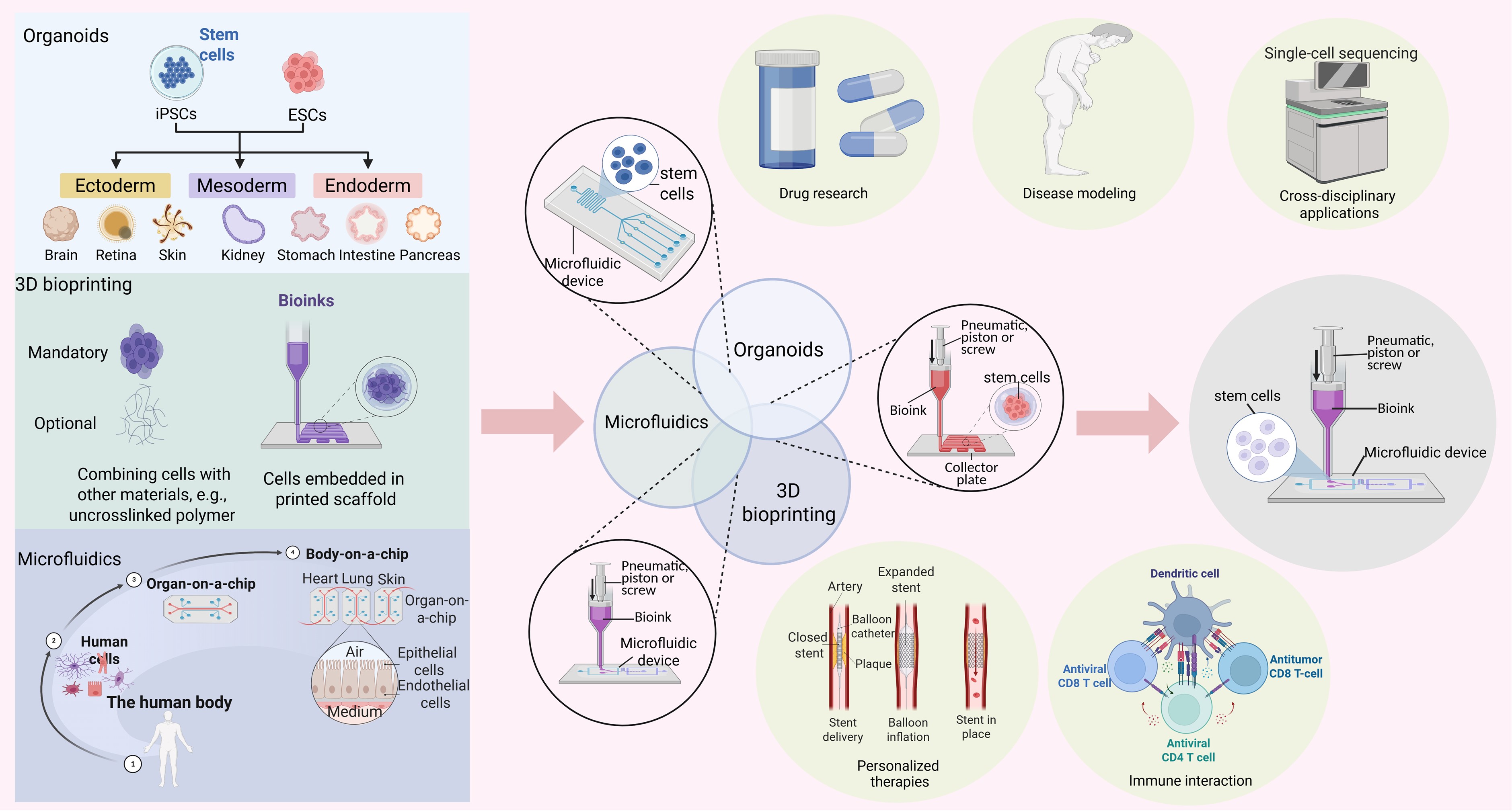
Integrated applications of microfluidics, organoids, and 3D bioprinting in in vitro 3D biomimetic models

Biomedical research has long faced challenges in accurately replicating human organ microenvironments and overcoming interspecies biological differences, thereby limiting the in-depth understanding of physiopathological mechanisms and hindering the development of cutting-edge therapeutic approaches. Recently, novel technologies such as organoids, microfluidics, and three-dimensional (3D) bioprinting offer promising solutions, fostering innovation, and accelerating progress in biomedical science. However, none of these technologies alone can serve as a fully representative preclinical model, underscoring the need for integrated approaches. This review provides a comprehensive overview of various strategies combining microfluidics, organoids, and 3D bioprinting to develop more physiologically relevant preclinical models. After briefly introducing each technology, we examine the advantages of their pairwise integrations and discuss their prospects for drug research, disease modeling, and beyond. In addition, we explore the potential of combining all three technologies, including the emerging concept of 4D culture systems, which incorporate the temporal dimension to better mimic dynamic biological processes. We anticipate that these integrated models will propel significant advances in biomedical research and contribute to the transformation of future healthcare.
1. Introduction
Advances in developmental biology, disease mechanism research, and drug discovery continue to be constrained by the limitations of current experimental models. To advance the development of physiologically relevant in vitro systems for biomedical applications, there is a pressing need for engineered models that faithfully replicate critical histological characteristics—including tissuespecific architecture, cellular heterogeneity, and functional microdomains—that mirror the structural organization and core biological activities of native human tissues. The structural and functional development of organisms, along with the dynamics of organ homeostasis, are orchestrated by physical and spatial cellular interactions. These interactions simultaneously modulate the epigenetic landscape and gene expression and are dynamically shaped by environmental cues, establishing a continuous feedback loop that regulates organ function.1 Organogenesis is governed not only by stimuli from the immediate local microenvironment but also by influences from other tissues and systemic factors, such as fluid flow and mechanical forces.2,3 Therefore, to develop clinically relevant model systems, it is essential to replicate the microenvironment of human organs or tissues using advanced technologies, such as three-dimensional (3D) cultures, extracellular matrix (ECM) supports, and microfluidic devices. These approaches aim to reconstruct the complex in vivo microenvironments that cells inhabit.4
Even though creating such advanced experimental models remains technically challenging, research efforts continue to surge in addressing these barriers. Animal models are the most widely used approach; however, due to interspecies differences, they often fail to accurately simulate and predict physiopathological processes and responses to interventions as they occur in humans. With advancements in cell biology and tissue engineering, cell cultures were initially performed on two-dimensional (2D) substrates such as Petri dishes and porous plates, where cells adhere and grow along the flat surfaces. However, traditional 2D culture systems cannot recapitulate the complex in vivo environment of human tissues, resulting in altered cell morphology and function, and limiting the development of tissue-specific architecture.5 Despite these limitations, the monolayer cell culture method remains valuable in the early stages of drug testing and compound screening due to its cost-effectiveness and ease of use.6 To address the limitations of both 2D models and speciesspecific differences in animal models, 3D cellular models derived from human cells have emerged. Among these, organoids have gained attention for their ability to selforganize into complex structures and simulate functional tissue states.7 Organoids more closely resemble in vivo organs in terms of gene transcription, protein expression, functional metabolism, and microstructure. Nonetheless, they are limited by issues such as incomplete maturation and functionality, low accessibility, high heterogeneity, and inconsistent model readouts.8–11 The advent of microfluidic technologies has further revolutionized the ability to recapitulate in vivo-like microenvironments by enabling precise spatiotemporal control of fluid dynamics. This facilitates biomimetic regulation of critical parameters such as shear stress, nutrient gradients, and intercellular communication.12 Microfluidics not only enhances nutrients and metabolic waste transport but also supports the integration of external stimuli (e.g., mechanical forces) and in situ monitoring of key parameters (e.g., pH), thereby contributing to the development of more robust human models. However, challenges remain, such as limited spatial control precision, low scalability, and reliance on manual processing. In addition, the integration of novel 3D bioprinting concepts into model fabrication enables more accurate construction of diverse cellular microenvironments. It provides physical boundaries to guide morphogenesis and facilitates signaling regulation across temporal and spatial dimensions. However, these pre-determined model structures may reduce intercellular communication and hinder the efficiency of material transport.13Table 1 briefly summarizes key comparisons among these technologies.
Table 1. Summary of microfluidics, organoids, and 3D bioprinting
Note: Figures were created in BioRender. Liang, L. (2025) https://BioRender.com/6zt3gft
Although many novel technologies have made significant progress in addressing the challenges of in vitro model construction, several critical issues persist. First, current models still fall short in simulating the biological complexity of real in vivo environments, especially in terms of cellular functions, cell–cell interactions, and tissue– tissue integration.14 Second, technological limitations hinder scalability, throughput, and cost-effectiveness, restricting widespread application. In addition, improving model maturity, functionality, and stability while accurately replicating the human tissue microenvironment remains a top priority.15 Therefore, integrating organoid culture, microfluidic systems, and 3D bioprinting—each offering distinct advantages—holds great promise for advancing the field of bioengineering. This paper first describes the individual technologies: organoids, microfluidics, and 3D bioprinting. It then focuses on the characteristics and applications of combining these technologies in areas such as drug research, disease modeling, immunological research, clinical therapy, and interdisciplinary integration. Finally, the paper discusses existing studies involving all three technologies and proposes an innovative 4D cultivation model that incorporates the temporal dimension.
2. Brief introduction to microfluidics, organoids, and 3D bioprinting
2.1. Microfluidics
Microfluidics is a system for studying and manipulating fluid flow at the sub-millimeter scale. It enables precise control of key factors such as the physico-mechanical properties and chemical composition of in vitro models. This technology primarily utilizes microfluidic chips to support cell cultures that more closely resemble in vivo conditions.16 Beyond being a mere tool, microfluidics is a multidisciplinary field that integrates principles from physics, engineering, chemistry, and biology to improve the biological relevance of experimental models and the accuracy of experimental results.17,18
The core component of microfluidic technology is the microfluidic chip. The first such chip in human history was the organ-on-a-chip reported by Huh et al., designed to mimic the natural anatomy of the target organ by incorporating key functional components essential for replicating physiological functions. Structurally, the chip successfully recreated a critical anatomical feature— the alveolar–capillary interface–by co-culturing human alveolar epithelial cells with microvascular endothelial cells. Functionally, the system exhibited barrier integrity and permeability, surfactant production, and the ability to mimic physiological respiratory movements through a computer-controlled vacuum system that induced cyclic stretching of the tissue–tissue interface.19
With the exponential growth of multidisciplinary research, microfluidics has undergone significant expansion and technological advancement. Improvements in precision and stability of fluid flow control have not only effectively improved its ability to transport a wide range of components (e.g., nutrients, metabolic wastes, oxygen, and immunomodulatory factors), but also permitted it to exert dynamic mechanical forces (e.g., vascular shear stress, intestinal peristalsis, skin tensions) within the cellular microenvironment. These capabilities make microfluidic systems an excellent platform for designing novel bioassays that allow both the precise manipulation of experimental parameters and seamless integration with other techniques.20
Models based on microfluidic devices have been widely developed across various biological systems. For example, a lung cancer brain metastasis model consists of two biomimetic units—an upstream “lung” and a downstream “brain”—connected by a functional region mimicking the blood–brain barrier (BBB). By monitoring, in real time, the progression from primary tumor growth to BBB penetration and eventual invasion of the brain parenchyma, researchers identified aldo-keto reductase family 1 B10 (AKR1B10) as a potential serum biomarker for patients with brain metastases of lung cancer. The study also suggested that AKR1B10 may play a role in mediating cancer cell extravasation.21 Similarly, microfluidic systems have been applied in culturing ovarian cancer spheroids to assess the feasibility of multi-class drug sensitivity assays. The stable transport of substances and controlled modulation of the microenvironment ensured the stabilization of the cell lines, further demonstrating the technique’s ability to regulate spatiotemporal variables and generate precise flow patterns at the microscopic scale.22 Furthermore, robust and streamlined microfluidic chips enable the incorporation of dynamic mechanical forces— such as fluid flow and shear stresses—into tumor spheroid cultures, providing a valuable paradigm for constructing physiologically relevant in vitro models.23,24
Undeniably, while microfluidics enables precise control of fluid dynamics at the microscopic scale, the models produced can only replicate key structural and functional features of target organs or tissues and remain biologically distinct from native organs.25 Additionally, microfluidic techniques alone lack spatial refinement for complex model sculpting and have yet to meet established benchmarks for advanced biomedical applications.26 In addition, the initial setup and cell seeding processes typically require manual operation, underscoring the urgent need for integration with complementary technologies to automate procedures, streamline data acquisition, and improve system accessibility.27
2.2. Organoids
Organoids are 3D structures derived from pluripotent stem cells (PSCs) or tissue-derived adult stem cells (AdSCs). They undergo growth and differentiation processes that mimic the tissue of origin and ultimately consist of organspecific cell types. These cells self-organize through lineage commitment, cell sorting, and spatial restriction.28
In general, organoids derived from PSCs are engineered based on the principle of sequential stem cell differentiation. Successful generation requires the timely and sequential addition of relevant signaling factors to guide cell fate decisions and support self-organization. Growth factors are administered in a sequence that mirrors embryonic development to establish correct regional identities and enable the identification of organ-specific lineages using unique biomarkers or functional assays. In contrast, AdSC-derived organoids are relatively simpler to generate. The process involves isolating tissue-specific stem cell populations, embedding them in an ECM, and providing defined combinations of growth factors to support their proliferation and differentiation.29,30 While PSC-derived organoids can give rise to complex structures such as vasculature and immune components by incorporating diverse cell types, they often require prolonged development times and present challenges in maintaining stable propagation over multiple passages. These factors limit their immediate application in disease modeling.
In contrast, AdSCs-derived organoids, sourced from patient tissues, retain the genetic background of the donor and demonstrate significant potential for personalized medicine. Notably, PSCs can also be derived autologously.31 Both types of organoids can effectively mimic the pathophysiological characteristics of patient tissues, offering powerful platforms for developing in vitro personalized disease models. Nevertheless, AdSC-derived organoids are generally restricted in their differentiation capabilities and typically recapitulate only specific parts of a given organ. For example, adult intestinal stem cells can differentiate exclusively into intestinal epithelial cells. Furthermore, inter-individual variability and high levels of heterogeneity underlie the instability of these organoids during long-term culture.29 Organoid technology is firmly rooted in classical developmental biology, drawing from extensive research in cell dissociation and recombination. By imposing rational spatial constraints, this innovative approach effectively guides the fate of progenitor cells, ultimately shaping the morphogenesis and organization of organoids.32,33 Building on this foundation, organoids formed through the spatial organization of multiple cell types—referred to as assembloids—allow for a more profound exploration of tissue functionality.34
Organoids often closely resemble human tissues, exhibiting microscale organization and physiological functions that are more similar to those of natural organs. These attributes were demonstrated by Sheridan et al.,35 who created embryonic trophoblast organoids. The cultured tissues not only successfully differentiated into syncytial and extrachorionic trophoblast structures but also functionally secreted placenta-specific peptides and hormones. For this very reason, organoids have been widely used in various areas of biological research. Patient-derived organoids have been demonstrated as functional models for predicting the pharmacological effects of anticancer agents. Culture systems established from specimens representing different stages of disease progression and histological grades in bladder cancer effectively maintain the histopathological features and molecular heterogeneity of the original tumors, including their multiclonal genetic characteristics. These culture systems consistently exhibit key genomic variation patterns that mirror those of parental tumors, with dynamic changes corresponding to tumor evolutionary trajectories observed through longitudinal analyses.36 In addition, the differentiation of human embryonic stem cells into thymic epithelial progenitor cells through precise regulation of developmental signaling molecules, including fibroblast growth factor 8, retinoic acid, sonic hedgehog, noggin, and bone morphogenetic protein 4, has further demonstrated the great potential of organoids for biomimetic modeling. When transplanted into swine thymus tissue with a supportive microenvironment, these progenitors not only successfully integrated into the grafts but also significantly enhanced thymocyte production efficiency and increased the reconstitution of CD4+ naive T cells in peripheral circulation.37
However, organoids also face notable limitations.38–41 They are often cultured for relatively short durations, limiting robust differentiation into the full cellular diversity available in the target organ and resulting in poor maturation and functionality. Their short life cycle is partly attributed to limited accessibility to nutrients and inefficient removal of metabolic waste.42 As organoids increase in size, nutrient and waste transport primarily occurs through diffusion, which becomes inefficient, ultimately compromising long-term viability. Moreover, the self-organizing nature of organoid formation and the stochasticity of cell fate decisions contribute to morphological and functional heterogeneity within the cultures.43,44
Material limitations and traditional monitoring methods also limit the interpretability of organoid model data. For example, commonly used substrates such as Matrigel lack sufficient biocompatibility and standardization, making accurate replication of the in vivo environment challenging.41,45 Additionally, traditional culture media often fail to deliver oxygen and nutrients uniformly, resulting in cellular inhomogeneity.46,47 Simultaneously, existing monitoring methods struggle to capture the complex dynamics within organoids in real time, and their 3D structure complicates data acquisition and analysis.48,49 Furthermore, internal tissue heterogeneity adds another layer of complexity to data interpretation.50
2.3. 3D bioprinting
Three-dimensional bioprinting is an innovative fabrication technology that utilizes computer-aided design and control systems to pattern and assemble biological and nonbiological materials into defined 2D or 3D organization for constructing complex biological tissue or organ models.51 Key components of this technology include the development of a digital blueprint using specialized software, the selection of an appropriate printing device compatible with the material and tissue characteristics, and the implementation of layer-by-layer deposition techniques.
Bioprinting devices usually consist of printheads, printing platforms, and control systems that facilitate precise printing and lamination processes. Optimization of the printing procedure involves adjusting printing parameters, determining the layering sequence, and designing support structures to ensure fidelity and functional outcomes.15,52 This technology enables the reconstruction of complex tissue structures with high precision, customization, personalization, and throughput, while also supporting automation.53
Layer-by-layer deposition techniques include nozzlebased bioprinting, light-based bioprinting, and hybrid systems that combine both methods. Within these techniques, key printing parameters in the control system serve as critical determinants for ensuring both high spatial precision and biologically compatible outcomes54 (see Table 2 for details). In extrusion bioprinting, stable extrusion of thermosensitive materials is achieved by regulating nozzle temperature, pressure, and material viscosity, thereby minimizing cell damage.55–58 Inkjet printing (drop-on-demand), on the other hand, relies on precise control of nozzle temperature, jet pressure, and ink viscosity to generate uniform droplets while maintaining cell viability.40,59–62 Cellular electro-writing requires accurate adjustment of voltage, nozzle-to-stage distance, and material viscosity to form stable fibers without compromising cell integrity.63 For light-based bioprinting techniques, such as digital light printing and volumetric bioprinting, parameters including light intensity, exposure time, material viscosity, and print speed are critical for controlling material curing and achieving high printing resolution.64–66 Multiphoton lithography attains nanoscale precision through controlled light intensity and scanning speed, while laser-induced forward transfer relies on precise regulation of laser pulse energy and focal distance for efficient material transfer.67,68 Furthermore, precise thermal regulation and the preservation of cell viability within bioinks are critical for successful bioprinting. Proper management of these printing parameters allows effective integration of material properties, cellular requirements, and printing precision, paving the way for innovative opportunities and expansive applications in the fields of tissue engineering and regenerative medicine.69,70
Table 2. Introduction to 3D bioprinting technology
| Name | Mechanism | Size | Advantages | Disadvantages | References |
|---|---|---|---|---|---|
| Nozzle-based bioprinting | |||||
| EB | Mechanically actuated or pneumatically extruded polymers | Filament diameter range 100–500 μm | The high degree of customization, control of microstructure, flexibility, the high degree of automation, lower costs | Increased risk of cell damage, limited resolution, not suitable for high-viscosity bioinks, prone to cell clogging, limited choice of biomaterials | 55–58 |
| DoD | Introducing strand breaks in polymer extrusion jets by means of heating, piezoelectricity, electrostatics, or electrodynamics | Droplet diameter range 5–2000 μm | 40,59–62 | ||
| CEW | Stabilized jets of filaments sprayed from polymers controlled by electric fields | Fiber diameter range 5–50 μm | 63 | ||
| Light-based bioprinting | |||||
| DLP | Layer-by-layer crosslinked resin printing using CAD-projected light voxels | Minimum feature resolution of 20 μm | High resolution, rapid prototyping, suitable for complex structures, biocompatible, precise control | Limited range of printable materials, limited by the focus of the beam and optical system, loss of bioactivity due to light curing, high environmental requirements, toxicity of photoinitiators, equipment limitations, high cost of systems, and long time to fabricate small structures | 64 |
| VBP | The photocrosslinked material is rotated with the aid of a laser-projected CAD file to create a holographic pattern. | 40 μm for positive features and 100 μm for negative features | 66 | ||
| MPL | The laser scans the focal point, activating the resin and polymerizing it layer by layer. | Lateral resolution of 100 nm and axial resolution of 300 nm | 67 | ||
| Combining 3D bioprinting based on light and nozzle principles | |||||
| LIFT | Utilizing a high-power pulsed laser focused on a thin film of ink, the ejected material forms voxels that are transferred to the surface of the receiver substrate. | Resolution of 20 μm | Fine structure and high resolution, fast print speeds, flexibility and versatility, precise control and customization for complex structures | Increased complexity, difficult technology integration, increased cost, print accuracy limitations, material compatibility challenges, and difficult technology debugging | 68 |
Abbreviations: CAD, computer-aided design; CEW, cellular electro-writing; DLP, digital light printing; DoD, droplet-on-demand; EB, extrusion bioprinting; LIFT, laser-induced forward transfer; MPL, multi-photon lithography; VBP, volumetric bioprinting.
Material selection for bioprinting and tissue engineering requires a systematic evaluation of physical, chemical, and mechanical properties to ensure biocompatibility, functionality, and structural integrity— critical factors for successful tissue formation and integration within the body. Biomaterials are generally classified into two broad categories based on their origin as follows: natural and synthetic. Regardless of their source, all materials must be biocompatible, printable, and cytocompatible (see Table 3 for more details). Natural materials such as chitosan and hyaluronic acid are widely utilized due to their exceptional biocompatibility and degradability.55 Chitosan exhibits good biocompatibility, and its mechanical strength and stiffness can be optimized by adjusting the degree of cross-linking.71–74 Its viscosity and rheological properties also vary significantly with molecular weight and concentration.71,74–76 In contrast, hyaluronic acid is valued for its high hydrophilicity and excellent hydration properties; its cross-linked form can form elastic hydrogels, though it exhibits reduced stiffness when uncross-linked.77 Synthetic materials such as poly(ε-caprolactone) and the Pluronic series provide distinct performance advantages.78–82 Poly(ε-caprolactone) is highly biodegradable and easily processed due to its low melting point, remaining solid at body temperature.78,80,81 Its mechanical strength can be further enhanced through cross-linking or compositing. Poly(ε-caprolactone) also exhibits high melt viscosity, favorable rheological properties, and low thixotropy.81 The Pluronic series, composed of nonionic surfactants, is known for its biocompatibility and thermosensitive properties.79 In aqueous environments, these materials form gels or micelles with low mechanical strength but high thixotropy, making them well-suited for temperaturesensitive applications.81 The integration of 3D bioprinting with micro- and nanotechnology enables precise, sitespecific delivery of bioactive compounds at the molecular level. This combination offers innovative solutions, such as incorporating micro- and nanoparticle structures that effectively minimize non-specific interactions with blood components and the mononuclear phagocyte systems, thereby facilitating targeted therapy for localized lesions.83 As 3D printing research advances, a wide range of smart materials has emerged, including shape memory polymers, hydrogels, liquid crystal polymers, shape memory alloys, dielectric elastomers, piezoelectric materials, magnetically active materials, and biologically functional particles or fillers—each offering diverse biomedical applications.84,85 Notably, shape memory polymers and hydrogels have gained prominence due to their superior printability, inherent biocompatibility, and capacity for structural reconfiguration, making them ideal for fabricating complex tissue architectures.85–87 Compared to traditional biological preparation methods, which often struggle to simulate complex biological tissue structures, 3D bioprinting enables precise layer-by-layer control of material placement and tissue architecture, thereby achieving a high degree of biomimicry. By finetuning printing parameters and material properties, tissue and organ models can be customized to reflect individual anatomical and physical characteristics.88 Moreover, automated printing systems and optimized protocols enable the rapid and efficient production of large quantities of biological models.89
Table 3. Introduction to 3D bioprinting materials
| Material | Attributes | Advantages | Disadvantages | Applications | Improvement | References |
|---|---|---|---|---|---|---|
| Natural polymers | ||||||
| Alginate | Water-soluble polysaccharide, derived mainly from brown seaweeds | Easy to print to form 3D structures; compatibility with ionic cross-linking; water absorption; low cost | Poor cell adhesion, limited mechanical properties, unstable degradation rate, low molding precision, and limited application range | Bones, muscles, cartilage, skin, nerves and blood vessels, and functional organs, including the heart, liver, kidneys, and bladder | Addition of other cell-attachable biomaterials or modification with adhesion molecule sequences | 205 |
| Chitosan | Polysaccharide derived from the deacetylation of chitin in crustacean shells and fungi | Relatively inexpensive; good non-toxicity; biodegradable; antimicrobial | Acidic environment unsuitable for cell survival, slow gelation rate, poor mechanical properties for bioprinting | Cartilage regeneration, bone tissue engineering, liver tissue engineering, and vascular tissue engineering, etc. | Adjusting solvent pH to neutral, mixing with other hydrogels, cross-linking with various compounds, and working with other polyelectrolytes | 71–73,206,207 |
| Agarose | Water-soluble polysaccharide from seaweeds | Inert; good thermal sensitivity | Non-adhesion, biodegradability | Forms cell aggregates and/or supports differentiation of pericytes, “sacrificial biomaterial” for scaffold vascularization | Mixed with collagen | 208 |
| HA | Glycosaminoglycan found in the ECM | Stimulates inflammatory response; water-soluble; the resulting solution has a high viscosity; tunable physical and biological properties | Time-consuming, possibly toxic to encapsulated cells, poor mechanical properties, and rapid degradation | Dermal fillers for wound healing, auxiliary materials to regulate the viscosity of solutions of other biomaterials in bioprinting, and suitable materials for cell incorporation | Curing methacrylates with UV light | 77 |
| Collagen | Abundant natural protein in the body | Natural receptor for cell attachment; soluble in slightly acidic aqueous solutions; polymerizes at 37°C and neutral pH in 60 min | Faster degradation, biocompatibility needs to be further improved, higher cost, difficult to control its mechanical properties, poor structural stability | Tissue scaffolds use the widest range of natural materials, including fat, bladder, blood vessels, bone, cartilage, heart, liver, nerves, and skin, among other tissues | Covalent bonding and irradiation cross-linking methods applied with thermal polymerization of collagen solutions; blends of collagen and other synthetic polymers | 209 |
| Gelatin | Derived from partial hydrolysis of collagen | Biocompatible; non-immunogenic, cell affinity; fully biodegradable in vivo | Temperature-sensitive, structurally unstable, and relatively weak mechanical properties | Osteochondral regeneration, wound healing, heart tissue repair, cornea, and blood vessel formation | Chemicals incorporating metal ions, glutaraldehyde, and other printable materials | 210,211 |
| Fibrin | Blood clot-forming protein active in wound healing | Inherent cell adhesion capacity | Low mechanical stability, rapid degradation, and limited viscosity | Creating cell-doped fibrin constructs that can serve as drug delivery systems for wound healing | Use of high concentrations of fibrinogen or thrombin; mixing with other biomaterials with better mechanical stability; addition of protease inhibitors; optimization of printing temperature, calcium concentration, and cell density; premixing of fibrinogen and thrombin solutions | 212-214 |
| dECM | ECM biomaterial obtained via tissue decellularization | Contains a variety Ofbiologically active molecules and proteins that promote cell growth and function. | Weak mechanical properties | Compatible with multiple technologies for tissue engineering, including heart, kidney, and liver | Use of other polymers as frames | 215, 216 |
| PBH | Synthetic peptides selfassembled into hydrogels | High biological activity | Poor printability, poor mechanical stability | Tissue engineering, drug delivery, cell culture, and building biosensors | Modifying elastin-like polypeptides to make them photosensitive; layering dipeptides with oppositely charged terminal residues to promote electrostatic interactions and form stable structures | 217-219 |
| Synthetic polymers | ||||||
| PCL | Biodegradable polyester degraded by hydrolysis | Low melting point (60 oC); hydrophobicity; slow degradation; thermoplastic behavior; considerable mechanical strength; hydrolysis-induced biodegradation | Melting temperature too high to maintain cell viability | Formation of mechanically stable 3D structures with good structural fidelity | Cell inoculation after printing with another hydrogel bioink during the scaffold fabrication and impregnation process | 77 |
| PBP | Composed of PEG and PEO via ethylene oxide polymerization | Water soluble; metabolically inert; biocompatible; high permeability | Limited support for protein binding and cell adhesion | Reduced immunogenicity after implantation, used to encapsulate cells for cell delivery | Modification with peptides with enhanced cell adhesion capacity | 220,221 |
| Pluronic | PEO-PPO-PEO triblock copolymer based on PEO and PPO arrangement | Heatsensitivej amphiphilic properties, surfactant properties | Limited cell adhesion, limited degradation, uncertain Cytocompatibility | Used as a sacrificial bioink for creating molds, channels, containers, or vascular systems for 3D bioprinting, or as a temporary support structure | NA | 222 |
| Composite polymer | ||||||
| HFP | Combination of two or more natural and/or synthetic polymers with synergistic properties | Enhanced mechanical and biological properties and rheological and indentation characteristics | Lower mechanical strength, water solubility, shrinkage, and strict reaction conditions | Complex of type I collagen and ECM protein; complex of alginate and type I collagen | NA | 55 |
| CHIF | Inorganic ceramics combined with natural or synthetic polymers | Superior strength and bioactivity; printable | Inhomogeneity, reduced mechanical properties, interfacial issues, increased cost, and biocompatibility issues | Provides scaffolding support and release of bioactive substances to promote bone regeneration and repair | NA | 223-225 |
Abbreviations: CHIF, composite hydrogels incorporating fillers; dECM, decellularized extracellular matrix; ECM, extracellular matrix; HA, hyaluronic acid; HFP, hydrogel forming polymers; NA, not available; PBH, peptide-based hydrogels; PBP, polyethylene-based polymers; PCL, poly-epsilon-caprolactone; PEG, polyethylene glycol; PEO, polyethylene oxide; PPO, polypropylene oxide.
Three-dimensional bioprinting has revolutionized the biomedical field through its advantages in customization, precision, efficiency, and versatility, laying a robust foundation for individualized medicine and advanced biomedical research. For example, Roth et al. utilized high-throughput automated inkjet bioprinting to generate complex multicellular patterns through the precise deposition of collagen. This printing technique not only ensures the high resolution of cell patterns but also reduces experimental contamination associated with manual handling.90 Hafa et al.91 engineered biologically active full-thickness skin tissue with high printing speed (0.66 mm3/s) and resolution (9 μm). The constructs retained key histological features of the epidermis and dermis and maintained structural integrity and metabolic activity for 41 days, demonstrating the promise of this technology in the field of tissue regeneration and organ transplantation. In another study, a homogeneous in vitro 3D model of colorectal cancer and its liver metastasis was efficiently constructed through 3D bioprinting of patient-derived primary tumor cells and bioinks. Notably, this model not only effectively preserved parental tumor biomarkers and mutation profiles but also exhibited substantial tumor heterogeneity in chemotherapeutic response, showcasing its utility in precision oncology and preclinical testing.92
Overall, 3D bioprinting enables the accurate replication of complex tissue structures while offering high degrees of personalization and customization for individual needs. Its high degree of automation and productivity makes it ideal for the rapid generation of tissue and organ models, accelerating advancements in biomedicine. However, certain limitations remain, including reduced cell–cell interactions, challenges in nutrient and metabolic waste exchange, difficulty in simulating and precisely controlling physicochemical factors, and discrepancies between the printed constructs and native tissue in vivo.93,94
3. Pairwise integration of microfluidics, organoids, and 3D bioprinting
3.1. Microfluidics and organoids
The integration of microfluidic technology into organoid models enables fine-tuned regulation of the microenvironment, which not only facilitates the incorporation of system-level parameters but also enhances vascularization and prolongs model viability. This dual-technology approach significantly enhances data interpretability and supports real-time imaging through synergistic functional enhancement.
3.1.1. Integration of system-level parameters
In vitro organoid cultures, derived from stem cells, recapitulate the cellular composition, microstructure, and vital functions of native tissues, making them highly valuable for clinical applications. However, achieving clinically translatable models requires organoids that more closely mimic physiological conditions. Fortunately, with the introduction of auxiliary technologies such as microfluidics, organoid culture will no longer be limited to the intrinsic self-organization of cells. Instead, it can be externally regulated to a controllable extent by manipulating the microenvironment to adjust system-level parameters (e.g., fluid flow, mechanical force, and tissue space dimensions), thereby enabling the construction of biologically authentic organoid models. For example, under static culture conditions, most models remain avascular and immature. In contrast, renal organoids cultured under flow conditions on a millifluidic chip developed a vascular network with perfusable lumens surrounded by mural cells, exhibited more mature pedunculated cells and tubular compartments, and showed enhanced cell polarity and adult gene expressions (Figure 1A).95 Similarly, for stomach organoids, integrating organ-on-a-chip systems with microfluidic modulation enabled the simulation of fluid flow and peristaltic-like motions within the luminal space. These features recapitulated physiological gastric functions and effectively reduced the risk of bacterial overgrowth, intestinal obstruction, and inflammatory bowel disease associated with impaired peristalsis (Figure 1B).96 Likewise, colon tumor organoids cultured on microfluidic chips successfully mimicked the in vivo mechanical stimulation of intestinal muscles through cyclic pressure-channel contractions, illustrating the potential of engineering approaches that integrate biophysical factors into organoid cultures.97
Figure 1. Microfluidics and organoids—Part I. (A) Kidney organoids cultured on a dynamic microfluidic chip showed more mature podocytes and renal tubular compartments, along with a large network of perfusable luminal vessels. Adapted with permission from ref.95, Copyright © Nature 2019. (B) Gastric organoids placed in a microfluidic device were exposed to rhythmic stretching and contraction, realistically simulating peristaltic-like stomach motions. Adapted with permission from ref.96, Copyright © Royal Society of Chemistry 2018. (C) Vascularized brain organoids were generated by co-cultivating iNSC-derived spheroids with perfusable blood vessels in an injection-molded microfluidic chip. Adapted with permission from ref.101. Copyright © Wiley 2022. Abbreviations: ECM, extracellular matrix; hGO, human gastric organoid; HUVEC, human umbilical vein endothelial cell; iNSC, induced neural stem cell; LF, lung fibroblast; PC, polycarbonate; PDMS, polydimethylsiloxane; PSA, pressure-sensitive adhesive; PSC, pluripotent stem cell.
3.1.2. Vascularization of organoid cultures
Vascularized organoid technology—arising from the integration of microfluidic principles with organoid models—is essential for replicating organ growth and function and for advancing this important field. The core of this technology is self-assembly, which stimulates the formation of microvascular networks through angiogenesis, while microfluidic systems enable dynamic perfusion.98,99 A common technique for generating vascularized organoids involves inducing the self-organization of PSCs to form the desired organoid and initiating vascularization following the application of specific inducing factors. In addition, shear stress introduced via increased fluid flow during organoid self-organization enhances vascularization by triggering mechanotransduction pathways in vascular endothelial cells. This stimulation promotes the secretion of pro-angiogenic factors, which drive endothelial cell migration and proliferation.95 Another strategy involves co-culturing stem cells with endothelial cells under specific conditions. This method facilitates the self-assembly of vascular networks through the intrinsic bioprogramming of both cell types.100 For example, Shin et al.101 utilized injection-molded microfluidic chips to co-culture spheroids derived from induced neural stem cells with human umbilical vein endothelial cells. This setup led to the development of a 3D model resembling a vascularized brain organoid (Figure 1C).
A further method entails the engineered assembly of organoids with microfluidic channels lined with endothelial cells. In this approach, organoids are formed from stem cell differentiation, while vascular networks develop by seeding endothelial cells into porous microfluidic channels. These cells then invade the adjacent ECM via angiogenesis, forming a perfusable vascular network. The geometry (diameter and length), density, branching, perfusion, stability, and permeability of these networks are highly dependent on biophysical (e.g., interstitial and intraluminal flow and matrix stiffness) and biochemical (e.g., vascular endothelial growth factor) cues provided by the surrounding matrix and stromal cells.102
The ultimate goal of vascularized organoid technology is to generate tissue-specific microvascular systems and to connect multiple organoids through dynamic vascular networks, creating integrated systems capable of responding to physiological changes. Currently, successful examples of such techniques have been reported in organoids representing the bone marrow, brain, heart, liver, pancreas, and intestines.103–108
3.1.3. Survival enhancement
Microfluidic devices can also be utilized to prolong the survival time of organoids. This benefit primarily stems from the ability of microfluidic systems to mimic the in vivo microenvironment and provide precise biological and physiological regulation, thereby creating a more favorable growth environment for organoids (Figure 2A).20 Through the use of microchannels and fluid control, microfluidic devices enable precise spatiotemporal regulation and delivery of nutrients, oxygen, and metabolites, which is essential for maintaining organoid cell viability.109,110 In addition, microfluidic systems promote inter-tissue interactions and cellular signaling, facilitating communication and coordination among cells. These interactions contribute to the maintenance of tissue structure and cellular function within the organoid. Consequently, microfluidic platforms effectively reduce cell death in the core regions of the culture, thereby enhancing cell proliferation and differentiation, and ultimately extending the survival time of the organoid (Figure 2B).111
Figure 2. Microfluidics and organoids—Part II. (A) A brain organoid–microfluidic platform with dynamic fluidic perturbation and oxygenation enabled sustained culture for over 50 days. Adapted with permission from ref.20, Copyright © Elsevier 2023. (B) Bladder cancer organoids maintained a long-term culture using a microfluidic device or microchamber. Adapted with permission from ref.111, Copyright © Nature 2017. (C) Visualization of organoid vascularization using a microfluidic device. Adapted with permission from ref.98, Copyright © Nature 2024. Abbreviation: PDMS, polydimethylsiloxane.
In summary, while organoids are capable of recapitulating key generational characteristics of primary tissues, they are limited in their ability to regulate the extracellular physicochemical microenvironment using conventional technologies. Therefore, the development of physiologically and pathologically relevant homeostatic organ models necessitates the incorporation of technologies such as microfluidics to support and enhance organoid growth and maintenance.
3.1.4. Improved data readability and real-time imaging
Organoid culture is a complex and dynamic process, and maximizing the utility of organoid technology requires enhanced data readability and dynamic monitoring capabilities. Given its current limited data readout (e.g., geometry, quantity, oxygen concentration, pH) and monitoring capabilities, there is a critical need for precise and accurate functional readouts and realtime imaging to maximize the benefits of organoid technology. The integration of microfluidic devices into organoid culture platforms enables real-time monitoring and computational imaging of physicochemical factors within the microenvironment, facilitated by embedded sensors.112 A representative example of this concept is the organoid-on-a-chip system. These microchip platforms replicate both the structural and functional aspects of biological systems, allowing for more accurate tracking of key biological and physiological parameters compared to conventional methods.
For example, Schneider et al.113 utilized hydrostatic self-assembly of induced PSC (iPSC)-derived cardiac spheroids to form aligned myofibers and integrated fluidic electrodes with an open-source pulse generator for electrical stimulation. A resin-embedded microfluidic chip with real-time optical oxygen sensing enabled dynamic environmental monitoring. This multimodal regulation and monitoring system provides a novel solution for automated high-density tissue fabrication. Furthermore, image-based analysis is indispensable for assessing the physiopathological parameters of organoids, and its effectiveness is greatly enhanced through integration with microfluidics. For example, Quintard et al.98 developed a microfluidic platform to dynamically image the formation of endothelial networks around mesenchymal and pancreatic islet spheroids, as well as the generation of vascular organoids from PSCs (Figure 2C).
3.2. Microfluidics and 3D bioprinting
The combination of microfluidics and 3D bioprinting enables in vitro models to incorporate fluid flow dynamics and spatial control, allowing cultures to more closely mimic the anatomical structure and functional states of in vivo organs.114,115 The implementation of this integrated technological platform supports the precise calibration of biomimetic system parameters through iterative optimization protocols.116 Additionally, this technology not only improves sterility standards during the culture process but also significantly enhances the consistency of experimental procedures due to the highly automated nature of bioprinting.27,117
3.2.1. Modeling complex organizational structures
Three-dimensional bioprinting integrated with microfluidics enables flexible and precise manipulation of small-scale biological components and fluids, facilitating the simulation of complex microstructures found in natural tissues. In traditional in vitro models, replicating the layered structure of native myocardium poses a substantial challenge in engineering functional cardiac tissues. Zhang et al.118 address this by directly bioprinting a vascular bed within a microfibrous hydrogel scaffold using a composite bio-ink, onto which cardiomyocytes were seeded. This resulted in an aligned myocardial tissue capable of spontaneous and synchronous contraction. The endothelialized myocardium was then embedded into a specially designed microfluidic device, which not only partially restored the in vivo myocardial structure but also enabled toxicity assessment of relevant cardiovascular drugs (Figure 3A).
Figure 3. Microfluidics and 3D bioprinting. (A) 3D bioprinted endothelialized myocardium embedded in a specially designed microfluidic device for cardiovascular toxicity testing. Adapted with permission from ref.118, Copyright © Elsevier 2016. (B) Integration of 3D bioprinting and microfluidics to simulate the blood–brain barrier and adjacent 3D perivascular tumor microenvironment. Adapted with permission from ref.119, Copyright © Wiley 2021. (C) Microfluidic printhead for real-time regulation of printed cell concentration. Adapted with permission from ref.123, Copyright © Wiley 2018. (D) Automated bioprinting of tumor spheroids into chip-based electrochemical oxygen sensor microvias. Adapted with permission from ref.117, Copyright © Royal Society of Chemistry 2022.
Similarly, Silvani et al.119 reconstructed a compartmentalized polymorphic glioblastoma microenvironment consisting of a functional BBB and adjacent 3D perivascular tumor ecotopes by selectively mimicking physiological shear stress and mechanical interactions, including cell–cell and cell–matrix communication. Specifically, they bioprinted brain endothelial cells encapsulated in gelatin methacryloyl (GelMA) and fibronectin into a ring structure, then deposited a GelMA-alginate hydrogel loaded with glioblastoma cells at the center of the ring, followed by layered bioprinting and external perfusion. This novel integration of microfluidics and 3D bioprinting recapitulates the complex structure of glioblastomas, providing a valuable biological tool for studying cancer mechanisms and therapeutic interventions (Figure 3B).
3.2.2. Forming a vascular network
Vascular network formation is a diverse area of bioengineering, and the approach discussed here differs from the organoid vascularization presented in the Section 3.1.2. This method refers to engineered vascularization using 3D bioprinting to replicate the native vascular topology of in vivo tissues, followed by the application of microfluidic principles to simulate blood flow through perfusion.
For example, Fritschen et al.120 integrated on-demand positioned bioprinting with robotic manipulation of microfluidic chips to develop an automated platform capable of precisely constructing three tissue models onchip within 60 s, while achieving continuous, unmanned fabrication of multi-organ chips. The core innovation lies in the creation of sealable post-printing microfluidic chips compatible with mainstream bioprinters and perfusion systems. When validated using a vascularized liver cancer model, complete 3D vascular networks formed within 14 days, with HepG2 cells exhibiting significant spheroidal proliferation and sustained albumin secretion— demonstrating the functional viability of the engineered tissue. However, coaxial bioprinting remains challenging for vascularizing large constructs with 3D interconnecting channels. This challenge can be partially addressed by applying void-free 3D bioprinting techniques to hydrogelbased customized microfluidics. Specifically, sacrificial bioinks containing endothelial cells are deposited layer-by-layer alongside matrix bioinks to generate void-free, multimaterial structures. The sacrificial material is subsequently removed to create well-defined templated flow channels, which were then connected to a peristaltic pump using a polydimethylsiloxane sleeve for controlled perfusion. This system enables the fabrication of large 3D vascularized constructs with interconnected channels while autonomously maintaining stable perfusion and cell viability.121
3.2.3. Optimizing bioprinting parameters
The integration of microfluidics with 3D bioprinting facilitates the optimization of key parameters, including print concentration, resolution, and stability. Traditional extrusion bioprinting techniques are limited by the predetermined concentration of cell-laden bioinks. However, Serex et al.122 developed a microfluidic-based printhead capable of adjusting cell concentration in real time. This system can deliver up to 10 million fibroblasts per milliliter, enabling the bioprinting of highly concentrated cells. This approach yields cell densities that more closely resemble those found in living tissues, reducing intercellular distances and promoting cell–cell communication. In addition, microfluidic enhancements to 3D bioprinting nozzles can significantly improve printing resolution and stability. Highley et al.123 created a droplet-based T-shaped microfluidic system that generates microgels capable of further crosslinking to enhance structural stability. These inks exhibit shear-thinning behavior—flowing under external force but rapidly recovering their mechanical properties post-extrusion. This approach minimizes discrepancies between bioprinted structures and their computer-aided designs while maintaining cell viability, thereby overcoming major limitations related to stability and resolution in 3D bioprinting (Figure 3C).
3.2.4. Automation and dynamic monitoring
The combination of microfluidics and 3D bioprinting enables automated handling and dynamic monitoring of biological models. Traditionally, cells are introduced into microfluidic devices manually using pipettes—a process that increases the risk of contamination by laboratory personnel and introduces variability. Using a 3D bioprinter in a sterile environment minimizes human interference, reduces contamination risk, and enhances reproducibility.27 In addition, while conventional bioprinting methods lack the capacity to deliver realtime data on cell metabolism and cell culture reliability, embedding 3D-printed constructs into sensor-integrated microfluidic devices allows for dynamic monitoring. Dornhof et al.117 demonstrated that combining bioprinting with an on-demand drop-processing microfluidic system can ensure environmental sterility, reliable data acquisition, and seamless sensor integration during model cultivation. Their device demonstrated accurate and stable electrochemical oxygen sensing across atmospheric to hypoxic conditions. Such engineered systems, with high automation and scalability, offer broad potential for future biomedical applications (Figure 3D).
3.3. Organoids and 3D bioprinting
The combination of organoids with 3D bioprinting technology enables the precise spatial placement of cells and biomaterials to construct localized stem cell microenvironments in a targeted manner. This approach not only effectively reduces the randomness and nonreproducibility of traditional culture processes but also facilitates the establishment of highly biomimetic models.124–126 In addition, integrating these techniques transforms conventional bioprocessing methods and significantly improves the throughput and fidelity of organoid production.127,128
3.3.1. Enhancing structural and functional complexity of organoid models
The combination of organoid and 3D bioprinting enhances the architectural complexity of tissue models, modulates intrinsic biological properties such as cell migration and proliferation, and improves the scalability and applicability of composite cultures. For instance, in the context of bone tissue engineering, 3D bioprinting enables the precise fabrication of osteochondral defects with layered structures. Based on the bilayered nature of native osteochondral cartilage, a study precisely prepared an anisotropic bicellular living hydrogel embedding articular chondrocyte progenitor cells and bone mesenchymal stem cells using dual-channel extrusion bioprinting. This construct demonstrated effective cartilage–bone–vessel crosstalk during testing, successfully reconstituting the harmonious cartilage–bone interface and offering insights into the fabrication of anisotropic living material for complex organ reconstruction.129
Furthermore, the synergy between organoids and 3D bioprinting enables the precise fabrication of biomimetic corneal architectures, as validated by Sorkio et al.130 They generated physiologically relevant 3D corneal constructs using corneal limbal epithelial stem cells and adipose tissue-derived stem cells for epithelial and stromal components, respectively. Recombinant human laminin and human collagen I were used in bioinks, combined with laser-assisted bioprinting (LaBP) to fabricate corneal tissues. The resulting constructs included stratified corneal epithelium, stroma, and the epithelium–stroma interface (Figure 4A). These tissues showcased structural integrity, proper physiological function, and expression of relevant marker proteins, confirming the feasibility of LaBP with human stem cells for corneal applications. The integration of 3D bioprinting into organoid fabrication is expected to yield structures with physiological resemblance to host tissues and robust functional performance.
Figure 4. 3D bioprinting and organoids. (A) Laser-assisted bioprinting of layered 3D tissues using human stem cells to mimic natural corneal tissue. Adapted with permission from ref.130, Copyright © Elsevier 2018. (B) Control of self-organization from millimeter to centimeter scales by combining 3D bioprinting and organoid technologies. Adapted with permission from ref.131, Copyright © Nature 2021. (C) Automated extrusion-based bioprinting improves the throughput, quality, scalability, and structure of kidney organoid production. Adapted with permission from ref.127, Copyright © Nature 2021. Abbreviations: CHIR, CHIR99201; FGF9, fibroblast growth factor 9.
3.3.2. Engineering stem cell microenvironments for reproducible organoid development
The combination of organoid and 3D bioprinting allows for precise spatial control of stem cells and biomaterials, facilitating the targeted generation of specific structures. This convergence provides unprecedented control over 3D spatial deposition, enabling tailored tissue engineering approaches. For instance, stem cells used as base materials for organoid construction can be deposited in positions conducive to spontaneous extracellular mesenchymal self-organization. By precisely regulating spatial location and cell density, researchers have fabricated centimeterscale tissues exhibiting self-organizing features, such as tubular lumens, branching vascular networks, and tubular epithelia (Figure 4B).131
In addition to cell deposition, local modulation of biophysical or biochemical cues can direct organoid development and reduce variability. Gjorevski et al.39,132 demonstrated this by using a locally softened hydrogel to guide the geometry and patterning of intestinal organoids. Crypt-like buds formed preferentially within softened regions, while none developed outside them. These pseudobuds extended into crypt-like structures, indicating that localized ECM mechanics and hydrogel topology can be used to control organoid size, shape, and developmental trajectory. This advancement addresses organoids’ inherent variability and low reproducibility, enhancing their utility in both basic and translational research.
3.3.3. Fabricating high-fidelity, high-throughput, and high-efficiency organoids
Organoids derived from PSCs offer promising models for disease research and drug screening due to their structural and functional similarity to native tissues. However, their development and application in both basic and clinical medicine are hindered by limitations such as high variability, low throughput, and small-scale production inherent to conventional methods. Stem cellbased 3D bioprinting enables efficient and reproducible fabrication of high-throughput organoid cultures with biomimetic fidelity. For example, Lawlor et al.127 employed extrusion-based 3D cell bioprinting to rapidly generate kidney organoids with highly consistent cell numbers and viability. They demonstrated that manual organoid generation could be replaced by bioprinting in 6- or 96-well formats, with the device capable of printing approximately 200 organoids in just 10 min. Experimental results further showed that the bioprinted kidney organoids exhibited mature renal structures. By leveraging 3D bioprinting’s ability to precisely manipulate biophysical properties— including organoid size, cell number, and geometry— the platform enables structural fidelity, quality control, improved throughput, and scalability, thereby facilitating both in vitro and in vivo applications of stem cell-derived human kidney tissue (Figure 4C). In addition, this dualtechnology approach facilitates the automation of high-throughput bioprocessing for patient-derived tumor organoids, thereby circumventing the risk of organoids adhering to the sides of multiwell plates and forming cultures confined to two-dimensional growth.133
3.4. Pairwise integration challenges among microfluidics, organoids, and 3D bioprinting
The integration of microfluidics and 3D bioprinting presents several challenges due to conflicts between fluid dynamics and material properties. Microfluidics relies on precise fluid control (e.g., laminar flow and pressure gradients), whereas 3D bioprinting requires bioinks with specific rheological properties (e.g., shear-thinning behavior and rapid gelation).134,135 The dynamic fluid environment in microfluidics may interfere with bioink stability, resulting in the collapse of printed structures or reduced resolution.136,137 For example, the shear stress within microfluidic chips can disrupt the crosslinking process of bioinks, directly affecting printing accuracy.138
In addition, a mismatch exists between the scale of microfluidic channels and the resolution of 3D bioprinting, making it difficult to seamlessly integrate complex printed structures (e.g., vascular networks) with microfluidic systems. This often leads to leakage or pressure imbalances.139,140 Another major issue involves the conflict between dynamic fluid environments and static scaffolds.141 Periodic perfusion in microfluidics can become uneven due to obstruction by printed scaffolds, causing local nutrient or oxygen gradient disparities and negatively affecting cell viability. For instance, printed scaffolds may block microfluidic channels or hinder uniform fluid diffusion.142
A critical challenge in integrating microfluidics with organoids lies in the contradiction between the dynamic culture environment provided by microfluidic systems and the stable conditions required for organoid self-organization.143 Dynamic perfusion can disrupt the mechanical stresses and chemical gradients essential for organoid development, leading to structural abnormalities or functional deficiencies.144 Material compatibility is another significant concern. Common microfluidic materials, such as polydimethylsiloxane, tend to absorb small molecules critical for organoid culture (e.g., growth factors, lipids), resulting in imbalanced media composition and impaired organoid development.145 Furthermore, the implementation of real-time monitoring remains limited. The dense 3D architecture of organoids often obstructs optical imaging signals, reducing data readability within microfluidic systems. Invasive sensors, while potentially useful, may damage organoid integrity and further hinder functional analysis.146,147
The fundamental conflict in combining 3D bioprinting with organoid technology stems from the mismatch between printing resolution and the native structural complexity of organoids.148 Current 3D bioprinting technologies may lack the resolution necessary to replicate the fine, intricate architecture of organoids, potentially resulting in a loss of physiological function.149 For instance, the bioprinted liver organoids may fail to form functional bile ducts due to the absence of subcellular-level topographical features.150 The mechanical properties of bioinks also pose a major constraint: scaffolds that are too stiff may hinder organoid self-organization—such as lumen expansion in intestinal organoids—and current technologies for dynamically tuning scaffold stiffness remain immature.151 Additionally, pre-designed vascularization strategies (e.g., sacrificial material-based methods) may not synergize well with angiogenic signals secreted by organoids, resulting in poor perfusion efficiency.152
These core challenges stem from mismatches in physical scale, imbalances between dynamic and static environments, incompatibility at the material–biological interface, and interference from monitoring tools with the native microenvironment.136,143,145 Addressing these incompatibilities will require innovations in biomaterials, multi-scale interface engineering, and interdisciplinary approaches.
4. Combination of microfluidics, organoids, and 3D bioprinting
While the integration of dual technologies has introduced new perspectives in the biomedical field, there remains a need for a third technology to complement and enhance their capabilities. Each of the three technologies— microfluidics, organoids, and 3D bioprinting—offers unique advantages that play irreplaceable roles in high-precision modeling, making their integration essential. As technological convergence advances, an emerging research paradigm is actively investigating the combined application of these three approaches. This integration aims to achieve higher-quality model construction and to develop more accurate biomedical research protocols, ultimately advancing the understanding of complex biological systems and enhancing the efficacy of biomedical applications.
4.1. Endothelialized myocardial fabrication and its application to cardiovascular toxicity assessment
Zhang et al.118 proposed an innovative strategy that integrates microfluidics, organoids, and 3D bioprinting to successfully engineer endothelialized myocardial tissue. In this integrated approach, endothelial cells were encapsulated within a microfiber lattice created through 3D bioprinting, while a functional vascular bed was formed by utilizing a microfluidic system to guide human umbilical vein endothelial cells to migrate to the periphery of the microfiber structure. This microarchitecture not only allowed for precise control over cell distribution but also provided an optimal supportive environment for the implantation of human iPSC (hiPSC)-derived cardiomyocytes. Building on this foundation, 3D bioprinting further enabled the precise construction of myocardial tissues by controlling cellular arrangement and tissue structure. This resulted in the formation of cardiomyocytes capable of spontaneous, synchronized contractions. Such cellular organization not only enhances the accuracy of functional simulation within the tissue but also significantly improves its physiological relevance.
Combined with a specially designed microfluidic perfusion bioreactor, this model demonstrated significant advantages in cardiovascular toxicity screening. The microfluidic system orchestrated the microenvironment of the endothelialized myocardial chips by regulating nutrient delivery and waste removal—processes essential for maintaining healthy tissue growth. This enhanced environmental control promotes optimal cellular function and offers a more physiologically relevant platform for assessing the effects of compounds on cardiovascular health, thereby improving the accuracy and reliability of toxicity assessments.
Compared with rat-derived myocardial organoids, hiPSC-derived myocardial organoids exhibited slightly lower endurance at all time points, underscoring the benefits of using organoid models to minimize species differences. This comprehensive model addresses limitations associated with individual technologies and enhances myocardial tissue simulation and function through the synergistic combination of all three. Through the strategic convergence of microfluidics, organoids, and 3D bioprinting, this integrated platform establishes an innovative paradigm for accelerated drug discovery and pathophysiological modeling, facilitating precise interrogation of human-specific biological responses while enhancing translational fidelity in preclinical evaluations.
4.2. Vascularized glioblastoma-on-a-chip model and its application in mechanobiological studies of brain tumors
Pioneering 3D organoid models that incorporate dynamic flow and volumetric cues have emerged as transformative platforms for bridging the current gap in effective cancer treatment within in vitro exploration platforms. These models are crucial for accurately mimicking the tumor microenvironment and facilitating studies on intercellular interactions under tumor-specific pathophysiological conditions. Silvani et al.119 successfully reconstructed the complex brain tumor microenvironment, including a functional BBB and surrounding 3D perivascular tumor microhabitat, by faithful recapitulating physiological shear stress and the mechanical interactions between cells and the matrix. A glioblastoma (GBM) model was assessed using a microfluidic chip under simulated microgravity conditions. The results indicated significant changes in cellular morphology and mechanotransduction responses, highlighting the critical role of gravity in the mechanoregulation of GBM.119
This model was developed to characterize a novel 3D microfluidic bioprinting system for vascularized GBM-on-a-chip constructs, designed to comprehensively replicate the pathophysiological conditions of tumors and their surrounding vascular microenvironment. In vivo, GBM typically manifests as a dense, spherical structure with distinct morphological characteristics associated with various regions of the brain tumor microenvironment. These regions include a necrotic core, a perivascular zone with a severely compromised BBB, and adjacent healthy brain tissue with an intact barrier that effectively restricts drug diffusion. A bioprinting strategy employing dual bioinks was used to engineer vascularized tissue constructs with perfusable lumens. Initially, a GelMA– fibronectin mixture encapsulating brain endothelial cells was printed as a ring-shaped outer region, followed by a GelMA–alginate core bioink loaded with GBM cells. Experimental results demonstrated that under gravity-free conditions, GBM cell invasiveness and aggregation were significantly suppressed.
This integrated model showcases the highly synergistic effects of microfluidics, organoids, and 3D bioprinting, offering a robust platform for simulating complex tumor microenvironments and investigating GBM pathophysiological.
4.3. Microfluidic printhead-based bioprinting with high cell concentration and its application to bladder-like organ fabrication
Organoids have become essential tools in preclinical research due to their remarkable ability to closely mimic human tissues. However, for applications such as drug screening, it is essential to ensure not only high fidelity but also methodological reliability and reproducibility. 3D bioprinting has emerged as a viable strategy to meet these criteria, offering precise control over tissue morphology and architecture. Nonetheless, conventional extrusion bioprinting often struggles to replicate the intricate tissue complexity found in native organs. This limitation arises because the technique typically involves dispensing cell solutions at predefined concentrations through a needle. To minimize cell lysis or loss in dead volume, researchers tend to employ diluted cell solutions—concentrations lower than those found in living tissues—which diminishes bioactivity and increases cost.122,153
Recognizing the importance of cell concentration for organoid formation, Serex et al.122 developed a microfluidics-based printhead capable of real-time adjustment of cell density. Their system achieved fibroblast concentrations of 10 million cells/mL with precise volumetric dispensing and was successfully applied to the generation of bladder organoids with preserved urothelial functionality. Hematoxylin and eosin staining confirmed that these organoids retained their original morphology, including a distinct central lumen and multilayered cellular structure.
Immunostaining further confirmed the expression of key urothelial markers such as CD44, CK13, and CK5, affirming cellular coherence and functional relevance. These results indicate that the microfluidic printing technology can generate organoids with multicellular layers and complex structures. Such advancements not only enhance the physiological relevance of organoids but also improve the controllability and reproducibility of the production process, offering an innovative and efficient solution for large-scale organoid generation. This approach significantly reduces variability caused by manual manipulation and standardizes the organoid formation process, thereby promoting the application of organoids in biomedical research. As a result, they represent powerful tools for disease modeling and drug screening.
4.4. Fabrication of 3D tumor spheroids via bioprinting and sensor integration for cellular metabolism monitoring
Three-dimensional cellular agglomerates, such as microtissues, organoids, and spheroids, are increasingly recognized as pivotal modeling tools in biomedical research. These structures are capable of accurately mimicking the functions of in vivo tissues under in vitro conditions and are increasingly utilized in cancer research and organ-on-a-chip systems. To further enhance the utility of these models, microsensors can provide crucial real-time information about cellular metabolism and the reliability of culture conditions. However, 3D cell cultures, particularly individual spheroids, still face persistent challenges related to reproducible formation, precise localization, and the acquisition of meaningful biosignals when integrated with sensors. These challenges become even more pronounced when working with high cell volume ratios in close proximity to sensing elements.117
To address these challenges, Dornhof et al.117 successfully automated the precise printing of tumor spheroids into the microvias of a chip-based electrochemical oxygen sensor array using advanced 3D bioprinting technology. This innovative approach overcomes issues of shape instability and culture failure in organoid fabrication caused by operator inexperience, achieving highly accurate and reproducible spheroid generation. The diameters of the spheroids can be controlled to approximately 200 μm, with a deposition accuracy of up to 25 μm and a volume of 22 nL per droplet.
Additionally, the microstructure and hydrogel-coated micropores are designed to precisely position individual MCF-7 breast cancer spheroids near the sensor electrodes. The microelectrode pores are encapsulated to facilitate rapid oxygen concentration measurements in a volume of 55 nL. The system exhibited excellent stability and accuracy as the electrochemical oxygen sensor transitioned from atmospheric to hypoxic conditions. Experimental results demonstrated that the cellular respiration rate of individual tumor spheroids could be measured within a range of 450–850 fmol/min, revealing significant changes in cellular metabolism upon drug exposure.
The study marks the first successful integration of 3D bioprinting with real-time monitoring technologies in 3D cell culture systems. It demonstrates an efficient process for parallelization, sensor integration, and drug delivery in both 3D cell culture and organ-on-a-chip platforms. The system achieves full automation and scalable manufacturing through the transition from conventional microfluidic architectures to a digitally programmable droplet manipulation system, thereby establishing a paradigm shift in liquid handling precision and operational flexibility. This advancement supports greater flexibility in spheroid formation and capture.
The potential applications of this technology are extensive, ranging from basic metabolic studies to standardized cell culture and toxicology experiments, as well as personalized medicine, such as patient-specific chemotherapy.
4.5. Challenges and prospects of combining microfluidics, organoids, and 3D bioprinting
Although the integration of microfluidics, organoids, and 3D bioprinting holds great promise for biomedical applications, their synergistic development still faces multiple challenges. First, the complexity of technological integration lies in achieving compatibility across scales. For example, dynamic perfusion in microfluidic systems and the structural precision of 3D bioprinting must be coordinated within a sub-millimeter to centimeter range, while the heterogeneous growth behavior of organoids may compromise the stability of printed structures.148,154 Second, the functional design of bioinks has yet to fully meet the demands of multi-technology coupling. Bioinks must not only support cell viability and provide a microenvironment conducive to organoid development during printing but also accommodate the fluid dynamics of microfluidic perfusion. This places increased demands on material rheology, degradation kinetics, and the ability to transmit biochemical signals.145 In addition, significant bottlenecks remain in the real-time acquisition and analysis of multimodal data. For instance, in situ monitoring of organoid functional evolution under dynamic culture conditions requires the development of novel biosensors with enhanced signal sensitivity, improved spatial resolution, and greater integration compatibility with hybrid microfluidic–bioprinting systems.155 Finally, the lack of standardized frameworks limits the scalability and translational potential of these integrated technologies. Unified standards are urgently needed for crossplatform workflow harmonization, quality assessment metrics, and clinical validation protocols.156
Despite the challenges, the deep integration of microfluidics, organoids, and 3D bioprinting presents a unique and promising future. By leveraging the precise microenvironmental control of microfluidics, the biological fidelity of organoids, and the complex structural fabrication capabilities of 3D bioprinting, this technological convergence has the potential to overcome many limitations of traditional in vitro models. For instance, dynamic perfusion enabled by microfluidics can enhance the maturation of vascular networks within bioprinted tissues, while high-resolution bioprinted biomimetic scaffolds can spatially guide organoid self-assembly, resulting in multifunctional tissues with physiologically relevant vascular–parenchymal interfaces.120,157 In terms of functional enhancement, this tri-technology integration allows simultaneous achievement of topological control over cell alignment (e.g., directed contraction in cardiac tissue), simulation of mechanical microenvironments (e.g., shear stress response in the BBB), and metabolic zonation (e.g., oxygen gradients in tumor spheroids), thereby significantly improving the pathophysiological relevance of the resulting models.119,154,158 Moreover, the combination of microfluidic-driven real-time cell density regulation with feedback-controlled bioprinting parameters could enable a closed-loop manufacturing platform for organoids. Such a system would support both high cell viability and high-throughput production, facilitating the standardized fabrication of patientspecific models for personalized medicine.122
Looking ahead, as interdisciplinary technical barriers are progressively overcome, the organic integration of these three technologies is expected to drive a paradigm shift— from static structural mimicry to dynamic functional biomimicry in in vitro models. This advancement will provide a more powerful technological engine for disease mechanism studies, drug development, and regenerative medicine.
5. Advances in the application of combined technology in biomedical fields
Conventional modeling systems face significant limitations in clinical translation, including the inability to adequately simulate the complex physiological environment of the human body, discrepancies in biomarker expression, genetic and metabolic differences, and the lack of disease-specific environmental cues. These shortcomings often hinder the direct application of experimental findings to human patients. The emergence of microfluidic, organoid models, and 3D bioprinting technologies provides new opportunities to overcome these barriers. However, given the inherent biological complexity, no single technology can fully replicate all aspects of human physiology. Consequently, the integration of multidisciplinary technologies to faithfully recapitulate human pathophysiological complexity has emerged as an imperative in contemporary biomedical research. This technological convergence has been widely applied in multiple biomedical research domains, including drug research, disease modeling, immunology, clinical treatment, and the integration of other advanced technologies. Its potential application value is substantial and will inevitably provide an important impetus for the advancement of the biomedical field.
5.1. Pharmaceutical research
The demand for pharmaceutical research—closely tied to human health and survival—is ever-increasing. In developing in vitro test models, drug research must address target tissue functionality, monitor drug metabolism, and improve both data readability and model throughput.
Although pharmacokinetics (PK) represents a critical discipline for drug development, conventional models often fail to simulate or predict human-relevant metabolic parameters with sufficient quantitative accuracy. Herland et al.159 constructed the first human physiological PK model based on the integration of microfluidic and organoid technologies. This model simulates the complete physiological processes of drug absorption, metabolism, and excretion and accomplishes an accurate prediction of PK parameters. At the core of the system is a fluid-coupled organoid chip that connects fluid streams via endothelial channels and shares a common arterial–venous mixing reservoir. This design not only ensures physiological stability and accurate data collection but also accomplishes the quantitative translation of PK parameters from in vitro to in vivo contexts (Figure 5A).
Figure 5. Application of dual-technology combinations—Part I. (A) A physiologically relevant pharmacokinetics model of drug absorption, metabolism, and excretion using microfluidics and organoids. Adapted with permission from ref.159, Copyright © Nature 2020. (B) High-throughput tumor organoid drug screening using bioprinting. Adapted with permission from ref.161, Copyright © Nature 2023. (C) Microfluidic culture of cortical organoids derived from hiPSCs used to model early brain development and VPA exposure. Adapted with permission from ref.163, Copyright © Nature 2020. Abbreviations: AV, arteriovenous; ATP, adenosine triphosphate; EB, embryoid body; hiPSC, human induced pluripotent stem cells; HSLCI, high-speed live-cell interferometry; VPA, valproic acid.
Similar to PK models, toxicology studies require high data fidelity and the ability to reproduce human-specific scenarios. Focusing on the liver, an area of high relevance for drug toxicity, Vurat et al.160 combined 3D bioprinting and microfluidic technology to construct a multicellular microtissue model mimicking the periodontal ligament– alveolar bone interface. The bioprinted constructs demonstrated stable mechanical, thermal, and rheological properties. Structures embedded with hydroxyapatite-coated magnetic nanoparticles exhibited enhanced compressive strength. Under dynamic culture conditions sustained over 10 days, the constructs retained their morphological integrity and supported uniformly distributed, viable cells. Immunofluorescence analysis revealed that various cell types expressed characteristic markers within their respective matrices and established direct interactions, indicating robust cellular permeability. Subsequent drug interaction studies demonstrated the platform’s capability to assess both drug uptake and toxicity, highlighting its potential application in periodontal disease research.
Drug screening—a critical step in pharmaceutical pipelines—has long been hindered by the inability of traditional platforms to accurately recapitulate human tissue architecture and to support clinically relevant, scalable screened. Tebon et al.161 developed bioprinted tumor organoids related to time-resolved imaging via machine learning-based single-organoid analysis and high-speed live-cell interferometry. These models allowed precise, parallel mass monitoring of multiple cultures, enabling identification of organoids with transient or sustained sensitivity or resistance to specific therapies. This approach effectively reflects drug efficacy and facilitates targeted drug screening (Figure 5B). In addition, the application of a 3D acoustic assembly device to cellular spheroid fabrication enabled large-scale production of viable cellular aggregates. Rigorous validation confirmed high-efficiency spheroid formation with sustained viability, underscoring the translational potential of integrating organoid technology into robust, high-throughput drug screening pipelines.162
In summary, the combined application of microfluidics, organoids, and 3D bioprinting enables the dynamic construction of physiological models, the accurate transformation of pharmacological parameters, and efficient high-throughput drug screening. This integration helps modern drug research surpass the limitations of isolated culture systems and move toward a more comprehensive, systematic drug evaluation framework.
5.2. Disease modeling
In disease modeling, researchers are particularly concerned with the degree of biological fidelity with which in vitro cultures replicate in vivo processes. The development of ideal human physiological models is essential for exploring the complex and diverse mechanisms of the human body and is critical for building a comprehensive understanding of biological systems. Furthermore, studying diseasespecific mechanisms across different organs requires the modeling of key structures and functions that accurately reflect corresponding physiological properties and adaptations.
Cui et al.163 developed a cortical organoid model using a micropillar array to mimic human brain development in a controlled manner. This model captures the physiological characteristics of early prenatal brain development and reveals the neurodevelopmental effects of the antiepileptic drug valproic acid, thereby expanding the breadth of research on brain development and neurological diseases (Figure 5C).
Yin et al.164 integrated microfluidic bioprinting with coaxial flows of polymer and crosslinker to fabricate thin, biocompatible, and reproducible hydrogel structures that mimic the epithelial features of the salivary gland. The platform supports the following two printing modes: solid hydrogel fibers with diameters below 100 µm, which can be arranged into millimeter-scale grids, and hollow tubular structures with outer diameters ranging from 0.6 to 2.2 mm and wall thicknesses between 45 and 80 µm, both exhibiting confirmed luminal patency. In both configurations, salivary gland cells were printed at high densities while maintaining their phenotype and viability, highlighting the potential of this strategy for multiscale hydrogel patterning and microscale tissue engineering applications.
Despite substantial advances in in vitro model development, there remains a dearth of models that can simultaneously recapitulate the full complexity of the tumor microenvironment, including its diverse cellular components and genetic properties. A vascularized lung cancer model integrating organoid and 3D bioprinting technologies addresses this limitation. This model incorporates idiopathic pulmonary fibrosis-derived lung fibroblasts and a novel porcine lung-derived decellularized ECM hydrogel, effectively replicating the local tumor microenvironment. Such a system enables the development of targeted therapies and the identification of biomarkers for lung cancer patients with fibrosis.165
In addition, a microfluidic intestinal organoid chip, modeled after the natural intestinal villus structure, successfully addresses the in vitro–in vivo oxygen dynamics mismatch by improving oxygen exchange efficiency and supporting complex and diverse biological responses. This system sets a paradigm for next-generation, multisystem-interacting organoid chips aimed at precision therapeutic applications.166
For cardiovascular toxicity evaluation, Zhang et al.118 proposed a strategy integrating three technologies to construct a vascularized cardiac tissue model. This model utilized 3D bioprinting to generate microfiber scaffolds and employed a microfluidic system to precisely control the spatial distribution of endothelial cells, thereby mimicking the vascular bed structure and providing functional support for hiPSC-derived cardiomyocytes. The engineered tissue not only exhibited synchronous contraction but also maintained a stable microenvironment through a perfusion system, thereby enhancing the sensitivity and accuracy of toxicity screening.
5.3. Mimicking immune interaction
The mammalian immune system, an extraordinary sensory system for detecting and neutralizing pathogens, plays a critical role in tumor development. The precise recapitulation of intercellular communication networks and faithful reconstruction of tissue-specific immune niches constitute fundamental imperatives in advancing contemporary immunological research paradigms. For example, hepatocellular carcinoma organoid microarrays constructed by co-culturing mesenchymal stromal cells with peripheral blood mononuclear cells partially replicate the tumor microenvironment and provide a promising platform for predicting immunotherapeutic responses in hepatocellular carcinoma patients (Figure 6A).167
Figure 6. Application of dual-technology combinations—Part II. (A) Co-culture of MSCs and PBMCs to construct HCC organoids mimicking the primitive tumor microenvironment. Adapted with permission from ref.167, Copyright © Wiley 2023. (B) Neural organoids derived from hiPSCs cultured in a specialized microfluidic device to generate artificial synapse bundles for transplantation. Adapted with permission from ref.171, Copyright © Springer Nature 2024. (C) Liver-on-a-chip model constructed by bioprinting hepatocyte extracellular vesicles onto a microfluidic chip to evaluate the metastatic behavior of reprogrammed triple-negative breast cancer cells in an in vitro 3D liver microenvironment. Adapted with permission from ref.173, Copyright © IVYSPRING 2023. Abbreviations: BM-MSC, bone marrow-derived mesenchymal stem cell; DSF, dual-scale flow; HCC, hepatocellular carcinoma; hiPSC, human induced pluripotent stem cells; PBMC, peripheral blood mononuclear cell; PDO, patient‐derived organoid; PMMA, polymethyl methacrylate.
Similarly, a co-culture tumor immunity model containing patient-derived gastric cancer organoids and tumor-infiltrating lymphocytes mimicked the migration and functional activation of tumor-infiltrating lymphocytes, providing preliminary insights into the mechanisms of both active and passive cell migration.168
In addition, a study combining 3D bioprinting and microfluidics demonstrated the potential of fusion technologies for immunoassays. Aimed at accelerating lateral chromatography immunoassays for novel coronavirus screening, the study used microfluidics and 3D bioprinting for protein deposition and shell unit formation, respectively, thereby optimizing and expediting the assay process.169
5.4. Personalized therapies
Individualized clinical therapy programs often provide interventions tailored to a patient’s specific condition and needs. These personalized therapies are innovative not only in their integration of emerging scientific and technological advancements but also in their use of bioinspired models to assess clinical feasibility and facilitate clinical application.
For example, in cardiovascular disease treatment, the combination of microfluidic molding and coaxial bioprinting technologies enables the fabrication of freestanding, perfusable vascular structures. These structures support the precise integration of relevant vascular cells in multilayered and biomimetic configurations. Mechanical evaluations in tension and bending confirmed the model’s feasibility in percutaneous coronary intervention, demonstrating its significant potential in vascular bionics.170 Similarly, in cases of severe peripheral nerve injury requiring autologous nerve grafts, specific microfluidic devices have been employed to promote the generation of artificial nerve synapse bundles derived from hiPSCs, with physiologically favorable correlations and functional states. This approach represents a promising, safe, and effective peripheral nerve treatment (Figure 6B).171
In addition, a study utilizing digital light processing printing successfully engineered and printed bone-healing tissue organoids. The resulting models exhibited stagespecific gene expression patterns reflecting biological processes such as endochondral ossification, underscoring their potential for rapid regeneration and recovery from bone injuries.172 Overall, the combination of organoids, 3D bioprinting, and microfluidics holds great promise for the development of sustainable and environmentally friendly medical solutions, contributing significantly to the future of medical innovation.
5.5. Cross-disciplinary applications
The current era is marked by the co-evolution of multiple disciplines, fostering innovation, and driving integration across fields. Within this broad context, advanced technologies such as organoids, microfluidics, and 3D bioprinting are playing increasingly pivotal roles, especially in their fusion with other advanced technologies. The synergistic integration of these technologies transcends conventional disciplinary boundaries in biomedicine, establishing a catalytic framework that propels interdisciplinary innovation across precision medicine, biomimetic engineering, and advanced material science. Specifically, cross-disciplinary integration not only compensates for the limitations inherent in any single technology but also facilitates a more accurate representation of physiological and pathological processes in vivo, thereby accelerating clinical translation. For example, Lu et al.173 demonstrated the feasibility of reprogramming triple-negative breast cancer cells into hepatocytes using a simplified reagent preparation method, integrated with microfluidics and 3D bioprinting technologies. Through single-cell sequencing analysis and liver-on-a-chip modeling, the study confirmed both the reprogrammed phenotype in a hepatic microenvironment and the ability to inhibit cancer cell metastasis, offering a novel therapeutic approach for triple-negative breast cancer liver metastasis (Figure 6C).
In the field of biomics, Bues et al.174 used DisCo microfluidics to address the challenge of low-input samples in single-cell RNA sequencing. By combining machine vision with microfluidics, this approach efficiently captured single-cell samples with high throughput and precision. Their analysis of intestinal organoids and mouse intestinal crypts demonstrated the innovative application of DisCo technology in revealing organoid heterogeneity and cellular composition.
In materials science, Qazi et al.175 developed a photosensitive polymer hydrogel enabling in vivo 3D bioprinting within living mouse tissues. They successfully localized complex structures such as the dermis, skeletal muscle, and brain, and demonstrated muscle fiber formation within mouse muscle tissue, opening a promising avenue for minimally invasive surgical organ repair. Similarly, Daly et al.176 applied bioprinting to transfer organoids into self-healing support hydrogels, facilitating the scalable production of high-cell-density microtissues that mimic cardiac scar tissue post-myocardial infarction. This technological combination enables both in vivo 3D bioprinting and the large-scale fabrication of functional microtissues, advancing applications in organ repair and disease modeling.
As the cross-application of organoid, microfluidic, and 3D bioprinting technologies progresses, their cross-disciplinary application marks the advent of a transformative era in regenerative medicine and biomedical engineering.
5.6. Obstacles to transformation and future prospects for technology integration
At the translational level, the widespread implementation of microfluidics, organoids, and 3D bioprinting faces multiple practical challenges. The complexity of integrating these technologies results in high development and maintenance costs. Additionally, the need for precision manufacturing and specialized materials raises the industrial entry threshold, limiting accessibility for small- and medium-sized institutions.177
A major barrier lies in the absence of unified technical standards and quality assessment systems. Significant variability in culture environments, printing parameters, and detection methodologies across platforms impedes the development of reproducible, standardized workflows, severely restricting inter-laboratory data sharing and translational efficiency.178 Clinically, long-term stability of organoid models, functional validation of vascularized constructs, and the immunocompatibility of bioprinted tissues remain critical concerns that require large-scale animal studies and long-term safety evaluations. Regulatory guidelines for approving such emerging integrated technologies remain underdeveloped, significantly extending the timeline from laboratory research to clinical implementation.179
Looking ahead, deepening interdisciplinary collaborative innovation is expected to drive breakthroughs across multiple dimensions. Advances in materials science and engineering will facilitate the development of lowcost, highly biocompatible bioinks, and microfluidic chips. When combined with automated control technologies, these innovations can facilitate the construction of intelligent platforms that significantly reduce operational complexity and enhance production efficiency.180
The establishment of standardized technical systems will be accelerated through collaboration among industry associations, research institutions, and enterprises. This will lead to comprehensive frameworks covering model construction, performance evaluation, and clinical application, offering clear guidance for technology translation.181 In terms of application expansion, integrated technological platforms will significantly empower precision medicine—for example, enabling the development of personalized tumor organoid models for targeted drug screening or the use of bioprinted vascularized tissues for organ repair—thus transitioning clinical practice from empirical approaches to precisionbased strategies.119 The incorporation of artificial intelligence and biosensing technologies will enable realtime monitoring and dynamic regulation of the culturing process, making tissue engineering more intelligent and controllable. This provides technical support for the precise fabrication of complex organ models.182 In the long term, building an innovation ecosystem that deeply integrates government, industry, academia, research institutions, and clinical practice—along with a robust ethical and regulatory framework—will accelerate the translation of technological advancements into healthcare benefits. This will create a seamless pipeline from laboratory research to clinical application, bringing revolutionary breakthroughs to regenerative medicine and precision healthcare.183
6. 4D culture
While the integration of microfluidics, organoids, and 3D bioprinting facilitates the creation of tissue models, significant differences remain compared to the developmental trajectories of real tissues and organs in vivo. These discrepancies primarily stem from the lack of precise and targeted induction of key biological processes such as cell differentiation, migration, proliferation, and apoptosis. Although current systems can support ab initio culture, they lack the auxiliary induction and precise regulation required at critical physiological developmental stages, resulting in a substantial gap between in vitro models and native tissue development.
Introducing time as a variable—transforming 3D into 4D culture—may further optimize the simulation of tissue growth and development. This approach could help narrow the gap between cultured models and the formation of functional, physiologically accurate tissues. Achieving this goal hinges on a thorough understanding of the regulatory factors orchestrating spatiotemporal induction, allowing for targeted interventions at key developmental checkpoints in accordance with the principles governing tissue development. Some experiments have already successfully demonstrated the precise induction of directed cell differentiation and development by modulating controllable factors. These findings provide valuable references for further optimizing tissue model development.
Specific growth geometries significantly influence directed cell differentiation. By shaping organoids to mimic organ-specific morphogenetic processes, stem cells can be guided to differentiate along predefined spatial boundaries and form tissues with distinct structural features. This strategy not only replicates the 3D architecture of various organs but also allows for precise control over stem cell fate and tissue morphogenesis. For instance, research by Liberali and Schier33 demonstrated how cystic gut-like organoids can be shaped into the crypt and villus structures characteristic of the intestinal tract. Through the synergistic integration of tissue engineering platforms and principles of cellular self-organization, intestinal stem cells were guided to recapitulate tubular epithelial morphogenesis with a patent lumen, resulting in physiologically relevant intestinal architectures. These organoids not only spatially reproduce the in vivo structures of crypts and villi but also exhibit key physiological features and strong regenerative capacity. Notably, specialized cell types rarely seen in conventional organoids were successfully induced, enhancing physiological relevance.
Further studies revealed that the emergence and formation of crypt structures in organoids can be predicted and controlled simply by manipulating their initial geometry. Researchers utilized localized light exposure to soften specific areas of the substrate, creating initial geometries that directed where crypts would form. Crypt formation occurred exclusively in the softened regions, while unaffected regions showed no structural changes. Additionally, the timing of substrate softening proved critical, initiating softening 2 days after the onset of stem cell differentiation significantly reduced crypt development.39
Regulation of signaling pathways is also a crucial factor in the process of directed cell differentiation. Specifically, various signaling pathways guide stem cell differentiation by being selectively activated or inhibited at precise time points. For instance, cardamonin has demonstrated the potential to induce osteogenic differentiation in human amniotic mesenchymal stem cells through targeted modulation using small-molecule compounds. At nontoxic concentrations, cardamonin significantly enhanced alkaline phosphatase activity and calcium deposition, while upregulating both early and late osteogenic markers. Pathway analysis revealed that this flavonoid regulates cell fate by increasing the phosphorylation of Janus kinase 2 (JAK2) and signal transducer and activator of transcription 3 (STAT3). Inhibition experiments further confirmed that its effects are mediated through the JAK2/STAT3 pathway, highlighting its potential as a small-molecule osteoinductive agent for regenerative medicine applications.184 Similarly, Cai et al.185 induced the differentiation of hiPSCs into valvular endothelial cells and valvular interstitial cells through the sequential activation of the Wnt, bone morphogenetic protein 4 (BMP4), vascular endothelial growth factor (VEGF), and nuclear factor of activated T cells 1 signaling pathways. They employed CHIR-99021, BMP4, VEGF-165, and forskolin to achieve this modulation. Comparative analyses revealed that the differentiated cells closely resembled primary heart valve endothelial cells and mesenchymal cells in both gene expression profiles and functional characteristics. Furthermore, sequencing technology indicated that the transcriptional trajectories during differentiation closely mirrored those observed in embryonic valve development.
In addition to biochemical cues, the physicochemical environment also plays a critical role in regulating cell differentiation. Oxygen concentration, for example, has a profound effect on erythropoiesis. While hypoxia is typically viewed as a limiting factor in vertebrate embryonic development—due to the need for sufficient oxygen to support metabolism—recent studies suggest that hypoxic conditions can actively induce stem cell differentiation and promote developmental processes. Red lineage differentiation begins early in embryogenesis through oxygen delivery; however, the specific mechanisms regulating this process remain to be fully elucidated.
One study revealed that the vgll4b gene in Zebrafish exhibits a sensitive response to oxygen levels.186 Researchers established a vgll4b mutant zebrafish model using CRISPR/Cas9 technology, which exhibited impaired heme synthesis and disrupted terminal differentiation of erythrocytes. This defect underscores the role of vgll4b as a critical regulator of HIF1α-dependent erythropoiesis. Moreover, it was discovered that the Vgll4 protein inhibits the negative regulation of alas2 expression and heme biosynthesis by binding to Irf2bp2. This mechanistic insight elucidates the pivotal role of oxygen tension in erythropoietic regulation and delineates how metabolic oxygen dynamics orchestrate cellular differentiation trajectories through discrete signaling cascades. Another vital regulatory component is mechanical signaling, which plays diverse roles in cell fate transitions, migration, and morphogenesis. Cells dynamically sense and respond to their external physical environment, including stimuli such as substrate mechanical properties and shear stress. Concurrently, the mechanical characteristics of the cell surface modulate intracellular signaling through feedback mechanisms. This bidirectional regulation underscores the profound influence of mechanical signaling on stem cell fate and further illustrates the complexity of cell differentiation processes.187
In summary, current in vitro factors known to induce directional cell differentiation include external signaling molecules, physical stimuli, and growth geometries. These factors activate specific signaling pathways in various ways, effectively guiding cellular processes. To achieve precise directed differentiation, a deep understanding of the activation mechanisms and regulatory conditions governing these signaling pathways is essential. Technological advancements have revolutionized experimental methodologies, gradually unveiling biological phenomena that were previously challenging to observe using traditional techniques.188 This progress demonstrates the interdependence between the exploration of signaling pathways and technological innovation—each enhancing the other, jointly driving the advancement of scientific frontiers.
The three technologies highlighted in this discussion— organoids, microfluidics, and 3D bioprinting—are pivotal to this endeavor. Organoids offer a physiologically relevant platform that supports the differentiation of stem cells into 3D structures closely resembling native tissues. Microfluidic systems provide spatiotemporally resolved regulation of exogenous signaling gradients and mechanical cues, while dynamically modulating physicochemical parameters to establish precisely controlled experimental milieus. Meanwhile, 3D bioprinting enables the customization of spatial structures and optimization of the culture environment. The integration of these three technologies not only incorporates diverse factors influencing directed differentiation but also facilitates stage-specific activation of key signaling pathways, enabling precise control of the cell differentiation process. Experimental evidence has already demonstrated the feasibility and synergistic benefits of combining these three approaches.95,189
Against this favorable backdrop, we introduce the novel concept of “4D culture,” which integrates organoids, 3D bioprinting, and microfluidics. The 4D culture model extends beyond spatial control to include temporal dynamics. In this system, 3D bioprinting engineers the spatial structure, organoids mimic physiologically relevant differentiation processes, and microfluidics generates a dynamic microenvironment necessary for sustaining these processes. Specifically, microfluidics allows for dynamic regulation of the microenvironment by simulating blood flow, removing necrotic cells, delivering and monitoring nutrients and oxygen, supplying growth factors, and introducing hydrodynamic forces.
This 4D culture paradigm emerges as a synergistic framework for next-generation biomedical technologies, establishing physiologically relevant translational platforms that bridge the gap between preclinical modeling and clinical implementation. Such innovations hold the potential to revolutionize disease management by enhancing predictive diagnostics and therapeutic development, thereby establishing new frontiers in precision medicine.
7. Conclusion
Microfluidics, organoids, and 3D bioprinting are expected to play a central role in modeling systems that require the structural and functional mimicry of natural human tissues. The successful construction of such models depends on the coordinated regulation of various factors, including stem cell proliferation and differentiation, ecological microenvironment control, and spatial precision. It is worth noting that no single technology can overcome all limitations independently; instead, the integrated application of multiple technologies is essential for developing optimal in vitro models.
Recent studies have frequently employed dualtechnology integration, and the resulting fusion models can be broadly categorized into three groups. The first category involves the combination of microfluidics and organoids—an approach that effectively modulates the microenvironment of cultures, facilitates the integration of system-level parameters, enhances vascularization and nutrient delivery, and improves data readability and real-time imaging capabilities. The second category encompasses the combination of microfluidics and 3D bioprinting; such technological integration significantly advances the microscale shaping of pathophysiological tissue structures and culture microenvironments, the formation of engineered vascular systems, the optimization of parameters such as resolution and structural stability, and the ability to automate and dynamically monitor cultures through the interplay of fluid flow control and spatial patterning. The final category consists of the integration of organoids and 3D bioprinting, which enhances the functional characterization of cultures, enables spatial manipulation, and facilitates high-fidelity, highly replicable, and high-throughput bioprocessing.
With continued exploration of microfluidics, organoids, and 3D bioprinting, combinatorial technologies are gaining traction for their clinical translational potential. Initial successes have been observed in areas such as drug evaluation, disease modeling, immunological exploration, therapeutic development, and broader multi-technology integration.
While dual-technology integration introduces novel paradigms for biomedical innovation, the absence of a third synergistic technological modality limits the full optimization of modeling systems. The unique advantages offered by the three technologies are indispensable for constructing physiologically relevant models, and thus, their combined use is essential. As the field evolves, a small number of studies have been geared toward exploring the tri-technology integration of organoids, microfluidics, and 3D bioprinting to achieve more optimal model selection and development.118,190 However, notable discrepancies persist between in vitro models developed through current fusion technologies and the developmental trajectories of real tissues and organs in vivo. Much of this discrepancy stems from the lack of precise, targeted chemotactic guidance for cell fate decisions, including differentiation, migration, proliferation, apoptosis, and the establishment of tissue morphology. Although current fusion systems can initiate culture from scratch, they often lack intentional auxiliary induction and temporal regulation at key physiological checkpoints. As a result, the developmental course of in vitro cultures diverges significantly from that of native tissues. To address this gap, introducing a temporal variable into the culture process offers a promising strategy to better simulate tissue growth and developmental dynamics, thereby narrowing the gap between in vitro models and natural tissue formation. This approach holds potential for broader application in related research fields. It is important to emphasize that classical studies have clearly demonstrated that the temporal activation of signaling pathways is critical for stem cell differentiation, migration, and morphogenesis. For instance, precise regulation of signaling pathways such as Wnt and transforming growth factor-beta not only optimizes the direction of cell differentiation but also facilitates the formation of tissue structures at specific developmental stages.191,192 Additionally, the mechanical properties of the ECM and the optimization of the physical microenvironment further enhance the physiological relevance of in vitro models.193,194
The integration of these factors lays the groundwork for constructing a 4D dynamic culture system, transforming in vitro models from static structures into multidimensional dynamic developmental systems. By introducing the time dimension, we aim to significantly narrow the gap between in vitro cultures and in vivo tissue development, thereby enhancing the physiological fidelity of these models. Such technological advancements not only open new avenues for precise cell differentiation research but also broaden the application potential in disease modeling and drug discovery. We propose, for the first time, a culture approach that adds a temporal dimension to the integrated use of organoids, 3D bioprinting, and microfluidics, and define this system as 4D culture.
In this model, 3D bioprinting is responsible for shaping the spatial architecture; organoids mimic the developmental process of primitive cells differentiating into mature cells; and microfluidics dynamically modulate the microenvironment of organ development and replicate the functions of blood flow, including the removal of necrotic cells, the transport and monitoring of nutrients and oxygen, the delivery of growth factors, and the application of fluidic forces. In addition, 4D culture can serve as a platform for incorporating various emerging technologies aimed at creating improved models for preclinical studies, ultimately enhancing the prediction, diagnosis, and treatment of diseases to better serve biomedical research.
The integration of multiple technologies in in vitro testing methods has profound implications for resource efficiency, data accuracy, and experimental reproducibility. Automation and standardized workflows reduce manual intervention, minimize the risk of human error, and— through the use of data visualization techniques— enable real-time monitoring of experimental data with enhanced resolution, thereby reducing both time and resource consumption. In the long term, these integrated technologies facilitate more accurate simulations of patient-specific biological characteristics, enhancing the precision of drug screening and preclinical trials. This not only increases the translational value of research but may also reduce failure rates in future clinical trial stages.195,196 With respect to data accuracy, microfluidic technology improves the imaging and data monitoring capabilities of 3D bioprinting and organoid technologies, enhancing the data readability of cell cultures—an essential factor in ensuring the accuracy of experimental results. Automated experimental procedures further minimize human error, significantly improving experimental reproducibility. Moreover, precise control over key external factors, such as growth factor concentrations and initial geometric configurations, supports consistent cell growth and differentiation, thereby strengthening the uniformity of experimental conditions and the reliability of repeated experiments. Although these methods offer clear advantages in long-term cost-effectiveness, the initial investment in equipment and technology can be substantial, potentially posing financial challenges for some research institutions and laboratories.197,198 Additionally, the integration of these technologies requires higher levels of technical expertise. Effective coordination between platforms, as well as the unification of data analysis workflow for technologies such as microfluidics, organoids, and 3D bioprinting, still requires further research and optimization.199
Dual-, triple-, and prospective 4D cultures— incorporating a temporal dimension alongside microfluidic, organoid, and 3D bioprinting technologies—offer unprecedented opportunities to mimic the developmental processes of human tissues. These integrated classes of technologies aim to accurately reproduce cell fate decisions and tissue morphogenesis during culture construction and can dynamically regulate the culture environment across temporal and spatial dimensions to more closely replicate organ development in vivo. This innovative paradigm holds transformative potential for redefining the frontiers of biomedical research by establishing physiologically relevant platforms for drug efficacy testing, pathomimetic disease modeling, and therapeutic regimen optimization. In doing so, it advances precision medicine initiatives through the development of high-fidelity, clinically translatable model systems.
1.Santos AK, Scalzo S, de Souza RTV, et al. Strategic use of organoids and organs-on-chip as biomimetic tools. Semin Cell Dev Biol. 2023;144:3-10.
doi: 10.1016/j.semcdb.2022.09.010
2.Mooney D, Dankers P, Roche E, Wang H. Engineering active materials for biomedical applications. Adv Mater. 2024;36(43):e2412651.
3.Perner F, Pahl HL, Zeiser R, Heidel FH. Malignant JAK-signaling: at the interface of inflammation and malignant transformation. Leukemia. 2025;39(5):1011-1030.
doi: 10.1038/s41375-025-02569-8
4.Bullman S. The intratumoral microbiota: from microniches to single cells. Cell. 2023;186(8):1532-1534.
doi: 10.1016/j.cell.2023.03.012
5.Luo Q, Shang K, Zhu J, et al. Biomimetic cell culture for cell adhesive propagation for tissue engineering strategies. Mater Horiz. 2023;10(11):4662-4685.
doi: 10.1039/d3mh00849e
6.Cao C, Memete O, Dun Y, et al. Promoting epithelial regeneration in chemically induced acute lung injury through Sox9-positive alveolar type 2 epithelial cells. Stem Cell Res Ther. 2025;16(1):13.
doi: 10.1186/s13287-024-04124-1
7.Unagolla JM, Jayasuriya AC. Recent advances in organoid engineering: a comprehensive review. Appl Mater Today. 2022;29:101582.
doi: 10.1016/j.apmt.2022.101582
8.Zhou Z, Cong L, Cong X. Patient-derived organoids in precision medicine: drug screening, organoid-on-a-chip and living organoid biobank. Front Oncol. 2021;11:762184.
9.Zhao Z, Chen X, Dowbaj AM, et al. Organoids. Nat Rev Methods Primers. 2022;2:94.
doi: 10.1038/s43586-022-00174-y
10.Hofer M, Lutolf MP. Engineering organoids. Nat Rev Mater. 2021;6(5):402-420.
doi: 10.1038/s41578-021-00279-y
11.Ahn Y, An JH, Yang HJ, et al. Human blood vessel organoids penetrate human cerebral organoids and form a vessel-like system. Cells. 2021;10(8):2036.
12.Saorin G, Caligiuri I, Rizzolio F. Microfluidic organoids-on-a-chip: the future of human models. Semin Cell Dev Biol. 2023;144:41-54.
doi: 10.1016/j.semcdb.2022.10.001
13.Ren Y, Yang X, Ma Z, et al. Developments and opportunities for 3D bioprinted organoids. IJB. 2024;7(3):364.
14.Xie M, Zhan Z, Li Y, et al. Functional microfluidics: theory, microfabrication, and applications. Int J Extrem Manuf. 2024;6(3):032005.
15.Koivisto M, Tolvanen TA, Toimela T, et al. Functional human cell-based vascularised cardiac tissue model for biomedical research and testing. Sci Rep. 2022;12(1):13459.
doi: 10.1038/s41598-022-17498-0
16.Sackmann EK, Fulton AL, Beebe DJ. The present and future role of microfluidics in biomedical research. Nature. 2014;507(7491):181-189.
doi: 10.1038/nature13118
17.Nunes JK, Stone HA. Introduction: microfluidics. Chem Rev. 2022;122(7):6919-6920.
doi: 10.1021/acs.chemrev.2c00052
18.Tien J, Dance YW. Microfluidic biomaterials. Adv Healthc Mater. 2021;10(4):e2001028.
19.Huh D, Matthews BD, Mammoto A, Montoya-Zavala M, Hsin HY, Ingber DE. Reconstituting organ-level lung functions on a chip. Science. 2010;328(5986):1662-1668.
20.Abdulla A, Chen S, Chen Z, et al. Three-dimensional microfluidics with dynamic fluidic perturbation promotes viability and uniformity of human cerebral organoids. Biosens Bioelectron. 2023;240:115635.
doi: 10.1016/j.bios.2023.115635
21.Liu W, Song J, Du X, et al. AKR1B10 (Aldo-keto reductase family 1 B10) promotes brain metastasis of lung cancer cells in a multiorgan microfluidic chip model. Acta Biomater. 2019;91:195-208.
doi: 10.1016/j.actbio.2019.04.053
22.Dadgar N, Gonzalez-Suarez AM, Fattahi P, et al. A microfluidic platform for cultivating ovarian cancer spheroids and testing their responses to chemotherapies. Microsyst Nanoeng. 2020;6:93.
doi: 10.1038/s41378-020-00201-6
23.Tan J, Sun X, Zhang J, et al. Exploratory evaluation of EGFR-targeted anti-tumor drugs for lung cancer based on lung-ona-chip. Biosensors (Basel). 2022;12(8):618.
doi: 10.3390/bios12080618
24.Besanjideh M, Shamloo A, Hannani SK. Evaluating the reliability of tumour spheroid-on-chip models for replicating intratumoural drug delivery: considering the role of microfluidic parameters. J Drug Target. 2023;31(2):179-193.
doi: 10.1080/1061186X.2022.2119478
25.Valverde MG, Mille LS, Figler KP, et al. Biomimetic models of the glomerulus. Nat Rev Nephrol. 2022;18(4):241-257.
doi: 10.1038/s41581-021-00528-x
26.Klak M, Bryniarski T, Kowalska P, et al. Novel strategies in artificial organ development: what is the future of medicine? Micromachines (Basel). 2020;11(7):646.
doi: 10.3390/mi11070646
27.Chliara MA, Elezoglou S, Zergioti I. Bioprinting on organon-chip: development and applications. Biosensors (Basel). 2022;12(12):1135.
doi: 10.3390/bios12121135
28.Clevers H. Modeling development and disease with organoids. Cell. 2016;165(7):1586-1597.
doi: 10.1016/j.cell.2016.05.082
29.Wang H, Ning X, Zhao F, Zhao H, Li D. Human organoids-on-chips for biomedical research and applications. Theranostics. 2024;14(2):788-818.
doi: 10.7150/thno.90492
30.Kim J, Koo BK, Knoblich JA. Human organoids: model systems for human biology and medicine. Nat Rev Mol Cell Biol. 2020;21(10):571-584.
doi: 10.1038/s41580-020-0259-3
31.Kim H, Perlingeiro RCR. Generation of human myogenic progenitors from pluripotent stem cells for in vivo regeneration. Cell Mol Life Sci. 2022;79(8):406.
doi: 10.1007/s00018-022-04434-8
32.Sen D, Voulgaropoulos A, Keung AJ. Effects of early geometric confinement on the transcriptomic profile of human cerebral organoids. BMC Biotechnol. 2021; 21(1):59.
doi: 10.1186/s12896-021-00718-2
33.Liberali P, Schier AF. The evolution of developmental biology through conceptual and technological revolutions. Cell. 2024;187(14):3461-3495.
doi: 10.1016/j.cell.2024.05.053
34.Kim E, Choi S, Kang B, et al. Creation of bladder assembloids mimicking tissue regeneration and cancer. Nature. 2020; 588(7839):664-669.
doi: 10.1038/s41586-020-3034-x
35.Sheridan MA, Fernando RC, Gardner L, et al. Establishment and differentiation of long-term trophoblast organoid cultures from the human placenta. Nat Protoc. 2020;15(10):3441-3463.
doi: 10.1038/s41596-020-0381-x
36.Minoli M, Cantore T, Hanhart D, et al. Bladder cancer organoids as a functional system to model different disease stages and therapy response. Nat Commun. 2023; 14(1):2214.
doi: 10.1038/s41467-023-37696-2
37.Gras-Peña R, Danzl NM, Khosravi-Maharlooei M, et al. Human stem cell-derived thymic epithelial cells enhance human T-cell development in a xenogeneic thymus. J Allergy Clin Immunol. 2022;149(5):1755-1771.
doi: 10.1016/j.jaci.2021.09.038
38.Corrò C, Novellasdemunt L, Li VSW. A brief history of organoids. Am J Physiol Cell Physiol. 2020;319(1):C151-C165.
doi: 10.1152/ajpcell.00120.2020
39.Gjorevski N, Nikolaev M, Brown TE, et al. Tissue geometry drives deterministic organoid patterning. Science. 2022;375(6576):eaaw9021.
40.Kim W, Lee Y, Kang D, Kwak T, Lee HR, Jung S. 3D inkjet-bioprinted lung-on-a-chip. ACS Biomater Sci Eng. 2023;9(5):2806-2815.
doi: 10.1021/acsbiomaterials.3c00089
41.Lee J, Park D, Seo Y, Chung JJ, Jung Y, Kim SH. Organlevel functional 3D tissue constructs with complex compartments and their preclinical applications. Adv Mater. 2020;32(51):e2002096.
42.Zhao X, Xu Z, Xiao L, et al. Review on the Vascularization of Organoids and Organoids-on-a-Chip. Front Bioeng Biotechnol. 2021;9:637048.
doi: 10.3389/fbioe.2021.637048
43.Yuan L, Xie S, Bai H, et al. Reconstruction of dynamic mammary mini gland in vitro for normal physiology and oncogenesis. Nat Methods. 2023;20(12):2021-2033.
doi: 10.1038/s41592-023-02039-y
44.Jiang S, Zhao H, Zhang W, et al. An automated organoid platform with inter-organoid homogeneity and inter-patient heterogeneity. Cell Rep Med. 2020;1(9):100161.
doi: 10.1016/j.xcrm.2020.100161
45.Zhu L, Yuhan J, Yu H, Zhang B, Huang K, Zhu L. Decellularized extracellular matrix for remodeling bioengineering organoid’s microenvironment. Small. 2023;19(25):e2207752.
46.Wörsdörfer P, Ergün S. The impact of oxygen availability and multilineage communication on organoid maturation. Antioxid Redox Signal. 2021;35(3):217-233.
47.LeSavage BL, Suhar RA, Broguiere N, Lutolf MP, Heilshorn SC. Next-generation cancer organoids. Nat Mater. 2022;21(2):143-159.
doi: 10.1038/s41563-021-01057-5
48.Dominijanni AJ, Devarasetty M, Forsythe SD, Votanopoulos KI, Soker S. Cell viability assays in three-dimensional hydrogels: a comparative study of accuracy. Tissue Eng Part C Methods. 2021;27(7):401-410.
doi: 10.1089/ten.TEC.2021.0060
49.de Medeiros G, Ortiz R, Strnad P, et al. Multiscale lightsheet organoid imaging framework. Nat Commun. 2022;13(1):4864.
doi: 10.1038/s41467-022-32465-z
50.Zhang Y, Li G, Wang J, Zhou F, Ren X, Su J. Small joint organoids 3D bioprinting: construction strategy and application. Small. 2024;20(8):e2302506.
51.Daly AC, Prendergast ME, Hughes AJ, Burdick JA. Bioprinting for the biologist. Cell. 2021;184(1):18-32.
doi: 10.1016/j.cell.2020.12.002
52.Wang Z, Wang L, Li T, et al. 3D bioprinting in cardiac tissue engineering. Theranostics. 2021;11(16):7948-7969.
doi: 10.7150/thno.61621
53.Matai I, Kaur G, Seyedsalehi A, McClinton A, Laurencin CT. Progress in 3D bioprinting technology for tissue/organ regenerative engineering. Biomaterials. 2020;226:119536.
doi: 10.1016/j.biomaterials.2019.119536
54.Bosmans C, Ginés Rodriguez N, Karperien M, et al. Towards single-cell bioprinting: micropatterning tools for organ-on-chip development. Trends Biotechnol. 2024;42(6):739-759.
doi: 10.1016/j.tibtech.2023.11.014
55.Chen XB, Fazel Anvari-Yazdi A, Duan X, et al. Biomaterials / bioinks and extrusion bioprinting. Bioact Mater. 2023;28:511-536.
doi: 10.1016/j.bioactmat.2023.06.006
56.Patrício SG, Sousa LR, Correia TR, et al. Freeform 3D printing using a continuous viscoelastic supporting matrix. Biofabrication. 2020;12(3):035017.
57.Schwab A, Levato R, D’Este M, Piluso S, Eglin D, Malda J. Printability and shape fidelity of bioinks in 3D bioprinting. Chem Rev. 2020;120(19):11028-11055.
doi: 10.1021/acs.chemrev.0c00084
58.Lee H, Kim J, Choi Y, Cho DW. Application of gelatin bioinks and cell-printing technology to enhance cell delivery capability for 3D liver fibrosis-on-a-chip development. ACS Biomater Sci Eng. 2020;6(4):2469-2477.
doi: 10.1021/acsbiomaterials.9b01735
59.Rukavina P, Koch F, Wehrle M, et al. In vivo evaluation of bioprinted prevascularized bone tissue. Biotechnol Bioeng. 2020;117(12):3902-3911.
doi: 10.1002/bit.27527
60.Yvanoff C, Willaert RG. Development of bone cell microarrays in microfluidic chips for studying osteocyte-osteoblast communication under fluid flow mechanical loading. Biofabrication. 2022;14(2):025014.
61.Carou-Senra P, Rodríguez-Pombo L, Awad A, Basit AW, Alvarez-Lorenzo C, Goyanes A. Inkjet printing of pharmaceuticals. Adv Mater. 2024;36(11):2309164.
62.Li X, Liu B, Pei B, et al. Inkjet bioprinting of biomaterials. Chem Rev. 2020;120(19):10793-10833.
doi: 10.1021/acs.chemrev.0c00008
63.Castilho M, Levato R, Bernal PN, et al. Hydrogelbased bioinks for cell electrowriting of well-organized living structures with micrometer-scale resolution. Biomacromolecules. 2021;22(2):855-866.
doi: 10.1021/acs.biomac.0c01577
64.Hong H, Seo YB, Kim DY, et al. Digital light processing 3D printed silk fibroin hydrogel for cartilage tissue engineering. Biomaterials. 2020;232:119679.
doi: 10.1016/j.biomaterials.2019.119679
65.Falandt M, Bernal PN, Dudaryeva O, et al. Spatial‐ selective volumetric 4D printing and single‐photon grafting of biomolecules within centimeter‐scale hydrogels via tomographic manufacturing. Adv Mater Technol. 2023;8(15):2300026.
66.Größbacher G, Bartolf-Kopp M, Gergely C, et al. Volumetric printing across melt electrowritten scaffolds fabricates multi-material living constructs with tunable architecture and mechanics. Adv Mater. 2023;35(32):e2300756.
67.Maibohm C, Silvestre OF, Borme J, Sinou M, Heggarty K, Nieder JB. Multi-beam two-photon polymerization for fast large area 3D periodic structure fabrication for bioapplications. Sci Rep. 2020;10(1):8740.
doi: 10.1038/s41598-020-64955-9
68.Moldovan D, Choi J, Choo Y, Kim WS, Hwa Y. Laserbased three-dimensional manufacturing technologies for rechargeable batteries. Nano Converg. 2021;8(1):23.
doi: 10.1186/s40580-021-00271-w
69.Zhang J, Wehrle E, Rubert M, Müller R. 3D bioprinting of human tissues: biofabrication, bioinks, and bioreactors. Int J Mol Sci. 2021;22(8):3971.
doi: 10.3390/ijms22083971
70.Ning L, Gil CJ, Hwang B, et al. Biomechanical factors in three-dimensional tissue bioprinting. Appl Phys Rev. 2020;7(4):041319.
doi: 10.1063/5.0023206
71.Mohabatpour F, Duan X, Yazdanpanah Z, et al. Bioprinting of alginate-carboxymethyl chitosan scaffolds for enamel tissue engineeringin vitro. Biofabrication. 2022;15(1): 015022.
doi: 10.1088/1758-5090/acab35015022
72.Sadeghianmaryan A, Naghieh S, Alizadeh Sardroud H, et al. Extrusion-based printing of chitosan scaffolds and their in vitro characterization for cartilage tissue engineering. Int J Biol Macromol. 2020;164:3179-3192.
doi: 10.1016/j.ijbiomac.2020.08.180
73.Kołodziejska M, Jankowska K, Klak M, Wszoła M. Chitosan as an underrated polymer in modern tissue engineering. Nanomaterials. 2021;11(11):3019.
doi: 10.3390/nano11113019
74.Abu-Jdayil B, Ghannam M, Alsayyed Ahmed K, Djama M. The effect of biopolymer chitosan on the rheology and stability of na-bentonite drilling mud. Polymers (Basel). 2021;13(19):3361.
75.Al-Hajri S, Negash BM, Rahman MM, Haroun M, Al-Shami TM. Effect of silica nanoparticles on polymer adsorption reduction on marcellus shale. ACS Omega. 2021;6(44):29537-29546.
76.Li Q, Li Y, Jin Z, Li Y, Chen Y, Zhou J. Viscoelasticity and solution stability of cyanoethylcellulose with different molecular weights in aqueous solution. Molecules. 2021;26(11):3201.
doi: 10.3390/molecules26113201
77.Li C, Zheng Z, Jia J, et al. Preparation and characterization of photocurable composite extracellular matrix-methacrylated hyaluronic acid bioink. J Mater Chem B. 2022;10(22):4242-4253.
doi: 10.1039/d2tb00548d
78.Zimmerling A, Yazdanpanah Z, Cooper DML, Johnston JD, Chen X. 3D printing PCL/nHA bone scaffolds: exploring the influence of material synthesis techniques. Biomater Res. 2021;25(1):3.
doi: 10.1186/s40824-021-00204-y
79.Pfau MR, Beltran FO, Woodard LN, et al. Evaluation of a self-fitting, shape memory polymer scaffold in a rabbit calvarial defect model. Acta Biomater. 2021;136: 233-242.
doi: 10.1016/j.actbio.2021.09.041
80.Fang Z, Xiao Y, Geng X, et al. Fabrication of heparinized small diameter TPU/PCL bi-layered artificial blood vessels and in vivo assessment in a rabbit carotid artery replacement model. Biomater Adv. 2022;133:112628.
doi: 10.1016/j.msec.2021.112628
81.Yang FT, Chen YM, Rwei SP. Influence of cross-linking and crystalline morphology on the shape-memory properties of PET/PEN/PCL copolyesters using trimesic acid and glycerol. Polymers (Basel). 2023;15(9):2082.
82.Liu X, Song S, Huang J, et al. HBC-nanofiber hydrogel scaffolds with 3D printed internal microchannels for enhanced cartilage differentiation. J Mater Chem B. 2020;8(28):6115-6127.
doi: 10.1039/d0tb00616e
83.Liang DS, Wen ZJ, Wang JH, et al. Legumain protease-sheddable PEGylated, tuftsin-modified nanoparticles for selective targeting to tumour-associated macrophages. J Drug Target. 2022;30(1):82-93.
doi: 10.1080/1061186X.2021.1906886
84.Xu X, Wang Z, Li M, et al. Reconstructed hierarchically structured keratin fibers with shape-memory features based on reversible secondary-structure transformation. Adv Mater. 2023;35(41):e2304725.
85.Yarali E, Mirzaali MJ, Ghalayaniesfahani A, Accardo A, Diaz-Payno PJ, Zadpoor AA. 4D printing for biomedical applications. Adv Mater. 2024;36(31):e2402301.
86.Leanza S, Wu S, Sun X, Qi HJ, Zhao RR. Active materials for functional origami. Adv Mater. 2024;36(9):e2302066.
87.McCracken JM, Donovan BR, White TJ. Materials as machines. Adv Mater. 2020;32(20):e1906564.
88.Wang Z, Xiang L, Lin F, Tang Y, Cui W. 3D bioprinting of emulating homeostasis regulation for regenerative medicine applications. J Control Release. 2023;353:147-165.
doi: 10.1016/j.jconrel.2022.11.035
89.Juraski AC, Sharma S, Sparanese S, et al. 3D bioprinting for organ and organoid models and disease modeling. Expert Opin Drug Discov. 2023;18(9):1043-1059.
doi: 10.1080/17460441.2023.2234280
90.Roth EA, Xu T, Das M, Gregory C, Hickman JJ, Boland T. Inkjet printing for high-throughput cell patterning. Biomaterials. 2004;25(17):3707-3715.
doi: 10.1016/j.biomaterials.2003.10.052
91.Hafa L, Breideband L, Ramirez Posada L, et al. Light sheet-based laser patterning bioprinting produces longterm viable full-thickness skin constructs. Adv Mater. 2024;36(8):e2306258.
92.Sun H, Sun L, Ke X, et al. Prediction of clinical precision chemotherapy by patient-derived 3D bioprinting models of colorectal cancer and its liver metastases. Adv Sci (Weinh). 2024;11(2):e2304460.
93.Herzog J, Franke L, Lai Y, Gomez Rossi P, Sachtleben J, Weuster-Botz D. 3D bioprinting of microorganisms: principles and applications. Bioprocess Biosyst Eng. 2024;47(4):443-461.
doi: 10.1007/s00449-023-02965-3
94.Ansaf RB, Ziebart R, Gudapati H, et al. 3D bioprinting-a model for skin aging. Regen Biomater. 2023;10:rbad060.
doi: 10.1093/rb/rbad060
95.Homan KA, Gupta N, Kroll KT, et al. Flow-enhanced vascularization and maturation of kidney organoids in vitro. Nat Methods. 2019;16(3):255-262.
doi: 10.1038/s41592-019-0325-y
96.Lee KK, McCauley HA, Broda TR, Kofron MJ, Wells JM, Hong CI. Human stomach-on-a-chip with luminal flow and peristaltic-like motility. Lab Chip. 2018;18(20):3079-3085.
doi: 10.1039/C8LC00910D
97.Fang G, Lu H, Al-Nakashli R, et al. Enabling peristalsis of human colon tumor organoids on microfluidic chips. Biofabrication. 2021;14(1):015006.
98.Quintard C, Tubbs E, Jonsson G, et al. A microfluidic platform integrating functional vascularized organoids-on-chip. Nat Commun. 2024;15(1):1452.
doi: 10.1038/s41467-024-45710-4
99.Lim J, Ching H, Yoon JK, Jeon NL, Kim Y. Microvascularized tumor organoids-on-chips: advancing preclinical drug screening with pathophysiological relevance. Nano Converg. 2021;8(1):12.
doi: 10.1186/s40580-021-00261-y
100.Song HHG, Lammers A, Sundaram S, et al. Transient support from fibroblasts is sufficient to drive functional vascularization in engineered tissues. Adv Funct Mater. 2020;30(48):2003777.
101.Shin N, Kim Y, Ko J, et al. Vascularization of iNSC spheroid in a 3D spheroid-on-a-chip platform enhances neural maturation. Biotechnol Bioeng. 2022;119(2):566-574.
doi: 10.1002/bit.27978
102.Nashimoto Y, Hayashi T, Kunita I, et al. Integrating perfusable vascular networks with a three-dimensional tissue in a microfluidic device. Integr Biol (Camb). 2017;9(6):506-518.
doi: 10.1039/c7ib00024c
103.Glaser DE, Curtis MB, Sariano PA, et al. Organ-on-a-chip model of vascularized human bone marrow niches. Biomaterials. 2022;280:121245.
doi: 10.1016/j.biomaterials.2021.121245
104.Shaji M, Tamada A, Fujimoto K, Muguruma K, Karsten SL, Yokokawa R. Deciphering potential vascularization factors of on-chip co-cultured hiPSC-derived cerebral organoids. Lab Chip. 2024;24(4):680-696.
doi: 10.1039/d3lc00930k
105.Huang YH, Watanabe M, Yamashita T, Sudo R. Construction of highly vascularized hepatic spheroids of primary hepatocytes via pro-angiogenic strategy in vitro. Biofabrication. 2025;17(3):035001.
106.Tronolone JJ, Mathur T, Chaftari CP, Jain A. A machine-learned microvasculature optimizes physiological insulin secretion in a vascularized pancreas-chip. FASEB J. 2022;36(S1):6103.
doi: 10.1096/fasebj.2022.36.S1.R6103
107.Weng KC, Kurokawa YK, Hajek BS, Paladin JA, Shirure VS, George SC. Human induced pluripotent stem-cardiac-endothelial-tumor-on-a-chip to assess anticancer efficacy and cardiotoxicity. Tissue Eng Part C Methods. 2020;26(1):44-55.
doi: 10.1089/ten.TEC.2019.0248
108.Seiler KM, Bajinting A, Alvarado DM, et al. Patient-derived small intestinal myofibroblasts direct perfused, physiologically responsive capillary development in a microfluidic Gut-on-a-Chip model. Sci Rep. 2020; 10(1):3842.
doi: 10.1038/s41598-020-60672-5
109.Pinho D, Santos D, Vila A, Carvalho S. Establishment of colorectal cancer organoids in microfluidic-based system. Micromachines (Basel). 2021;12(5):497.
doi: 10.3390/mi12050497
110.Rizzuti M, Melzi V, Brambilla L, et al. Shaping the neurovascular unit exploiting human brain organoids. Mol Neurobiol. 2024;61(9):6642-6657.
doi: 10.1007/s12035-024-03998-9
111.Gheibi P, Zeng S, Son KJ, et al. Microchamber cultures of bladder cancer: a platform for characterizing drug responsiveness and resistance in PDX and primary cancer cells. Sci Rep. 2017;7(1):12277.
doi: 10.1038/s41598-017-12543-9
112.Huang Y, Liu T, Huang Q, Wang Y. From organ-on-a-chip to human-on-a-chip: a review of research progress and latest applications. ACS Sens. 2024;9(7):3466-3488.
doi: 10.1021/acssensors.4c00004
113.Schneider O, Moruzzi A, Fuchs S, et al. Fusing spheroids to aligned μ-tissues in a Heart-on-Chip featuring oxygen sensing and electrical pacing capabilities. Mater Today Bio. 2022:15:100280.
doi: 10.1101/2022.02.26.482011
114.Yang C, Wu W, Yang 1†, et al. Controlled preparation of droplets for cell encapsulation by air-focused microfluidic bioprinting. Int J Bioprint. 2024;10(1):1102.
doi: 10.36922/ijb.1102
115.Chen L, Xiao Y, Wu Q, et al. Emulsion designer using microfluidic three-dimensional droplet printing in droplet. Small. 2021;17(39):e2102579.
116.Ouyang L, Armstrong JPK, Chen Q, Lin Y, Stevens MM. Void-free 3D bioprinting for in-situ endothelialization and microfluidic perfusion. Adv Funct Mater. 2020;30(26):1909009.
117.Dornhof J, Zieger V, Kieninger J, et al. Bioprinting-based automated deposition of single cancer cell spheroids into oxygen sensor microelectrode wells. Lab Chip. 2022;22(22):4369-4381.
doi: 10.1039/d2lc00705c
118.Zhang YS, Arneri A, Bersini S, et al. Bioprinting 3D microfibrous scaffolds for engineering endothelialized myocardium and heart-on-a-chip. Biomaterials. 2016;110:45-59.
doi: 10.1016/j.biomaterials.2016.09.003
119.Silvani G, Basirun C, Wu H, et al. A 3D-bioprinted vascularized glioblastoma-on-a-chip for studying the impact of simulated microgravity as a novel pre-clinical approach in brain tumor therapy. Adv Ther. 2021;4(11): 2100106.
120.Fritschen A, Lindner N, Scholpp S, et al. High-scale 3D-bioprinting platform for the automated production of vascularized organs-on-a-chip. Adv Healthc Mater. 2024;13(17):e2304028.
121.Singh NK, Han W, Nam SA, et al. Three-dimensional cell-printing of advanced renal tubular tissue analogue. Biomaterials. 2020;232:119734.
doi: 10.1016/j.biomaterials.2019.119734
122.Serex L, Sharma K, Rizov V, Bertsch A, McKinney JD, Renaud P. Microfluidic-assisted bioprinting of tissues and organoids at high cell concentrations. Biofabrication. 2021;13(2):025006.
123.Highley CB, Song KH, Daly AC, Burdick JA. Jammed microgel inks for 3D printing applications. Adv Sci (Weinh). 2019;6(1):1801076.
124.Maharjan S, Ma C, Singh B, et al. Advanced 3D imaging and organoid bioprinting for biomedical research and therapeutic applications. Adv Drug Deliv Rev. 2024;208:115237.
doi: 10.1016/j.addr.2024.115237
125.Hu Y, Zhu T, Cui H, Cui H. Integrating 3D bioprinting and organoids to better recapitulate the complexity of cellular microenvironments for tissue engineering. Adv Healthc Mater. 2025;14(3):e2403762.
126.Han Y, King M, Tikhomirov E, et al. Towards 3D bioprinted spinal cord organoids. Int J Mol Sci. 2022;23(10):5788.
doi: 10.3390/ijms23105788
127.Lawlor KT, Vanslambrouck JM, Higgins JW, et al. Cellular extrusion bioprinting improves kidney organoid reproducibility and conformation. Nat Mater. 2021;20(2):260-271.
doi: 10.1038/s41563-020-00853-9
128.Zhang T, Sheng S, Cai W, et al. 3-D bioprinted human-derived skin organoids accelerate full-thickness skin defects repair. Bioact Mater. 2024;42:257-269.
doi: 10.1016/j.bioactmat.2024.08.036
129.Zhang Y, Li D, Liu Y, et al. 3D-bioprinted anisotropic bicellular living hydrogels boost osteochondral regeneration via reconstruction of cartilage–bone interface. Innovation (Camb). 2024;5(1):100542.
doi: 10.1016/j.xinn.2023.100542
130.Sorkio A, Koch L, Koivusalo L, et al. Human stem cell based corneal tissue mimicking structures using laser-assisted 3D bioprinting and functional bioinks. Biomaterials. 2018;171:57-71.
doi: 10.1016/j.biomaterials.2018.04.034
131.Brassard JA, Nikolaev M, Hübscher T, Hofer M, Lutolf MP. Recapitulating macro-scale tissue self-organization through organoid bioprinting. Nat Mater. 2021;20(1):22-29.
doi: 10.1038/s41563-020-00803-5
132.Rezakhani S, Gjorevski N, Lutolf MP. Extracellular matrix requirements for gastrointestinal organoid cultures. Biomaterials. 2021;276:121020.
doi: 10.1016/j.biomaterials.2021.121020
133.Clark CC, Yoo KM, Sivakumar H, et al. Immersion bioprinting of hyaluronan and collagen bioink-supported 3D patient-derived brain tumor organoids. Biomed Mater. 2022;18(1):015014.
134.Wen K, Gorbushina AA, Schwibbert K, Bell J. Microfluidic platform with precisely controlled hydrodynamic parameters and integrated features for generation of microvortices to accurately form and monitor biofilms in flow. ACS Biomater Sci Eng. 2024;10(7):4626-4634.
doi: 10.1021/acsbiomaterials.4c00101
135.Hu T, Cui X, Zhu M, et al. 3D-printable supramolecular hydrogels with shear-thinning property: fabricating strength tunable bioink via dual crosslinking. Bioact Mater. 2020;5(4):808-818.
doi: 10.1016/j.bioactmat.2020.06.001
136.Luo Z, Zhang H, Chen R, et al. Digital light processing 3D printing for microfluidic chips with enhanced resolution via dosing- and zoning-controlled vat photopolymerization. Microsyst Nanoeng. 2023;9:103.
doi: 10.1038/s41378-023-00542-y
137.Carvalho MR, Caballero D, Kundu SC, Reis RL, Oliveira JM. Biomimetic and soft lab-on-a-chip platform based on enzymatic-crosslinked silk fibroin hydrogel for 3D cell coculture. Biomed Mater. 2024;19(6):065032.
138.Reineke B, Paulus I, Löffelsend S, et al. On-chip fabrication and in-flow 3D-printing of microgel constructs: from chip to scaffold materials in one integral process. Biofabrication. 2024;16(2):025038.
139.Fournié V, Venzac B, Trevisiol E, et al. A microfluidics-assisted photopolymerization method for high-resolution multimaterial 3D printing. Addit Manuf. 2023;72:103629.
doi: 10.1016/j.addma.2023.103629
140.Böcherer D, Li Y, Rein C, Franco Corredor S, Hou P, Helmer D. High-resolution 3D printing of dual-curing thiol-ene/ epoxy system for fabrication of microfluidic devices for bioassays. Adv Funct Mater. 2024;34(29):2401516.
141.Liu P, Fu L, Song Z, et al. Three dimensionally printed nitrocellulose-based microfluidic platform for investigating the effect of oxygen gradient on cells. Analyst. 2021;146(17):5255-5263.
doi: 10.1039/d1an00927c
142.Cantoni F, Barbe L, Pohlit H, Tenje M. A perfusable multihydrogel vasculature on-chip engineered by 2-photon 3D printing and scaffold molding to improve microfabrication fidelity in hydrogels. Adv Mater Technol. 2024;9(4):2300718.
143.Khan I, Prabhakar A, Delepine C, Tsang H, Pham V, Sur M. A low-cost 3D printed microfluidic bioreactor and imaging chamber for live-organoid imaging. Biomicrofluidics. 2021;15(2):024105.
doi: 10.1063/5.0041027
144.Fiorotto R, Mariotti V, Taleb SA, et al. Cell-matrix interactions control biliary organoid polarity, architecture, and differentiation. Hepatol Commun. 2023;7(4):e0094.
doi: 10.1097/HC9.0000000000000094
145.Becker ML, Burdick JA. Introduction: polymeric biomaterials. Chem Rev. 2021;121(18):10789-10791.
doi: 10.1021/acs.chemrev.1c00354
146.Yang X, Jiang T, Liu L, et al. Observing single cells in whole organs with optical imaging. J Innov Opt Health Sci. 2023;16(01):2330002.
doi: 10.1142/S1793545823300021
147.Velicky P, Miguel E, Michalska JM, et al. Dense 4D nanoscale reconstruction of living brain tissue. Nat Methods. 2023;20(8):1256-1265
doi: 10.1016/j.compositesa.2021.106799
148.Bernal PN, Bouwmeester M, Madrid-Wolff J, et al. Volumetric bioprinting of organoids and optically tuned hydrogels to build liver-like metabolic biofactories. Adv Mater. 2022;34(15):e2110054.
149.Yu K, Zhang X, Sun Y, et al. Printability during projectionbased 3D bioprinting. Bioact Mater. 2022;11:254-267.
doi: 10.1016/j.bioactmat.2021.09.021
150.Budi NYP, Lai WY, Huang YH, Ho HN. 3D organoid cultivation improves the maturation and functional differentiation of cholangiocytes from human pluripotent stem cells. Front Cell Dev Biol. 2024;12:1361084.
doi: 10.3389/fcell.2024.1361084
151.Rnjak-Kovacina J, Choi YS, Lim KS. Biofabrication applications. Adv Healthc Mater. 2022;11(24):e2202934.
152.Wang Y, Kaplan D. Special issue: leaders in biomedical engineering. ACS Biomater Sci Eng. 2020;6(5): 2495-2497.
doi: 10.1021/acsbiomaterials.0c00606
153.Cabral M, Cheng K, Zhu D. Three-dimensional bioprinting of organoids: past, present, and prospective. Tissue Eng Part A. 2024;30(11-12):314-321.
doi: 10.1089/ten.TEA.2023.0209
154.Weltin A, Hammer S, Noor F, Kaminski Y, Kieninger J, Urban GA. Accessing 3D microtissue metabolism: lactate and oxygen monitoring in hepatocyte spheroids. Biosens Bioelectron. 2017;87:941-948.
doi: 10.1016/j.bios.2016.07.094
155.Esene JE, Burningham AJ, Tahir A, Nordin GP, Woolley AT. 3D printed microfluidic devices for integrated solidphase extraction and microchip electrophoresis of preterm birth biomarkers. Anal Chim Acta. 2024;1296: 342338.
doi: 10.1016/j.aca.2024.342338
156.Kluwe F, Michelet R, Mueller-Schoell A, et al. Perspectives on model-informed precision dosing in the digital health era: challenges, opportunities, and recommendations. Clin Pharmacol Ther. 2021;109(1):29-36.
doi: 10.1002/cpt.2049
157.Murphy JF, Lavelle M, Asciak L, et al. Biofabrication and biomanufacturing in Ireland and the UK. Biodes Manuf. 2024;7(6):825-856.
doi: 10.1007/s42242-024-00316-z
158.Rahimifard M, Bagheri Z, Hadjighassem M, et al. Investigation of anti-cancer effects of new pyrazino[1,2-a] benzimidazole derivatives on human glioblastoma cells through 2D in vitro model and 3D-printed microfluidic device. Life Sci. 2022;302:120505.
doi: 10.1016/j.lfs.2022.120505
159.Herland A, Maoz BM, Das D, et al. Quantitative prediction of human pharmacokinetic responses to drugs via fluidically coupled vascularized organ chips. Nat Biomed Eng. 2020;4(4):421-436.
doi: 10.1038/s41551-019-0498-9
160.Vurat MT, Şeker Ş, Lalegül-Ülker Ö, Parmaksiz M, Elçin AE, Elçin YM. Development of a multicellular 3D-bioprinted microtissue model of human periodontal ligamentalveolar bone biointerface: towards a pre-clinical model of periodontal diseases and personalized periodontal tissue engineering. Genes Dis. 2022;9(4):1008-1023.
doi: 10.1016/j.gendis.2020.11.011
161.Tebon PJ, Wang B, Markowitz AL, et al. Drug screening at single-organoid resolution via bioprinting and interferometry. Nat Commun. 2023;14(1):3168.
doi: 10.1038/s41467-023-38832-8
162.Miao T, Chen K, Wei X, et al. High-throughput fabrication of cell spheroids with 3D acoustic assembly devices. Int J Bioprint. 2023;9(4):733.
doi: 10.18063/ijb.733
163.Cui K, Wang Y, Zhu Y, et al. Neurodevelopmental impairment induced by prenatal valproic acid exposure shown with the human cortical organoid-on-a-chip model. Microsyst Nanoeng. 2020;6(1):49.
doi: 10.1038/s41378-020-0165-z
164.Yin Y, Vázquez-Rosado EJ, Wu D, et al. Microfluidic coaxial 3D bioprinting of cell-laden microfibers and microtubes for salivary gland tissue engineering. Biomater Adv. 2023;154:213588.
doi: 10.1016/j.bioadv.2023.213588
165.Choi YM, Lee H, Ann M, Song M, Rheey J, Jang J. 3D bioprinted vascularized lung cancer organoid models with underlying disease capable of more precise drug evaluation. Biofabrication. 2023;15(3):034104.
166.Huang J, Xu Z, Jiao J, et al. Microfluidic intestinal organoid-on-a-chip uncovers therapeutic targets by recapitulating oxygen dynamics of intestinal IR injury. Bioact Mater. 2023;30:1-14.
doi: 10.1016/j.bioactmat.2023.07.001
167.Zou Z, Lin Z, Wu C, et al. Micro‐engineered organoid‐ on‐a‐chip based on mesenchymal stromal cells to predict immunotherapy responses of HCC patients. Adv Sci. 2023;10(27):2302640.
168.Flores-Torres S, Dimitriou NM, Pardo LA, et al. Bioprinted multicomponent hydrogel co-culture tumor-immune model for assessing and simulating tumor-infiltrated lymphocyte migration and functional activation. ACS Appl Mater Interfaces. 2023;15(28):33250-33262.
169.Alrashoudi AA, Albalawi HI, Aldoukhi AH, et al. Fabrication of a lateral flow assay for rapid in-field detection of covid-19 antibodies using additive manufacturing printing technologies. IJB. 2024;7(4):399.
170.Ching T, Vasudevan J, Chang SY, et al. Biomimetic vasculatures by 3D-printed porous molds. Small. 2022;18(39):e2203426.
171.Nishijima T, Okuyama K, Shibata S, et al. Novel artificial nerve transplantation of human iPSC-derived neurite bundles enhanced nerve regeneration after peripheral nerve injury. Inflamm Regener. 2024;44(1):6.
doi: 10.1186/s41232-024-00319-4
172.Xie C, Liang R, Ye J, et al. High-efficient engineering of osteo-callus organoids for rapid bone regeneration within one month. Biomaterials. 2022;288:121741.
doi: 10.1016/j.biomaterials.2022.121741
173.Lu Z, Miao X, Song Q, et al. Detection of lineagereprogramming efficiency of tumor cells in a 3D-printed liver-on-a-chip model. Theranostics. 2023;13(14):4905-4918.
doi: 10.7150/thno.86921
174.Bues J, Biočanin M, Pezoldt J, et al. Deterministic scRNA-seq captures variation in intestinal crypt and organoid composition. Nat Methods. 2022;19(3):323-330.
doi: 10.1038/s41592-021-01391-1
175.Qazi TH, Blatchley MR, Davidson MD, et al. Programming hydrogels to probe spatiotemporal cell biology. Cell Stem Cell. 2022;29(5):678-691.
doi: 10.1016/j.stem.2022.03.013
176.Daly AC, Davidson MD, Burdick JA. 3D bioprinting of high cell-density heterogeneous tissue models through spheroid fusion within self-healing hydrogels. Nat Commun. 2021;12(1):753.
doi: 10.1038/s41467-021-21029-2
177.Javanmardi E, Maresova P, Xie N, Mierzwiak R. Exploring business models for managing uncertainty in healthcare, medical devices, and biotechnology industries. Heliyon. 2024;10(4):e25962.
doi: 10.1016/j.heliyon.2024.e25962
178.Zushin PJH, Mukherjee S, Wu JC. FDA Modernization Act 2.0: transitioning beyond animal models with human cells, organoids, and AI/ML-based approaches. J Clin Invest. 2023;133(21):e175824.
doi: 10.1172/JCI175824
179.Shishkina LN, Mazurkov OY, Bormotov NI, et al. Safety and pharmacokinetics of the substance of the anti-smallpox drug NIOCH-14 after oral administration to laboratory animals. Viruses. 2023;15(1):205.
doi: 10.3390/v15010205
180.Shang L, Zhao Y, Cui W. Regenerative medicine entering a new era. Small. 2022;18(36):e2204625.
181.Boccardi M, Gold M, Mahant V, Marincola FM, Gunn A. Why should academia care about the Target Product Profile? J Transl Med. 2024;22(1):716.
doi: 10.1186/s12967-024-05520-0
182.Fu J, Gao Q, Li S. Application of intelligent medical sensing technology. Biosensors. 2023;13(8):812.
doi: 10.3390/bios13080812
183.Lee ED, Aurand ER, Friedman DC, Engineering Biology Research Consortium Microbiomes Roadmapping Working Group. Engineering microbiomes-looking ahead. ACS Synth Biol. 2020;9(12):3181-3183.
doi: 10.1021/acssynbio.0c00558
184.Xing Y, Zhang MS, Xiao JH, Liu RM. Galangin induces the osteogenic differentiation of human amniotic mesenchymal stem cells via the JAK2/STAT3 signaling pathway. Eur J Pharmacol. 2022;935:175326.
doi: 10.1016/j.ejphar.2022.175326
185.Cai Z, Zhu M, Xu L, et al. Directed differentiation of human induced pluripotent stem cells to heart valve cells. Circulation. 2024;149(18):1435-1456.
doi: 10.1161/CIRCULATIONAHA.123.065143
186.Wang Y, Liu X, Xie B, Yuan H, Zhang Y, Zhu J. The NOTCH1-dependent HIF1α/VGLL4/IRF2BP2 oxygen sensing pathway triggers erythropoiesis terminal differentiation. Redox Biol. 2020;28:101313.
doi: 10.1016/j.redox.2019.101313
187.De Belly H, Paluch EK, Chalut KJ. Interplay between mechanics and signalling in regulating cell fate. Nat Rev Mol Cell Biol. 2022;23(7):465-480.
doi: 10.1038/s41580-022-00472-z
188.Zhou Z, Pang Y, Ji J, et al. Harnessing 3D in vitro systems to model immune responses to solid tumours: a step towards improving and creating personalized immunotherapies. Nat Rev Immunol. 2024;24(1):18-32.
doi: 10.1038/s41577-023-00896-4.
189.Nikolaev M, Mitrofanova O, Broguiere N, et al. Homeostatic mini-intestines through scaffold-guided organoid morphogenesis. Nature. 2020;585(7826):574-578.
doi: 10.1038/s41586-020-2724-8
190.Salmon I, Grebenyuk S, Abdel Fattah AR, et al. Engineering neurovascular organoids with 3D printed microfluidic chips. Lab Chip. 2022;22(8):1615-1629.
doi: 10.1039/d1lc00535a
191.Yi SA, Zhang Y, Rathnam C, Pongkulapa T, Lee K. Bioengineering approaches for the advanced organoid research. Adv Mater. 2021;33(45):2007949.
192.Jin H, Xue Z, Liu J, Ma B, Yang J, Lei L. Advancing organoid engineering for tissue regeneration and biofunctional reconstruction. Biomater Res. 2024;28:0016.
doi: 10.34133/bmr.0016
193.Chethikkattuveli Salih AR, Hyun K, Asif A, et al. Extracellular matrix optimization for enhanced physiological relevance in hepatic tissue-chips. Polymers (Basel). 2021;13(17):3016.
194.Bhar B, Das E, Manikumar K, Mandal BB. 3D bioprinted human skin model recapitulating native-like tissue maturation and immunocompetence as an advanced platform for skin sensitization assessment. Adv Healthc Mater. 2024;13(15):e2303312.
doi: 10.1002/adhm.202303312. Epub 2024 Mar 26. PMID: 38478847
195.Karwasra R, Sharma S, Sharma I, Shahid N, Umar T. Diabetology and nanotechnology: a compelling combination. Recent Pat Nanotechnol. 2025;19(1):4-16.
doi: 10.2174/0118722105253055231016155618
196.Joshi P, Nascimento HSD, Kang SY, et al. Dynamic culture of bioprinted liver tumor spheroids in a pillar/perfusion plate for predictive screening of anticancer drugs. Biotechnol Bioeng. 2025;122(4):995-1009.
doi: 10.1002/bit.28924
197.Borriello M, Tarabella G, D’Angelo P, et al. Lab on a chip device for diagnostic evaluation and management in chronic renal disease: a change promoting approach in the patients’ follow up. Biosensors. 2023;13(3):373.
doi: 10.3390/bios13030373
198.Gao X. Frontiers in biomedical engineering. Adv Funct Mater. 2020;30: 2005265.
199.Khodaverdi K, Naghib SM, Mozafari MR, Rahmanian M. Chitosan/hydroxyapatite hydrogels for localized drug delivery and tissue engineering: a review. Carbohydr Polym Technol Appl. 2025;9:100640.
doi: 10.1016/j.carpta.2024.100640
200.Marcos LF, Wilson SL, Roach P. Tissue engineering of the retina: from organoids to microfluidic chips. J Tissue Eng. 2021;12:20417314211059876.
doi: 10.1177/20417314211059876
201.Liu X, Sun A, Brodský J, et al. Microfluidics chips fabrication techniques comparison. Sci Rep. 2024;14(1):28793.
doi: 10.1038/s41598-024-80332-2
202.Duanmu L, Yu Y, Meng X. Microdroplet PCR in microfluidic chip based on constant pressure regulation. Micromachines (Basel). 2023;14(6):1257.
doi: 10.3390/mi14061257
203.Gu Y, Zhang W, Wu X, Zhang Y, Xu K, Su J. Organoid assessment technologies. Clin Transl Med. 2023;13(12):e1499.
doi: 10.1002/ctm2.1499
204.Baptista LS, Porrini C, Kronemberger GS, Kelly DJ, Perrault CM. 3D organ-on-a-chip: the convergence of microphysiological systems and organoids. Front Cell Dev Biol. 2022;10:1043117.
doi: 10.3389/fcell.2022.1043117
205.Lee J, Hong J, Kim W, Kim GH. Bone-derived dECM/alginate bioink for fabricating a 3D cell-laden mesh structure for bone tissue engineering. Carbohydr Polym. 2020;250:116914.
doi: 10.1016/j.carbpol.2020.116914
206.Ning L, Guillemot A, Zhao J, Kipouros G, Chen X. Influence of flow behavior of alginate-cell suspensions on cell viability and proliferation. Tissue Eng Part C Methods. 2016;22(7):652-662.
doi: 10.1089/ten.TEC.2016.0011
207.Coşkun S, Akbulut SO, Sarıkaya B, Çakmak S, Gümüşderelioğlu M. Formulation of chitosan and chitosan-nanoHAp bioinks and investigation of printability with optimized bioprinting parameters. Int J Biol Macromol. 2022;222(Pt A):1453-1464.
doi: 10.1016/j.ijbiomac.2022.09.078
208.Dravid A, McCaughey-Chapman A, Raos B, O’Carroll SJ, Connor B, Svirskis D. Development of agarose– gelatin bioinks for extrusion-based bioprinting and cell encapsulation. Biomed Mater. 2022;17(5):055001.
209.Amirrah IN, Lokanathan Y, Zulkiflee I, Wee MFMR, Motta A, Fauzi MB. A comprehensive review on collagen type I development of biomaterials for tissue engineering: from biosynthesis to bioscaffold. Biomedicines. 2022;10(9):2307.
doi: 10.3390/biomedicines10092307
210.Netti F, Aviv M, Dan Y, Rudnick-Glick S, HalperinSternfeld M, Adler-Abramovich L. Stabilizing gelatin-based bioinks under physiological conditions by incorporation of ethylene-glycol-conjugated Fmoc-FF peptides. Nanoscale. 2022;14(23):8525-8533.
doi: 10.1039/d1nr08206j
211.Wu D, Zheng K, Yin W, et al. Enhanced osteochondral regeneration with a 3D-Printed biomimetic scaffold featuring a calcified interfacial layer. Bioactive Mater. 2024;36:317-329.
doi: 10.1016/j.bioactmat.2024.03.004
212.Yi K, Li Q, Lian X, Wang Y, Tang Z. Utilizing 3D bioprinted platelet-rich fibrin-basedmaterials to promote theregeneration of oral soft tissue. Regen Biomater. 2022;9:rbac021.
doi: 10.1093/rb/rbac021
213.Laki K. The polymerization of proteins: the action of thrombin on fibrinogen. Arch Biochem Biophys. 2022;726:109244.
doi: 10.1016/j.abb.2022.109244
214.Al Kayal T, Buscemi M, Cavallo A, Foffa I, Soldani G, Losi P. Plasminogen-loaded fibrin scaffold as drug delivery system for wound healing applications. Pharmaceutics. 2022;14(2):251.
doi: 10.3390/pharmaceutics14020251
215.Zhang X, Chen X, Hong H, Hu R, Liu J, Liu C. Decellularized extracellular matrix scaffolds: recent trends and emerging strategies in tissue engineering. Bioact Mater. 2022;10:15-31.
doi: 10.1016/j.bioactmat.2021.09.014
216.Ning L, Yang B, Mohabatpour F, et al. Process-induced cell damage: pneumatic versus screw-driven bioprinting. Biofabrication. 2020;12(2):025011.
217.Dai M, Belaïdi JP, Fleury G, et al. Elastin-like polypeptide-based bioink: a promising alternative for 3D bioprinting. Biomacromolecules. 2021;22(12):4956-4966.
doi: 10.1021/acs.biomac.1c00861
218.Ding X, Zhao H, Li Y, et al. Synthetic peptide hydrogels as 3D scaffolds for tissue engineering. Adv Drug Deliv Rev. 2020;160:78-104.
doi: 10.1016/j.addr.2020.10.005
219.Jian H, Wang M, Dong Q, et al. Dipeptide self-assembled hydrogels with tunable mechanical properties and degradability for 3D bioprinting. ACS Appl Mater Interfaces. 2019;11(50):46419-46426.
220.Monfared M, Mawad D, Rnjak-Kovacina J, Stenzel MH. 3D bioprinting of dual-crosslinked nanocellulose hydrogels for tissue engineering applications. J Mater Chem B. 2021;9(31):6163-6175.
doi: 10.1039/d1tb00624j
221.Bandyopadhyay A, Mandal BB, Bhardwaj N. 3D bioprinting of photo-crosslinkable silk methacrylate (SilMA)-polyethylene glycol diacrylate (PEGDA) bioink for cartilage tissue engineering. J Biomed Mater Res A. 2022;110(4):884-898.
doi: 10.1002/jbm.a.37336
222.de Castro KC, Coco JC, Dos Santos ÉM, et al. Pluronic® triblock copolymer-based nanoformulations for cancer therapy: a 10-year overview. J Control Release. 2023;353:802-822.
doi: 10.1016/j.jconrel.2022.12.017
223.Serafin A, Murphy C, Rubio MC, Collins MN. Printable alginate/gelatin hydrogel reinforced with carbon nanofibers as electrically conductive scaffolds for tissue engineering. Mater Sci Eng C Mater Biol Appl. 2021;122:111927.
doi: 10.1016/j.msec.2021.111927
224.Monavari M, Homaeigohar S, Fuentes-Chandía M, et al. 3D printing of alginate dialdehyde-gelatin (ADA-GEL) hydrogels incorporating phytotherapeutic icariin loaded mesoporous SiO2-CaO nanoparticles for bone tissue engineering. Mater Sci Eng C 2021;131:112470.
doi: 10.1016/j.msec.2021.112470
225.McClements DJ. Composite hydrogels assembled from foodgrade biopolymers: fabrication, properties, and applications. Adv Colloid Interface Sci. 2024;332:103278.

