.1088/2631-7990/ad878c
- Zhou Z, Pang Y, Ji J, et al. Harnessing 3D in vitro systems to model immune responses to solid tumours: a step towards improving and creating personalized immunotherapies. Nat Rev Immunol. 2024;24(1):18-32. doi: 10.1038/s41577-023-00896-4
- Zhao K-y, Du Y-x, Cao H-m, Su L-y, Su X-l, Li X. The biological macromolecules constructed Matrigel for cultured organoids in biomedical and tissue engineering. Article. Colloids Surf B Biointerfaces. 2025;247:114435. doi: 10.1016/j.colsurfb.2024.114435
- Di Piazza E, Pandolfi E, Cacciotti I, et al. Bioprinting technology in skin, heart, pancreas and cartilage tissues: progress and challenges in clinical practice. Int J Environ Res Public Health. 2021;18(20):10806. doi: 10.3390/ijerph182010806
- Wang Y, Li H, Zhang J, Chen M, Pan Y, Lou X. 3D bioprinting inner ear organ of corti organoids induce hair cell regeneration. J Biomed Mater Res A. 2025;113(3): e37892. doi: 10.1002/jbm.a.37892
- Shukla P, Yeleswarapu S, Heinrich MA, Prakash J, Pati F. Mimicking tumor microenvironment by 3D bioprinting: 3D cancer modeling. Biofabrication. 2022;14(3):6d11. doi: 10.1088/1758-5090/ac6d11
- Lee Y, Min J, Kim S, Park W, Ko J, Jeon NL. Recapitulating the cancer-immunity cycle on a chip. Adv Healthc Mater. 2025;14(1):e2401927. doi: 10.1002/adhm.202401927
- Fan H, Demirci U, Chen P. Emerging organoid models: leaping forward in cancer research. J Hematol Oncol. 2019;12(1):142. doi: 10.1186/s13045-019-0832-4
- Mohamed E-T, Syed Arman R, Rasha B, et al. Unraveling the tumor microenvironment: insights into cancer metastasis and therapeutic strategies. Cancer Lett. 2024;591:216894. doi: 10.1016/j.canlet.2024.216894
- Wu X, Jin Z, Li B, et al. Deciphering of intra-tumoural heterogeneity and the interplay between metastasis-associated meta-program and myofibroblasts in gastric cancer. Clin Transl Med. 2025;15(5):e70319. doi: 10.1002/ctm2.70319
- Julia AL, Lance LM, Rakesh KJ. Compressive stresses in cancer: characterization and implications for tumour progression and treatment. Nat Rev Cancer. 2024;24(11):768-791. doi: 10.1038/s41568-024-00745-z
- Francisco B, Joana C, Maria M, João JS, Carla V. 3D bioprinting models for glioblastoma: from scaffold design to therapeutic application. Adv Mater. 2025;37(18):e2501994. doi: 10.1002/adma.202501994
- Yan L, Haijun C, Haitao C. Precision spatial control of tumor‐stroma interactions in cancer models via 3D bioprinting for advanced research and therapy. Adv Funct Mater. 2025; 2503391. doi: 10.1002/adfm.202503391
- Rong J, Xia L, Qian Z, et al. Anti-tumor immune potentiation targets-engineered nanobiotechnologies: design principles and applications. Prog Mater Sci. 2024;142:101230. doi: 10.1016/j.pmatsci.2023.101230
- Pengcheng Z, Xuanlong D, Weilu J, Kun F, Yewei Z. Engineered extracellular vesicles for targeted reprogramming of cancer-associated fibroblasts to potentiate therapy of pancreatic cancer. Signal Transduct Target Ther. 2024;9(1):1. doi: 10.1038/s41392-024-01872-7
- Hermida MA, Kumar JD, Schwarz D, et al. Three dimensional in vitro models of cancer: bioprinting multilineage glioblastoma models. Adv Biol Regul. 2020;75:100658. doi: 10.1016/j.jbior.2019.100658
- Sun Q, Tan SH, Chen Q, et al. Microfluidic formation of coculture tumor spheroids with stromal cells as a novel 3D tumor model for drug testing. ACS Biomater Sci Eng. 2018;4(12):4425-4433. doi: 10.1021/acsbiomaterials.8b00904
- Godier C, Baka Z, Lamy L, et al. A 3D bio-printed-based model for pancreatic ductal adenocarcinoma. Diseases. 2024;12(9):206. doi: 10.3390/diseases12090206
- Meng F, Meyer CM, Joung D, Vallera DA, McAlpine MC, Panoskaltsis-Mortari A. 3D bioprinted in vitro metastatic models via reconstruction of tumor microenvironments. Adv Mater (Deerfield Beach, Fla). 2019;31(10):e1806899. doi: 10.1002/adma.201806899
- Drost J, Clevers H. Organoids in cancer research. Nat Rev Cancer. 2018;18(7):407-418. doi: 10.1038/s41568-018-0007-6
- Li Y, Liu J, Xu S, Wang J. 3D bioprinting: an important tool for tumor microenvironment research. Int J Nanomed. 2023;18:8039-8057. doi: 10.2147/ijn.S435845
- Heinrich MA, Bansal R, Lammers T, Zhang YS, Michel Schiffelers R, Prakash J. 3D-bioprinted mini-brain: a glioblastoma model to study cellular interactions and therapeutics. Adv Mater (Deerfield Beach, Fla). 2019;31(14):e1806590. doi: 10.1002/adma.201806590
- Zhou X, Zhu W, Nowicki M, et al. 3D bioprinting a cell-laden bone matrix for breast cancer metastasis study. ACS Appl Mater Interfaces. 2016;8(44):30017-30026. doi: 10.1021/acsami.6b10673
- Hughes AM, Kolb AD, Shupp AB, Shine KM, Bussard KM. Printing the pathway forward in bone metastatic cancer research: applications of 3D engineered models and bioprinted scaffolds to recapitulate the bone-tumor niche. Cancers (Basel). 2021;13(3):507. doi: 10.3390/cancers13030507
- Mazzocchi A, Soker S, Skardal A. 3D bioprinting for high-throughput screening: drug screening, disease modeling, and precision medicine applications. Appl Phys Rev. 2019;6(1):011302. doi: 10.1063/1.5056188
- Kim J, Jang J, Cho D-W. Recapitulating the cancer microenvironment using bioprinting technology for precision medicine. Micromachines. 2021;12(9):1122. doi: 10.3390/mi12091122
- Langer EM, Allen-Petersen BL, King SM, et al. Modeling tumor phenotypes in vitro with three-dimensional bioprinting. Cell Rep. 2019;26(3):608-623.e6. doi: 10.1016/j.celrep.2018.12.090
- Calandrini C, Drost J. Normal and tumor-derived organoids as a drug screening platform for tumor-specific drug vulnerabilities. STAR Protoc. 2022;3(1):101079. doi: 10.1016/j.xpro.2021.101079
- Wu P, Asada H, Hakamada M, Mabuchi M. Bioengineering of high cell density tissues with hierarchical vascular networks for ex vivo whole organs. Adv Mater (Deerfield Beach, Fla). 2023;35(9):e2209149. doi: 10.1002/adma.202209149
- Bjerring JS, Khodour Y, Peterson EA, Sachs PC, Bruno RD. Intercellular mitochondrial transfer contributes to microenvironmental redirection of cancer cell fate. FEBS J. 2025;292(9):2306-2322. doi: 10.1111/febs.70002
- Khan AO, Rodriguez-Romera A, Reyat JS, et al. Human bone marrow organoids for disease modeling, discovery, and validation of therapeutic targets in hematologic malignancies. Article. Cancer Discov. 2023;13(2):364-385. doi: 10.1158/2159-8290.Cd-22-0199
- Chen H, Wu Z, Gong Z, et al. Acoustic bioprinting of patient-derived organoids for predicting cancer therapy responses. Adv Healthc Mater. 2022;11(13):2102784. doi: 10.1002/adhm.202102784
- Choi Y-m, Lee H, Ann M, Song M, Rheey J, Jang J. 3D bioprinted vascularized lung cancer organoid models with underlying disease capable of more precise drug evaluation. Biofabrication. 2023;15(3):034104. doi: 10.1088/1758-5090/acd95f
- Jungeun K, Hoe Suk K, Ga Yeon K, et al. Abstract P5-02- 02: development of automated 3d high-throughput drug screening platform for patient-derived breast cancer organoids. Cancer Res. 2022;82:2. doi: 10.1158/1538-7445.sabcs21-p5-02-02
- Hou S, Tiriac H, Sridharan BP, et al. Advanced development of primary pancreatic organoid tumor models for high-throughput phenotypic drug screening. SLAS Discov. 2018;23(6):574-584. doi: 10.1177/2472555218766842
- Arutyunyan I, Jumaniyazova E, Makarov A, Fatkhudinov T. In vitro models of head and neck cancer: from primitive to most advanced. J Pers Med. 2023;13(11):1575. doi: 10.3390/jpm13111575
- Azhakesan A, Kern J, Mishra A, et al. 3D bioprinted head and neck squamous cell carcinoma (HNSCC) model using tunicate derived nanocellulose (NC) bioink. Adv Healthc Mater. 2025;14(7):e2403114. doi: 10.1002/adhm.202403114
- Baka Z, Godier C, Lamy L, et al. A coculture based, 3D bioprinted ovarian tumor model combining cancer cells and cancer associated fibroblasts. Macromol Biosci. 2023;23(3):e2200434. doi: 10.1002/mabi.202200434
- Tebon PJ, Wang B, Markowitz AL, et al. Drug screening at single-organoid resolution via bioprinting and interferometry. Nat Commun. 2023;14(1):3168. doi: 10.1038/s41467-023-38832-8
- Nhan P, Jenny JH, Bobby T, et al. A simple high-throughput approach identifies actionable drug sensitivities in patient-derived tumor organoids. Commun Biol. 2019;2(1):1. doi: 10.1038/s42003-019-0305-x
- Krendl FJ, Primavesi F, Oberhuber R, et al. The importance of preclinical models for cholangiocarcinoma drug discovery. Expert Opin Drug Discov. 2025;20(2):205-216. doi: 10.1080/17460441.2025.2457637
- Joshi P, Nascimento HSD, Kang SY, et al. Dynamic culture of bioprinted liver tumor spheroids in a pillar/perfusion plate for predictive screening of anticancer drugs. Biotechnol Bioeng. 2025;122(4):995-1009. doi: 10.1002/bit.28924
- Kalla J, Pfneissl J, Mair T, Tran L, Egger G. A systematic review on the culture methods and applications of 3D tumoroids for cancer research and personalized medicine. Cell Oncol. 2025;48(1):1-26. doi: 10.1007/s13402-024-00960-8
- Maloney E, Clark C, Sivakumar H, et al. Immersion bioprinting of tumor organoids in multi-well plates for increasing chemotherapy screening throughput. Micromachines. 2020;11(2):208. doi: 10.3390/mi11020208
- Gong Z, Mao Y, Huang L, et al. Acoustic printing of patient-derived organoids that preserve tumor microenvironment for personalized drug screening. Adv Mater Technol. 2023;8(11):2201942. doi: 10.1002/admt.202201942
- Nieto D, Jiménez G, Moroni L, López-Ruiz E, Gálvez-Martín P, Marchal JA. Biofabrication approaches and regulatory framework of metastatic tumor-on-a-chip models for precision oncology. Med Res Rev. 2022;42(5):1978-2001. doi: 10.1002/med.21914
- Wang F, Song P, Wang J, et al. Organoid bioinks: construction and application. Biofabrication. 2024;16(3):3467c. doi: 10.1088/1758-5090/ad467c
- Wu Z, Liu R, Shao N, Zhao Y. Developing 3D bioprinting for organs-on-chips. Lab Chip. 2025;25(5):1081-1096. doi: 10.1039/d4lc00769g
- O’Connor CE, Zhang F, Neufeld A, et al. Bioprinted platform for parallelized screening of engineered microtissues in vivo. Cell Stem Cell. 2025;1;32(5):838-853.e6. doi: 10.1016/j.stem.2025.03.002
- Capeling MM, Czerwinski M, Huang S, et al. Nonadhesive alginate hydrogels support growth of pluripotent stem cell-derived intestinal organoids. Stem Cell Rep. 2019;12(2):381-394. doi: 10.1016/j.stemcr.2018.12.001
- Baptista LS, Porrini C, Kronemberger GS, Kelly DJ, Perrault CM. 3D organ-on-a-chip: the convergence of microphysiological systems and organoids. Front Cell Dev Biol. 2022;10:1043117. doi: 10.3389/fcell.2022.1043117
- Bengtsson A, Andersson R, Rahm J, Ganganna K, Andersson B, Ansari D. Organoid technology for personalized pancreatic cancer therapy. Cell Oncol (Dordr). 2021;44(2):251-260. doi: 10.1007/s13402-021-00585-1
- Mahdavi R, Hashemi-Najafabadi S, Ghiass MA, et al. Design, fabrication, and characterization of a user-friendly microfluidic device for studying liver zonation-on-chip (ZoC). Biomed Microdevices. 2025;27(1):8. doi: 10.1007/s10544-025-00738-1
- Myszczyszyn A, Muench A, Lehmann V, et al. A hollow fiber membrane-based liver organoid-on-a-chip model for examining drug metabolism and transport. Biofabrication. 2025;17(2):206. doi: 10.1088/1758-5090/adc3ce
- Zheng F, Xiao Y, Liu H, Fan Y, Dao M. Patient-specific organoid and organ-on-a-chip: 3D cell-culture meets 3D printing and numerical simulation. Adv Biol (Weinh). 2021;5(6):e2000024. doi: 10.1002/adbi.202000024
- Park B, Park J, Han S, et al. Advances in organoid-on-a-chip for recapitulation of human physiological events. Mater Today. 2025;84:75-94. doi: 10.1016/j.mattod.2025.02.002
- Tonon F, Giobbe GG, Zambon A, et al. In vitro metabolic zonation through oxygen gradient on a chip. Sci Rep. 2019;9(1):13557. doi: 10.1038/s41598-019-49412-6
- McCarty WJ, Usta OB, Yarmush ML. A microfabricated platform for generating physiologically-relevant hepatocyte zonation. Sci Rep. 2016;6(1):26868. doi: 10.1038/srep26868
- Mitani S, Takayama K, Nagamoto Y, et al. Human ESC/ iPSC-derived hepatocyte-like cells achieve zone-specific hepatic properties by modulation of WNT signaling. Mol Ther. 2017;25(6):1420-1433. doi: 10.1016/j.ymthe.2017.04.006
- Wang Q, Liu J, Yin W, et al. Microscale tissue engineering of liver lobule models: advancements and applications. Front Bioeng Biotechnol. 2023;11:1303053. doi: 10.3389/fbioe.2023.1303053
- Saw TB, Doostmohammadi A, Nier V, et al. Topological defects in epithelia govern cell death and extrusion. Nature. 2017;544(7649):212-216. doi: 10.1038/nature21718
- Gupta K, Ng IC, Balachander GM, et al. Bile canaliculi contract autonomously by releasing calcium into hepatocytes via mechanosensitive calcium channel. Biomaterials. 2020;259:120283. doi: 10.1016/j.biomaterials.2020.120283
- Warmflash A, Sorre B, Etoc F, Siggia ED, Brivanlou AH. A method to recapitulate early embryonic spatial patterning in human embryonic stem cells. Nat Methods. 2014;11(8):847-54. doi: 10.1038/nmeth.3016
- Kang R, Park S, Shin S, Bak G, Park JC. Electrophysiological insights with brain organoid models: a brief review. BMB Rep. 2024;57(7):311-317. doi: 10.5483/BMBRep.2024-0077
- Zhou J, Vijayavenkataraman S. 3D-printable conductive materials for tissue engineering and biomedical applications. Bioprinting. 2021;24:e00166. doi: 10.1016/j.bprint.2021.e00166
- Liu P. 3D da yin wei liu kong xin pian xi bao fen xi ping tai de gou jian ji qi ying yong [Construction and Application of a 3D-Printed Microfluidic Chip-Based Cell Analysis Platform] [dissertation]. Shandong Normal University; 2023. h t t p s : / / d . w a n f a n g d a t a . c o m . c n / t h e s i s / vChhUaGVzaXNOZXdTMjAyNDA5MjAxNTE3MjUS CUQwMzAyNDMyOBoIe XRhZ mhk ZXM%3D.
- Mai S, Inkielewicz-Stepniak I. Graphene oxide nanoparticles and organoids: a prospective advanced model for pancreatic cancer research. Int J Mol Sci. 2024;25(2):1066. doi: 10.3390/ijms25021066
- Salmon I, Grebenyuk S, Abdel Fattah AR, et al. Engineering neurovascular organoids with 3D printed microfluidic chips. Lab Chip. 2022;22(8):1615-1629. doi: 10.1039/d1lc00535a
- Park YG, Kim S, Min S, et al. Soft 3D bioelectrodes for intraorganoid signal monitoring in cardiac models. Nano Lett. 2025;25(16):6481-6490. doi: 10.1021/acs.nanolett.5c00069
- Lee S, Chung WG, Jeong H, et al. Electrophysiological analysis of retinal organoid development using 3D microelectrodes of liquid metals. Adv Mater (Deerfield Beach, Fla). 2024;36(35):e2404428. doi: 10.1002/adma.202404428
- Dong K, Liu WC, Su Y, et al. Scalable electrophysiology of millimeter-scale animals with electrode devices. BME Front. 2023;4:0034. doi: 10.34133/bmef.0034
- Acha C, George D, Diaz LC, et al. Neuromodulation in neural organoids with shell MEAs. bioRxiv. 2025. doi: 10.1101/2025.02.18.637712
- Saleh MS, Ritchie SM, Nicholas MA, et al. CMU array: a 3D nanoprinted, fully customizable high-density microelectrode array platform. Sci Adv. 2022;8(40):eabj4853. doi: 10.1126/sciadv.abj4853
- Patel D, Shetty S, Acha C, et al. Microinstrumentation for brain organoids. Adv Healthc Mater. 2024;13(21): e2302456. doi: 10.1002/adhm.202302456
- Li TL, Liu Y, Forro C, et al. Stretchable mesh microelectronics for the biointegration and stimulation of human neural organoids. Biomaterials. 2022;290:121825. doi: 10.1016/j.biomaterials.2022.121825
- Mao M, Han K, Gao J, et al. Engineering highly aligned and densely populated cardiac muscle bundles via fibrin remodeling in 3D-printed anisotropic microfibrous lattices. Adv Mater (Deerfield Beach, Fla). 2025;37(9):e2419380. doi: 10.1002/adma.202419380
- Zilinskaite N, Shukla RP, Baradoke A. Use of 3D printing techniques to fabricate implantable microelectrodes for electrochemical detection of biomarkers in the early diagnosis of cardiovascular and neurodegenerative diseases. ACS Meas Sci Au. 2023;3(5):315-336. doi: 10.1021/acsmeasuresciau.3c00028
- Kalmykov A, Huang C, Bliley J, et al. Organ-on-e-chip: three-dimensional self-rolled biosensor array for electrical interrogations of human electrogenic spheroids. Sci Adv. 2019;5(8):eaax0729. doi: 10.1126/sciadv.aax0729
- Spedicati M, Tivano F, Zoso A, et al. 3D bioartificial stretchable scaffolds mimicking the mechanical hallmarks of human cardiac fibrotic tissue. Int J Bioprint. 2024;10(3):2247. doi: 10.36922/ijb.2247
- Vashistha R, Kumar P, Dangi AK, Sharma N, Chhabra D, Shukla P. Quest for cardiovascular interventions: precise modeling and 3D printing of heart valves. J Biol Eng. 2019;13:12. doi: 10.1186/s13036-018-0132-5
- Chen A, Su J, Li Y, et al. 3D/4D printed bio-piezoelectric smart scaffolds for next-generation bone tissue engineering. Int J Extreme Manuf. 2023;5(3):8. doi: 10.1088/2631-7990/acd88f
- Simonneau C, Duschmalé M, Gavrilov A, et al. Investigating receptor-mediated antibody transcytosis using blood-brain barrier organoid arrays. Fluids Barriers CNS. 2021; 18(1):43. doi: 10.1186/s12987-021-00276-x
- Shen C, Zhang ZJ, Li XX, et al. Intersection of nanomaterials and organoids technology in biomedicine. Front Immunol. 2023;14:1172262. doi: 10.3389/fimmu.2023.1172262
- Paone LS, Benmassaoud MM, Curran A, Vega SL, Galie PA. A 3D-printed blood-brain barrier model with tunable topology and cell-matrix interactions. Biofabrication. 2023;16(1):260. doi: 10.1088/1758-5090/ad0260
- Marino A, Tricinci O, Battaglini M, et al. A 3D real-scale, biomimetic, and biohybrid model of the blood-brain barrier fabricated through two-photon lithography. Small. 2018;14(6):2959. doi: 10.1002/smll.201702959
- Carton F, Malatesta M. In vitro models of biological barriers for nanomedical research. Int J Mol Sci. 2022;23(16):8910. doi: 10.3390/ijms23168910
- Sharma A, Fernandes DC, Reis RL, et al. Cutting-edge advances in modeling the blood-brain barrier and tools for its reversible permeabilization for enhanced drug delivery into the brain. Cell Biosci. 2023;13(1):137. doi: 10.1186/s13578-023-01079-3
- Reina-Mahecha A, Beers MJ, van der Veen HC, Zuhorn IS, van Kooten TG, Sharma PK. A review of the role of bioreactors for iPSCs-based tissue-engineered articular cartilage. Tissue Eng Regen Med. 2023;20(7):1041-1052. doi: 10.1007/s13770-023-00573-6
- Xiu Z, Yang Q, Xie F, Han F, He W, Liao W. Revolutionizing digestive system tumor organoids research: exploring the potential of tumor organoids. J Tissue Eng. 2024;15:20417314241255470. doi: 10.1177/20417314241255470
- Labour MN, Le Guilcher C, Aid-Launais R, et al. Development of 3D hepatic constructs within polysaccharide-based scaffolds with tunable properties. Int J Mol Sci. 2020;21(10):3644. doi: 10.3390/ijms21103644
- Shin J, Kang R, Hyun K, et al. Machine learning-enhanced optimization for high-throughput precision in cellular droplet bioprinting. Adv Sci (Weinh). 2025:12(20):e2412831. doi: 10.1002/advs.202412831
- Hwang HH, You S, Ma X, et al. High throughput direct 3D bioprinting in multiwell plates. Biofabrication. 2021;13(2):2200434. doi: 10.1088/1758-5090/ab89ca
- Hu W, Cao M, Liao L, et al. An automated digital microfluidic system based on inkjet printing. Micromachines (Basel). 2024;15(11):2247. doi: 10.3390/mi15111285
- Bai L, Wu Y, Li G, Zhang W, Zhang H, Su J. AI-enabled organoids: Construction, analysis, and application. Bioact Mater. 2024;31:525-548. doi: 10.1016/j.bioactmat.2023.09.005
- Ear PH, Marinoni I, Dayton T, et al. NET models meeting 2024 white paper: the current state of neuroendocrine tumour research models and our future aspirations. Endocr Oncol. 2024;4(1):e240055. doi: 10.1530/eo-24-0055
- Ma W, Lu H, Xiao Y, Wu C. Advancing organoid development with 3D bioprinting. OR. 2025;1(1):40004. doi: 10.36922/or025040004

