Differentiation of iPSC-derived neural progenitors into motor neurons in 3D-printed bioscaffolds

Translational medicine for neurodegenerative diseases can advance through the use of in vitro models incorporating human neural cells derived from patient-specific induced pluripotent stem cells (iPSCs). Previously, we investigated whether motor neuron (MN) progenitors derived from human iPSCs could differentiate from MNs within three-dimensional (3D)-printed scaffolds. While extensive neurite arborization was observed on the scaffold surface, no neurite outgrowth occurred within the scaffold interior. Here we showed the extensive growth of the neurites from iPSC-derived neural progenitors, imbedded into the gelatin scaffolds during 30 days of experimental time. We presented a bioink formulation that softens the scaffold while preserving its 3D structure, thereby facilitating neurite outgrowth throughout the scaffold. MN differentiation, evidenced by extensive neurite arborization and the expression of choline acetyltransferase (ChAT), was verified in 3D images deep within the scaffold structure. Notably, the degree of MN differentiation appeared to depend on two factors as follows: the delivery of MN differentiation factors via mesoporous silica particles (MSPs) embedded in the bioink and the method used to generate MN progenitors prior to 3D printing. In this paper, we provide a detailed protocol for 3D-printing human iPSC-derived MN progenitors, enabling their differentiation and survival within gelatin scaffolds. This protocol could be expanded to incorporate additional cell types, allowing the creation of more complex and standardized 3D neural tissues. Such advancements could facilitate investigations into the pathophysiology of motor neuron diseases and the development of new therapeutic strategies.
1. Introduction
Organoids can be defined as in vitro cellular systems that emerge by self-organization or as three-dimensional (3D) culture engineered by bioprinting, which include multiple cell types and exhibit some cytoarchitectural and functional features reminiscent of an organ or organ region.1 For the nervous system, organoids are generally constructed from pluripotent stem cells, but can also be generated from donor tissue with growth potential.1 Organoids generated from different brain regions have revolutionized neuroscience research, offering unprecedented opportunities to study human brain development, disease mechanisms, and therapeutic interventions.2–4 Unlike traditional 2D cell cultures, neural organoids closely resemble the spatial organization and cellular diversity of the human brain, making them invaluable for modeling complex neurological processes.
However, not all neural tissues possess the innate ability to self-organize into 3D organoids. The spinal cord, for instance, requires external scaffolds or guided assembly to mimic its elongated, segmented structure and functional organization. Unlike the brain, which has a more spherical and layered architecture, the spinal cord’s linear and highly structured arrangement is challenging to replicate without external cues. This fundamental difference underscores the need for innovative techniques, such as 3D bioprinting, to construct spinal cord organoids or hybrid neural systems.5,6
3D bioprinting has emerged as a transformative technology to address these challenges.7 Using bioinks composed of stem cells and biomaterials, it is possible to precisely control the spatial arrangement of neural progenitors, extracellular matrix components, and other factors. For spinal cord models, this approach is essential to achieve reproducible and functional structures that mimic the complex organization of this critical neural tissue. When applied to brain organoids, 3D bioprinting enables improved reproducibility, scalability, and integration with vascular and supporting structures, overcoming limitations of self-assembly alone. One of the major challenges using the technique is to design bioink formulations that ensure adequate printability and achieve the required level of functionality, for example, the differentiation of human motor neurons. The interaction between the imposed extrusion printing flow (bioprinting parameters) and the bioink, together with the temperature dynamics during solidification, is ultimately what determines the multiscale structure of a scaffold.8,9 In this framework, the rheological properties of the bioinks during bioprinting10 and during organoid differentiation are crucial.
The intersection of self-assembling brain organoids and engineered spinal cord constructs exemplifies the potential of combining biological and engineering innovations. The development of 3D neural tissue constructs holds significant potential for the replacement or repair of damaged tissue in vivo, as well as for in vitro studies of disease mechanisms, biomarker discovery, and therapeutic screening. By employing neural cells differentiated from patient-derived iPSCs, this approach can be personalized, enabling the investigation of patient-specific pathology and individualized treatment options.11
Our ultimate aim is to establish a standardized protocol for constructing human spinal cord tissue from patient-derived iPSCs.12 This protocol will enable personalized in vitro modeling of motor neuron (MN) diseases, such as amyotrophic lateral sclerosis (ALS) or spinal muscular atrophy (SMA). As a foundational step, we sought to develop a method for 3D printing standardized constructs of human iPSC-derived MNs. Our previous studies demonstrated that human iPSC-derived MN progenitors (MNPs) can survive and differentiate on the surface of gelatin-based scaffolds, exhibiting extensive neurite outgrowth.13 However, cells deep within the scaffold layer lacked MN marker expression and failed to extend neurites. Possible explanations for this outcome are insufficient diffusion of nutrients and growth factors essential for MN differentiation into the deeper parts of the construct, in combination with a very high bioink density to permit neurite outgrowth. To address these limitations, we developed a softer bioink that retains sufficient stiffness to maintain the 3D structure during the prolonged differentiation process. Furthermore, we systematically quantified material composition and optimized the preparation and printing process parameters. Recognizing the importance of viscoelastic properties in scaffold formation and tissue organization,14–18 we monitored bioink rheological properties throughout the protocol.
To support MN differentiation deep within the scaffold, we added mesoporous silica particles loaded with peptide mimetics of ciliary neurotrophic factor (Cintrofin, CNTF) and glial-derived neurotrophic factor (Gliafin, GDNF). The motivation was to achieve a prolonged, slow release of these factors inside the bioscaffold layers. Previously, we have successfully used this delivery system to guide stem cell differentiation towards MNs in vitro and in vivo after transplantation.19,20 Using the modified bioink and scaffold formulation, the iPSC-derived neural progenitors survived and achieved differentiation and robust neurite outgrowth within the scaffold during 30 days after printing, until the end of our analysis.
We propose that this 3D printing protocol can serve as a platform for producing standardized spinal cord tissue models from patient-derived iPSCs. With appropriate modifications to incorporate additional cell types, this protocol has the potential to create complex tissue models for studying MN diseases and spinal cord injuries, identifying new biomarkers, and screening therapeutic candidates in a precision medicine framework.
1.1. Research objective
A scaffold replicates the structural organization of the spinal cord, which, unlike the brain, has a linear and segmented architecture that requires external support for accurate modeling. The goal of 3D-printed constructs is to advance in vitro studies on disease mechanisms, biomarker discovery, and therapeutic screening. To this end, we aim to develop a 3D multicellular model called a spinal cord organoid (SCO) derived from human iPSCs. The findings from this research will lay the groundwork for future efforts in regenerating damaged spinal tissue in vivo. Additionally, the study’s findings will facilitate the development of in vitro models that can be used to test new drugs designed to treat spinal cord diseases. These models will allow researchers to evaluate the effectiveness and safety of potential treatments before they are tested in living subjects, ultimately accelerating the discovery of new therapies.
The key objective of this study is to develop a 3D-printed scaffold embedded with cells to support neurite outgrowth. To achieve this, we designed a structured lattice mesh with a height of 0.3 mm and a width of 0.75 mm, incorporating cells within the matrix of a 3-layer structure. Based on our previous research,13 a layer thickness of 0.3 mm facilitates the diffusion of nutrients, oxygen, and differentiation factors throughout the scaffold, ensuring that even cells located at its core receive adequate support. At the same time, a layer width of 0.75 mm maintains mechanical stability, while an increase in width could greatly restrict diffusion, hinder the removal of byproducts, and compromise cellular viability.
A lattice mesh structure was chosen for its suitability in 3D bioprinting, enabling precise control over the size and interconnectivity of lattice cells. The design featured dimensions of 14 mm × 14 mm to fit within a 12-well plate, with individual cell units measuring 2 mm × 2 mm. This structural configuration supported extensive neurite outgrowth throughout the construct inside the extruding layers while maintaining its stability for at least 1 month in culture.
2. Materials and methods
2.1. Boundary cap neural crest stem cell (BC) culture The Uppsala Regional Ethics Committee for Research on Animals of the Swedish Board of Agriculture approved all animal procedures. Boundary cap neural crest stem cells (BCs) were prepared from 11-day-old (E11) transgenic mouse embryos harboring red fluorescent protein under the universal actin promoter and cultured as previously described.21 The spinal cord was exposed and dorsal root ganglia, including their attachment with the spinal cord, gently separated and mechano-enzymatically dissociated using collagenase/dispase (1 mg/mL) and DNase (0.5 mg/ mL) for 30 min at room temperature. Cells were plated at 0.5–1 × 105 cells/cm2 in DMEM/F-12 medium (Gibco, 11330032) containing B27 (ThermoFisher, 17504044) and N2 (ThermoFisher, 17502048) as well as epidermal growth factor (EGF; R&D Systems, 236-EG) and basic fibroblast growth factor (bFGF; R&D Systems, 3718-FB; 20 ng/ mL, respectively). After 12 h of culture, cells that had not adhered were removed together with half of the medium, and a fresh medium was added. The medium was changed every second day, and neurospheres could be observed after about 2 weeks of culture.
2.2. Culture of human iPSCs prior to 3D printing
Prior 3D printing, we used iPSCs cultured as monolayer or as neurospheres. The human iPS cell line ALS52 was purchased from the biorepository at Cedar Sinai and the work was performed in line with the material transfer agreement (MTA) between the University of Sheffield and Cedar Sinai. iPSC-derived neural progenitor cells (NPCs) were produced by cultivating iPSCs on Matrigel-coated plates in mTeSR plus medium (Stem Cell Technologies, Canada). After achieving 100% confluence, the cells were passaged 1:1 using ReLeSR onto Matrigel-coated plates in mTeSR plus and 10 μM Y27632 (Tocris, 1254) for 24 h. The Matrigel was obtained from Corning (356230). The media was then changed to the neural induction medium (Table 1). The medium was changed every day for 6 days and then was switched to the NPC medium. Every day for 6 days, the medium was changed. Thereafter, the cells were passaged 1:6 with Accutase onto Matrigel-coated plates in NPC medium with 10 μM Y27632 (for the first 24 h only) and valproic acid (0.5 mM) for expansion. Then, the NPC medium was replaced with the MNP medium to induce differentiation into MNPs after 6 days. The wells were rinsed with warm HBSS (Stem Cell Technologies, Canada), and room-temperature Accutase (ThermoFisher, A1110501) was then used for 10 min in the incubator until the material could be lifted from the well, then the cells were collected for printing with bioink, after several times of gentle pipetting.
Table 1. Recipes of culture media used to grow human iPSCs into monolayers before 3D printing
| Neural induction medium | NPC medium | MNP medium | |||
|---|---|---|---|---|---|
| KnockOut™ DMEM/F-12 (ThermoFisher, 12660012) | 50% | KnockOut™ DMEM/F-12 | 50% | KnockOut™ DMEM/F-12 | 50% |
| Neurobasal medium (ThermoFisher, 21103049) | 50% | Neurobasal medium | 50% | Neurobasal medium | 50% |
| N2 Supplement 100× | 0.5× | N2 Supplement 100× | 0.5× | N2 Supplement 100× | 0.5× |
| B27 Supplement 50× | 0.5× | B27 Supplement 50× | 0.5× | B27 Supplement 50× | 0.5× |
| Glutamax (ThermoFisher, 35050061) | 1× | Glutamax | 1× | Glutamax | 1× |
| Penicillin-streptomycin (Gibco, 15140122) | 1% | Penicillin-streptomycin | 1% | Penicillin-streptomycin | 1% |
| CHIR (Tocris, 4423) | 3 µM | CHIR | 3 µM | RA | 0.1 µM |
| DMH1 (Tocris, 4126) | 2 µM | DMH1 | 2 µM | PMN (Pur) | 0.5 µM |
| SB (Tocris, 1614) | 2 µM | SB | 2 µM | ||
| RA (STEMCELL Technologies, 72262) | 0.1 µM | ||||
| PMN (Pur) (Tocris, 4551) | 0.5 µM | ||||
Abbreviations: iPSCs, induced pluripotent stem cells; MNP, motor neuron progenitor; NPC, neural progenitor cell.
The work with human iPSCs, cultured as neurospheres, was performed with the approval of the Southeastern Norway Regional Ethics Committee (approval REK 2017/110). 15,000 iPSCs/cm2 were plated as single cells in E8 medium with ROCK inhibitor on GelTrex-coated wells in a 6-well plate and incubated overnight at 37°C and 5% CO2. The medium was aspirated from each well and induction was initiated with N1-medium containing 50% DMEM/F12, 50% Neurobasal medium, 0.5% N2-supplement, 0.5% B-27 without RA, 1% NEAA 100×, 1% GlutaMax 100×, 2 μM SB431542, 0.3 μM LDN193189, and 3 μM CHIR99021 (Table 2). The induction medium was changed every day until rosettes appeared, typically after 4 days. Cells were then split using STEMdiff™ Neural Rosette Selection Reagent (SNRS; Stem Cell Technologies, Canada) by first removing media and then incubated with 1 mL SNRS for 2 h at 37°C and 5% CO2.
Table 2. Recipes of culture media used to grow human iPSCs into neurospheres before 3D printing
| N1-medium | N2-medium | N3-medium | |||
|---|---|---|---|---|---|
| DMEM/F-12 | 50% | DMEM/F-12 | 50% | DMEM/F-12 | 50% |
| Neurobasal medium | 50% | Neurobasal medium | 50% | Neurobasal medium | 50% |
| N2 Supplement 100× | 0.5× | N2 Supplement 100× | 0.5× | N2 Supplement 100× | 0.5× |
| B27 Supplement 50× | 0.5× | B27 Supplement 50× | 1× | B27 Supplement 50× | 1× |
| NEAA 100× (Gibco, 11140050) | 1% | NEAA 100× | 1% | NEAA 100× | 1% |
| GlutaMax | 1% | GlutaMax | 1% | GlutaMax | 1% |
| SB431542 | 2 µM | LDN193189 | 0.3 μM | Ascorbic acid | 200 μM |
| LDN193189 (Tocris, 6053) | 0.3 µM | Ascorbic acid | 200 μM | RA | 0.1 μM |
| CHIR99021 | 3 µM | SB431542 | 2 μM | PMN (Pur) | 0.5 μM |
| CHIR99021 | 3 μM | ||||
| RA | 0.1 μM | ||||
| PMN (Pur) | 0.5 μM | ||||
Abbreviations: iPSCs, induced pluripotent stem cells.
SNRS was removed and 1 mL washing buffer, containing DMEM/F-12 with 1% GlutaMax and 15 mM Hepes, was splashed into each well. Washing buffer with cells was gently transferred to a 14 mL centrifugation tube. This step was repeated five times. Cells were then centrifuged at 100 × g for 5 min. The supernatant was removed, and the cell pellet was resuspended in N2-medium. N2-medium was changed every other day. At day 12, the medium was removed, and 1 mL accutase was added to each well and incubated on the bench until the cells started to detach. Accutase was removed and 1 mL washing buffer, as described above, was splashed into each well to generate clusters of cells. Washing buffer with clusters was then gently transferred to a 14 mL centrifugation tube using a 10 mL pipette. This step was repeated four times. Clusters were centrifuged 100 × g for 7 min. The supernatant was removed, and the pellet was splashed with 4 mL N2-medium and transferred to one well in a low attachment 6-well plate. The plate was swirled to get all the clusters in the middle of the well and incubated overnight at 37°C and 5% CO2.
All neurospheres were gently transferred to a 14 mL centrifugation tube using a 10 mL pipette and placed on the bench until all the neurospheres had sedimented, typically in 4–5 min, depending on the size. The supernatant was removed and the pellet was splashed with 4 mL N3-medium and transferred into the same well in the low attachment 6-well plate. The medium was changed every other day until mixed with the bioink for printing.
2.3. Scaffold preparation
2.3.1. Hydrogel ink printing process: Method 1
10% gelatin was heated up to 37 °C and mixed with 10% 30 mg/mL microbial transglutaminase (mTG) and 10 µg/ mL laminin (BioLamina, LN521) to reach 4% final gelatin– cell mixture, which then was loaded into 3 mL luer-locker syringe. The syringe with bioink was placed in 4 °C fridge for 30 min, then incubated in a water bath set at 27°C, and placed back in the fridge until the point at which it would not melt at 27°C; the whole process usually takes 6–8 h. Thereafter, single cells were mixed with the bioink using two syringes connected by a luer-locker connector. Lastly, the final mixture was connected to GESIM BioScaffolder 3.3 PRIME printer (Gesim mbH, Germany) with a temperature-controlled printing head and printing bed of 4°C. Three-layer scaffolds were printed in the 12-well plate according to the settings for the printer. After printing, the medium was added before further culturing in the incubator.
2.3.2. Hydrogel ink printing process: Method 2
10% gelatin was heated up to 37°C and mixed with single cells and 10% 30 mg/mL mTG and 10 µg/mL laminin to reach 4% final gelatin–cell mixture, which was then loaded into 3 mL luer-locker syringe for printing. The syringe with the bioink was placed in a 4°C fridge for 10 min and connected to GESIM BioScaffolder 3.3 PRIME printer with temperature-controlled printing head and printing bed of 4°C. Three-layer scaffolds were printed in the 12-well plate following the setting (Table 1). After printing, each printed scaffold was soaked in 30 mg/mL mTG for 10 min at 4 °C, then the medium was added before culturing in the incubator.
2.4. Rheology tests
All rheological experiments were performed on an Anton Paar MCR705e Space rotational rheometer (Anton Paar, Austria) in a separate motor-transducer configuration, using a 50 mm diameter parallel plate measuring geometry and 1 mm gap. The bottom plate consisting of a Peltier heating element and a temperature Peltier hood was used to ensure a uniform temperature in the measuring gap. To avoid drying at temperatures above the sol–gel transition temperature of gelatin during extensive testing, a standard plastic solvent trap was added to the setup, with water as a sealant. An overview of the setup configuration is presented in Figure S1 (Supporting Information). Four types of tests were performed: (i) temperature ramp tests (gelation), (ii) time-dependent constant-temperature (gelation vs. crosslinking), (iii) temperature oscillation tests, and (iv) cooling tests.
A simple temperature ramp oscillatory shear test (i) from 37 to 15°C was performed to confirm the sol–gel transition of the compositions. The cooling rate was set to 1°C/min and the strain amplitude and angular frequency were set to 4% and 1 rad/s, respectively. In test (ii), the temperature was set to 4°C, the strain amplitude to 2% and angular frequency to 1 rad/s, and the linear viscoelastic material reponse was monitored with time.
In test (iii), a custom time–temperature multi-interval test was applied (Figure S2, Supporting Information). The custom test was designed to mimic the lab procedure used to assess flowability and gel formation prior to printing. Thus, the temperature was varied sequentially between 27°C for 10-min intervals and 4°C for 30-min intervals to a total of approximately 17 h. Finally, approximately every 15 h the temperature was increased to 37°C and a time sweep was performed for 200 s. Thus, the test could probe how and when the samples become rheological gels under the influence of mTG during low-temperature storage.
For test (iv), the gelatin was pre-heated to 37°C for 30 min. Thereafter, it was quickly mixed with mTG and placed on the bottom plate, which had been pre-heated to 37°C. Thereafter, the upper geometry was lowered and the test was immediately started. The cooling rate was determined by direct measurement of a 2 mL syringe containing a 10% gelatin solution pre-heated at 37°C and then placed in the fridge. The temperature was recorded using a thermocouple inserted into the bulk of the solution through the syringe nozzle. The thermocouple was connected to a standard multimeter to record the temperature as a function of time (see Figure S3, Supporting Information). The data were found to be well described by the so-called Newton’s law of cooling (see Equation S1, Supporting Information). Thus, the interpolated datapoints were used to impose the temperature as a function of time during measurements.
2.5. Delivery of MN differentiation factors with mesoporous silica particles
To promote MN differentiation, mesoporous silica particles (MSP) loaded with peptide mimetics of glia-derived neurotrophic factor (Gliafin) and ciliary neurotrophic factor (Cintrofin)20 were added to the bioink of some scaffolds before printing. The final concentration of growth factors added to the bioink was 2 μg/mL. To examine the distribution of MSP within the scaffold, we used rhodamine-labeled MSPs.20
2.6. Live cell analysis
Droplets with BCs prepared in two different methods were placed in 4-well plates with 1 mL proliferation medium, then 50 μM Calcein AM Viability Dye (1:10; Invitrogen, #15560597) was added for a 30-min incubation at 37°C. After PBS washing for three times, Hoechst 33342 dye (1:1000; Invitrogen, 2433875) was added into each well for 5 min. The droplets were taken out of the 4-well plates and placed on slides for imaging by Zeiss Axio Imager Z2 (Carl Zeiss AG, Germany).
2.7. Culture of 3D-printed scaffolds
3D scaffolds with monolayer culture were placed in 12-well plates, and cultured with 3 mL of the MNP medium (Table 1) for 5 days, with changing of medium every day. On day 5, the medium was changed to the Young MN medium (Table 3) and the medium was changed every day. After 9 days with the Young MN medium, the MN medium (Table 3) was used instead until all scaffolds were sectioned at different time points. The 3D scaffolds with neurosphere culture were placed in 12-well plates, and cultured with 3 mL of the N3-medium (Table 1) for 10 days, with changing of medium every other day. On day 10, the medium was changed to the N4-medium (Table 3) until all scaffolds were fixed and sectioned at different time points.
Table 3. Recipe of media for culturing 3D-printed scaffolds
| Young MN medium | MN medium | N4-medium | |||
|---|---|---|---|---|---|
| KnockOut™ DMEM/F-12 | 50% | Neurobasal medium | 100% | Neurobasal medium | 100% |
| Neurobasal medium | 50% | B27 Supplement 50× | 2× | B27 Supplement 50× | 2× |
| N2 Supplement 100× | 0.5× | BDNF | 10 ng/mL | NEAA 100× | 1% |
| B27 Supplement 50× | 0.5× | CNTF | 10 ng/mL | GlutaMax | 1% |
| Glutamax | 1× | IGF | 10 ng/mL | GDNF | 10 ng/mL |
| Penicillin-streptomycin | 1% | BDNF | 10 ng/mL | ||
| RA | 0.5 μM | ||||
| PMN (Pur) | 0.1 μM | ||||
| Compound-E (Tocris, 6476) | 0.1 μM | ||||
| BDNF (PeproTech, AF-450-02) | 10 ng/mL | ||||
| CNTF (PeproTech, 450-13) | 10 ng/mL | ||||
| IGF (PeproTech, 100-11) | 10 ng/mL | ||||
Abbreviations: BDNF, brain-derived neurotrophic factor; CNTF, ciliary neurotrohic factor; GDNF, glial cell line-derived neurotrophic factor; IGF, insulin-like growth factor; MN, motor neuron; NEAA, non-essential amino acids aolution.
2.8. Immunohistochemistry
3D-printed scaffolds were fixed in 4% paraformaldehyde for 15 min and washed three times in phosphate-buffered saline (PBS). The fixed scaffolds were soaked in Tissue-Tek (Sakura Finetek, Alphen aan den Rijn, The Netherlands) overnight, and then frozen on dry ice for sectioning on a cryostat. Consecutive serial sections were prepared through the scaffold and distributed on five slides so that all levels of the scaffold were represented on each slide. The sections were covered by pre-incubation solution (1% bovine serum albumin, 0.1% sodium azide NaN3, and 0.3% Triton X-100 in PBS) for 30 min at room temperature, and thereafter incubated at 4°C with antibodies to glial fibrillary acidic protein (GFAP; rabbit, 1:400, Dako, Z0334) and beta-tubulin (bTUB; mouse, 1:400, ZYMED,32-2600) overnight. Some slides were stained for choline acetyltransferase (ChAT; goat, 1:200; Millipore, AB144P). Secondary antibodies Alexa Fluor 488 goat anti-rabbit (H+L; 1:800, Life Technologies, A21206) and Texas Red goat anti-mouse (H+L; 1:800, Invitrogen, 1862) were applied for 4 h at room temperature. After washing for three times, Hoechst 33342 dye (1:1000, Invitrogen, 2433875) was added for 5 min. The stained sections were washed three times in PBS and embedded in 6 μL of mounting solution (50% glycerol in PBS and 100 mM propyl gallate) (Sigma-Aldrich) and covered with coverslips for imaging.
2.9. Imaging
The stained slides of scaffolds, which had been in culture for 5, 10, 20, or 30 days, were subjected to confocal imaging with a Carl Zeiss LSM 700 Laser Scanning Microscope with a 63×/1.4 oil lens. 3D images of the scaffold sections were taken with a z-stack of 1 µm interval and formed by Zen Blue software in different angles. For cell counts, images of the stained slides of scaffolds were taken with a Zeiss Axio Imager Z2 with a 20×/0.75 and 40×/0.95 lens.
2.10. Assessment of cell differentiation
Differentiation of MNs was assessed by estimating the number of Chat-positive cells, as well as by the extent of neurite outgrowth.22 For the quantitative analyses, three images from three scaffolds of each condition (n = 9) were taken. The total neurite length per cell was determined by counting the number of intersections between neurites and test lines of an unbiased counting frame superimposed on images from different angles of sections of fluorescent-labeled MNs. The ratio between the number of neurite intersections and the number of cell bodies was used as a measure of MN differentiation. The effect of MSP and non-MSP exposure, as well as the effect of pre-culture of MN progenitors, on neurite outgrowth was estimated with the same method, using 20×/0.75 lens for counting.
2.11. Statistical analysis
GraphPad software was used for statistical analysis and graph presentation. t-test was employed in comparisons to determine the statistical significance in the cell differentiation study.
3. Results
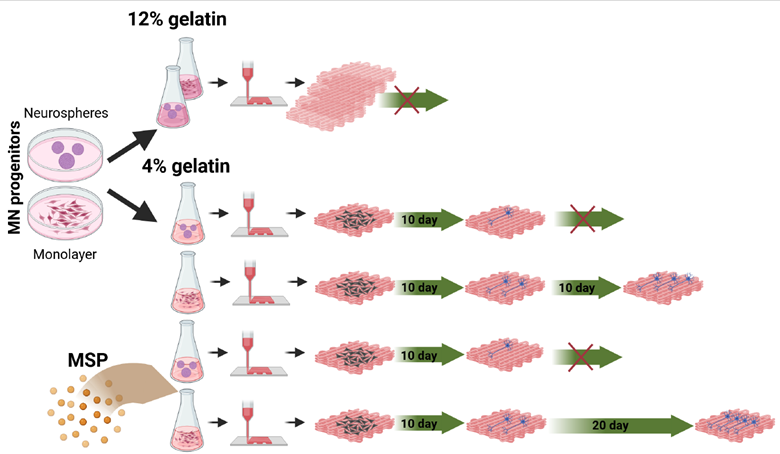
Our previous findings demonstrated that MNPs, which were embedded in 3D-printed scaffolds, differentiated to MNs on the surface of a gelatin scaffold, but failed to achieve neurite outgrowth within the scaffold.13 To address this limitation, our objective here was to achieve neuron survival, MN differentiation, and extensive neurite outgrowth throughout the scaffold. We hypothesized that reducing the gelatin concentration to 4% would increase bioink softness, enhancing the permeability of the scaffold to nutrients and growth factors while promoting neurite outgrowth.
To support scaffold integrity despite the lower gelatin concentration, we used rheological tests to design a protocol to facilitate rapid scaffold setting through the inclusion of the crosslinker microbial transglutaminase (mTG). This is due to the importance of viscoelastic properties both in the printing and cell differentiation stage. This method aimed to balance scaffold softness with sufficient structural stability for sustained MN differentiation. Furthermore, to enhance differentiation within the scaffold, we preloaded the bioink with MSPs containing the neurotrophic peptide mimetics Cintrofin (CNTF) and Gliafin (GDNF) and compared their effect on MN differentiation compared to MSP-free scaffolds, in which cells received differentiation factors only through the culture medium. MSPs loaded with differentiation factors have previously been shown to effectively guide MN differentiation in vitro and after the transplantation of MN progenitors into animal models.19,20 To achieve a stable 3D scaffold capable of retaining its structure at physiological temperatures (37°C), we developed and compared two preparation strategies (Figure 1). In the first approach (Preparation Method 1, M1), gelatin and crosslinker were mixed without cells, and cells were added only after allowing sufficient time for the crosslinker to activate. This step ensured scaffold stabilization prior to cell incorporation. In the second approach (Preparation Method 2, M2), scaffolds were printed and then soaked in a crosslinker solution post-printing to enhance stiffness.
Figure 1. Overview of the 3D printing process. (A) Method 1 (M1): performing gelation prior to mixing the cells (MNP) with bioink and printing. (B) Method 2 (M2): directly mixing the cells (MNP) with bioink (gelatin) and performing fast gelation after printing through coverage with mTG. Abbreviations: MNP, motor neuron progenitor; mTG, microbial transglutaminase.
To refine and standardize the preparation protocol, we performed rheological tests in parallel with preliminary printing trials. These experiments provided a quantitative understanding of the scaffold’s material response and evolution during the preparation process. Viscoelastic properties, including the sol–gel transition temperature, were identified as critical determinants for both printability and subsequent cellular differentiation.17 The sol–gel transition temperature of the gelatin solution was confirmed to be approximately 27°C (Figure 2A). In gelation tests, the absence of a measurable storage modulus indicated that the gelatin solution behaved as a near-pure viscous (Newtonian) fluid.
Figure 2. Overview of the sol–gel transition and the contribution of microbial transglutaminase (cross-linking) to the gelation process. (A) Gelation test identifying the sol-gel temperature as the crossing between the dynamic (shear) moduli, G′, G″, with decreasing temperature (37-15°C) using 10% gelatin (G10). (B) Influence of microbial transglutaminase (mTG) in 4% gelatin (G4) on changes in the time-dependent storage modulus ( G′) during a constant temperature test (4°C).
The primary challenges for successful scaffold preparation included achieving sufficient consistency for effective printing and ensuring the crosslinker retained sufficient action time to stabilize the printed scaffolds at 37°C. Addressing these challenges required iterative optimization of gelatin concentration, crosslinker timing, and rheological properties to support both scaffold integrity and cellular differentiation.
We then investigated the interplay between gelation and crosslinking in hydrogel formation by conducting a constant-temperature test at 4°C (Figure 2B). In this test, the time-dependent storage modulus, Gʹ(t), was normalized by the initial storage modulus at the beginning of the experiment, G′(t = 0). During the initial phase (t < 3 h), crosslinking significantly influenced the process, as evidenced by a notable relative increase in modulus for the mTG-containing composition. Beyond this period, a consistent ~20% difference in G′ was observed between the samples, with both compositions displaying a similar rate of increase in modulus. This convergence in behavior beyond ~2 h is attributed to the prolonged timescales required for the gelation process to reach equilibrium.23
To mimic the conditions of Preparation Method 1 (M1), we conducted temperature oscillation tests between 4°C (refrigeration temperature) and 27°C (sol– gel transition temperature), with a final evaluation of viscoelastic properties at 37 °C, simulating the incubation environment (Figures S2 and S4, Supporting Information). As expected, at 4°C, the hydrogels exhibited gel-like behavior, with a steady increase in dynamic moduli over time. At 27°C, both 4% and 10% gelatin samples displayed a gel-like response after 2 h. After 31–32 h at 37°C, the storage modulus for the 4% gelatin-mTG sample stabilized at approximately 0.21 kPa, while the 10% sample reached ~0.46 kPa, both with G′ > G″ (Figure 2A). However, we note that one critical environmental parameter, relative humidity, could not be fully replicated during these tests. The rheological experiments required a solvent trap to prevent drying, which introduced additional moisture into the system through condensation within the trap (Figure S1, Supporting Information). This supplemental moisture likely influenced the hydrogel properties, resulting in conditions that did not perfectly replicate those in M1. Nevertheless, based on the data in Figure 2B, we infer that temperature oscillations could modulate both crosslinking kinetics and gelation, potentially slowing structural development.
This effect was particularly evident during the early stages of testing (t < 10 t) in Figure S4, Supporting Information. At a constant 4°C, dynamic moduli increased over time due to ongoing gel-structure formation. Conversely, at 27°C, dynamic moduli decreased over time, likely due to softening effects associated with the sol–gel transition. Despite this, the cumulative effect of successive temperature oscillations was an overall increase in dynamic moduli. This outcome highlights the importance of environmental parameters such as temperature and humidity in modulating the viscoelastic properties and structural development of the hydrogels.
These findings emphasize the need for precise control of environmental conditions during scaffold preparation to ensure reproducible outcomes. The interplay of gelation and crosslinking, influenced by temperature variations, provides critical insights into the design of bioinks and printing parameters optimized for 3D bioprinting applications.
We subsequently conducted cooling tests to simulate the conditions of Preparation Method 2 (M2), by applying a cooling profile on the rheometer that corresponded to the temperature changes experienced during scaffold preparation. These tests aimed to determine the minimum time required for the formulations to achieve a predominant elastic response (G′ > G″), indicative of gel-like behavior, under transient temperature conditions and specific geometrical constraints. The results indicated that the minimum time required to achieve this transition was approximately 10 min, irrespective of the presence of MSP (Figure 3A, also see Figure S5 in the Supporting Information).
Figure 3. Summary of the (A) method 2 (M2) cooling tests (see Figure S3, Supporting Information) and (B) temperature oscillation tests (see Figure S2, Supporting Information), comparing several material response parameters. (A) Minimum gelation time of 4% gelatin with and without MSP and 10% gelatin (lower panel) and storage modulus at the end of the cooling tests (upper panel), as the temperature was decreased from 37 to 4°C (see Figure S5, Supporting Information). (B) Comparison of stiffness between 4% and 10% gelatin with Method 1 (M1) after approximately 31–33 h at 37 °C in a temperature oscillation test (see data in Figure S4, Supporting Information), and 4% gelatin final product with Method 2 (M2).
When the gelatin concentration was reduced from 10% to 4%, this minimum time increased by approximately 33%. In complementary preliminary tests, we applied a cooling procedure to 8°C and observed no significant differences in either the onset of the elastic response or the final mechanical properties of the hydrogel (data not shown). However, these conditions proved insufficient for maintaining scaffold stability in preliminary 3D printing experiments. When using a printing bed temperature of 8°C, the scaffolds printed using M2 exhibited structural instability. Specifically, after 2.5 h, the elastic modulus (G′) of the 4% gelatin formulation decreased by approximately 74% compared to the 10% gelatin formulation. This substantial reduction in mechanical stability highlights the challenge of balancing scaffold softness with structural integrity in low-concentration gelatin formulations.
Overall, these findings underscore the importance of optimizing cooling protocols and gelatin concentrations to ensure scaffold stability during and after printing. While the 4% gelatin formulation provides a softer environment conducive to neurite outgrowth and nutrient diffusion, its reduced mechanical stability necessitates careful adjustment of the preparation parameters, particularly in M2, where post-printing stabilization relies heavily on crosslinking dynamics and gelation kinetics.
The following two preliminary conclusions can be drawn: (i) a reduction in gelatin concentration primarily affects gel stiffness/softness, and (ii) the presence of MSP does not significantly influence the viscoelastic properties of the hydrogels. Based on the rheology tests and preliminary printing attempts, we could conclude that a decrease in gelatin concentration predominantly alters gel stiffness/softness, with a lesser effect on the time required to achieve gel-like behavior. This is particularly important in confirming that the preliminary printing tests could be evaluated within similar time scales. Furthermore, the stiffness of the gel can change by over 50% between 10% and 4% gelatin compositions, depending on the preparation method and environmental conditions. As a result, we determined the time and temperature during the printing process for both M1 (Figure 1A) and M2 (Figure 1B). The stiffness of 4% gelatin scaffolds generated using M2 was 0.5 kPa (Figure 3B), which is suitable for neuronal differentiation. However, since the ambient humidity in the rheology tests could not be matched to the conditions in the scaffold preparation environment (Figure S13, Supporting Information), we could not accurately determine the stiffness of the scaffolds in M1.
To assess and compare cell loss during the procedures, BCs were tested in M1 (Figure 4A) and M2 (Figure 4B) before printing. Comparing the two methods, the fraction of viable cells after mixing with liquid bioink was significantly higher (0.81 ± 0.06) than that after mixing with gelled bioink (0.51 ± 0.04, **p < 0.005; Figure 4C). The BC spheres were also tested in both methods. Using M1, the BC spheres were disrupted (Figure 4D), while with M2, the BC spheres maintained a well-defined spherical structure after being mixed with bioink (Figure 4E). Therefore, M2 was chosen for printing.
Figure 4. Overview of viability of boundary cap neural crest stem cells (BCs) printed with different methods (M1 and M2). (A, B) Boundary cap neural crest stem cell (BC) viability in the bioink from Method 1 (A) and Method 2 (B) before printing. Green: Calcein; Blue: Hoechst. (C) The percentage of live cells was assessed after mixing BCs with bioink before printing. (D, E) Images of BC spheres from method 1 (D) and method 2 (E). The average cell number was estimated using ImageJ via virtual measurements. Data and means are from three independent experiments. **p < 0.005. Scale bar = 100 µm. M1 and M2 refer to the scaffold preparation methods described in Figure 1.
Our ultimate goal is to develop a 3D-printed bioscaffold generated from iPSC-derived cells, allowing them to survive for extended periods, establish interconnections, and differentiate into functional cells. Therefore, we tested whether we could generate MNs from human iPSCs, cultured either as a monolayer (Figure 5A) or as neurospheres (Figure 5B), prior to 3D printing the scaffold (Figure 5C). In some cases, the bioink was also mixed with MSP loaded with peptide mimetics Cintrofin (CNTF) and Gliafin (GDNF).19,20 The uniform distribution of MSPs in the scaffolds after 3D printing was confirmed using redfluorescent MSPs (Figure S6, Supporting Information).
Figure 5. Culture of human iPSCs prior to 3D printing. (A, B) iPSC-derived NPCs cultured as monolayer (A) and iPSC-derived NPCs as neurospheres (B) in culture dish before the printing process (see Section 2). (C) 3D-printed scaffold covered with mTG (see Research objective Objective background in Section 2). Scale bar: 200 µm (A, B); 1 cm (C). Abbreviations: iPSCs, induced pluripotent stem cells; mTG, microbial transglutaminase; NPCs, neural progenitor cells.
The printed 300 µm scaffolds were placed in MN differentiation medium for 20–30 days. Three scaffolds per group were collected at various time points, with some cultures maintained for up to 30 days after printing. At day 5 and day 10, three scaffolds from each group were collected for immunohistochemical analysis. Thick sections, each 12 µm, were prepared using a cryostat and stained with antibodies against the neuronal marker betatubulin (bTUB) and the astrocytic marker glial fibrillary acidic protein (GFAP), along with Hoechst nuclear staining. During antibody incubation, the sections swelled due to the hydrophilic properties of gelatin.
On day 5, we observed more bTUB-positive cells in MSP-containing scaffolds compared to MSP-free scaffolds, both when cells were cultured as spheres prior to dissociation and printing (Figure 6A, B, and I) and when cultured as monolayer (Figure 6E, F, and J). However, by day 10, there was no significant difference in the percentage of bTUB-expressing cells in MSP-treated scaffolds compared to MSP-free scaffolds (Figure 6C, D, and G–J). Neurite outgrowth became clearly visible by day 10 in scaffolds with cells cultured as monolayers prior to printing (Figure 6G and H), whereas scaffolds containing cells cultured as neurospheres showed no neurite extensions on day 10 (Figure 6C). There was no significant difference in the level of differentiation on day 10 in scaffolds treated with MSP compared to MSP-free scaffolds (not shown). At the same time, a higher degree of astrocyte differentiation was observed in MSP-containing scaffolds both on day 5 and day 10 (Figure 6B, D, F, and H).
Figure 6. Differentiation in 3D-printed scaffolds after 5 days without MSP (A, C) and with MSP (B, D), and after 10 days without MSP (E, G) and with MSP (F, H). Left panel (A–D): cells were grown as neurospheres before printing; right panel (E–H): cells were grown as monolayers before 3D printing. Red: bTUB; green: GFAP; blue: Hoechst. Scale bar = 5 µm. (I, J) Percentage of bTUB-positive cells on day 5 and day 10 in 3D-printed scaffolds with or without MSP after pre-culture of cells as neurospheres (I) or monolayers (J). Three images from three scaffolds of each condition (n = 9) were taken with a 20×/0.75 lens for analysis. The percentage of bTUB-positive cells was calculated by dividing the number of bTUB-positive cells by the total number of cells. **p ≤ 0.005, ***p ≤ 0.001.
At later stages of the experiment, we did not observe differentiation in the scaffolds containing dissociated NPCs cultured as neurospheres. In contrast, the differentiation of MNP cultured as monolayers maintained extensive neurite outgrowth throughout the 30 days of scaffold culture (Figure 7).
Figure 7. Differentiation of motor neurons in MSP-treated and MSP-untreated scaffolds. Overview of neural differentiation on day 20 in MSP-free (A) and MSP-treated (B) 3D-printed scaffolds and on day 30 after printing in MSP-treated scaffold (C). The sections (A, B, C, E, F) were prepared as composite images from Z-stacks through 20-µm-thick cryosections. Red: bTUB; green: GFAP; blue: Hoechst. Scale bar = 5 µm. All scaffolds were prepared from iPSC-derived progenitors cultured as monolayer prior to 3D printing. Three images from three scaffolds of each condition (n = 9) were taken with a 40×/0.95 lens to estimate neuronal differentiation (neurite intersections per cell)22 (***p ≤ 0.001). (E) ChAT-positive MNs (combined image, green) on day 20 in MSP-free (E) and MSP-treated (F). Scale bar = 10 µm. The percentage of ChAT-positive cells on day 20 is higher in MSP-containing scaffolds compared to the MSP-free scaffolds (G, **p ≤ 0.005). Three images of three scaffolds of each condition (n = 9) were taken with a 5×/0.15 lens. Abbreviations: bTUB, beta tubulin; ChAT, choline acetyltransferase; iPSC, induced pluripotent stem cell; MN, motor neuron; MSP, mesoporous silica particle.
Neurite outgrowth, as a characteristic of neuronal differentiation, was quantified by counting neurite intersections with testlines in a counting frame, normalized to the number of cells in the section.22 Neuronal differentiation was greater in MSP-treated scaffolds on day 20 compared to scaffolds with MSP untreated cells (Figure 7A, B, and D). Expression of the MN marker choline acetyltransferase (ChAT) was detected from day 20 of differentiation (Figure 7E and F) both in treated and untreated scaffolds. MSP-containing scaffolds exhibited a significantly higher fraction of ChAT-positive cells and more extensive neurite arborization on day 20 compared to MSP-free scaffolds (Figure 7E–G). Extensive neurite outgrowth was detected throughout the scaffolds, as shown by 3D reconstructions (Figure 8), and neuronal differentiation on day 20 was significantly greater in scaffolds treated with MSP compared to MSP-free scaffolds (Figure 8).
Figure 8. Overview of neurite growth in 3D printed scaffolds reconstructed from Z-stuck images at different view angles demonstrating effect of MSP and of 4% gelatin on neurite growth inside the bioscaffolds: 12% gelatin (A) shows no growth inside the scaffold (20 day after printing, no MSP) whereas 4% gelatin (B) shows neurite outgrowth inside the scaffold (20 days, no MSP) and even more extensive neurite outgrowth inside the scaffold when treated with MSP (C) (20 days, MSP treated). The fibers extensions further increased in MSP treated scaffolds on 30 days after 3D-printing (D) in 4% gelatin scaffold (30 days, MSP treated).
At day 30, at the end of the experiment, we detected extensive neurite outgrowth at all levels within the scaffolds treated with MSPs (Figures 7C and D; Figure 8).
4. Discussion
In this study, we developed a 3D printing bioink and protocol that supports neuron viability and promotes neurite outgrowth throughout a 300 µm thick scaffold, which remains structurally stable for at least 1 month in culture. Over this period, the cultured neurons demonstrated significant increase in neurite outgrowth and expressed ChAT, the enzyme responsible for synthesizing acetylcholine. ChAT expression is characteristic of an MN phenotype, though it is possible that other ChAT-positive neuron types, such as spinal interneurons, are also present. Interestingly, neural progenitors printed in the bioink under identical conditions matured into MNs with longterm survival only when predifferentiated as monolayers, whereas those predifferentiated as neurospheres did not survive. These observations underscore the critical influence of pre-printing culture conditions on MN survival and differentiation in 3D-printed scaffolds.
In a previous study, MNs extended neurites only along the surface of scaffolds printed using 12% gelatin bioink.13 Here, we demonstrated that reducing the gelatin concentration to 4%, carefully regulating thermal parameters on the printing bed, and promptly transferring the scaffold into the culture medium post-printing enabled robust neurite outgrowth throughout the entire scaffold. This highlights the importance of bioink optimization for achieving extensive 3D MN integration.
3D-printed scaffolds provide both chemical and physical cues essential for the survival and differentiation of embedded cells.24–26 Gelatin is an ideal bioink component due to its biocompatibility, biodegradability, low toxicity, and hydrogel properties, which allow for creation of a porous matrix facilitating nutrient and bioactive agent diffusion.26 While chemical factors are well-established mediators of neural differentiation, recent studies have emphasized the significance of physical properties, such as scaffold stiffness.25,27–29 To evaluate the effect of gelatin concentration on the mechanical properties of bioinks, we examined the stiffness of scaffolds prepared using two methods (M1 and M2). We adjusted the temperature during the ink preparation and printing process to improve the printability of low-concentration gelatin and maintained the stability of the printed structure by temporarily covering the scaffolds with a high concentration of the crosslinker. Rheology tests revealed that the stiffness of 4% gelatin bioink prepared with M2 was tuned to 0.5 kPa (Figure 2B), a value conducive to MN differentiation. These findings align with previous research showing that optimal scaffold stiffness for neurons is <1 kPa, whereas higher stiffness values favor differentiation into other cell types like oligodendrocytes (~7 kPa) and astrocytes (1–3 kPa).24
Unexpectedly, the stiffness of 10% gelatin bioink prepared using M1 was lower than that of the 4% gelatin bioink prepared with M2 (Figure 3B). This discrepancy may be attributed to water vapor condensation during M1 processing (Figure S3, Supporting Information), which was subsequently absorbed by the scaffold. Increased scaffold softness facilitated culture medium penetration throughout the structure, although it did not ensure that differentiation factors reached deeper cells. To address this, we incorporated MSPs loaded with differentiation factors directly into the bioink before printing.
We previously demonstrated that MSPs can sustain the release of differentiation factors promoting MN differentiation both in vitro and after in vivo implantation.19,20 In this study, MSPs loaded with ciliary neurotrophic factor (CNTF) and glial-derived neurotrophic factor (GDNF) peptide mimetics (Cintrofin and Gliafin) were uniformly distributed within the scaffolds, ensuring consistent access to differentiation factors. This approach significantly advanced neurite outgrowth inside the scaffold and increased the proportion of differentiated ChAT-positive MNs. These results demonstrate the utility of MSPs in bridging the gap between traditional in vitro differentiation techniques and 3D bioprinting by virtue of its potential to provide sustained, localized delivery of differentiation factors and mimic in vivo conditions through enhancing MN survival and differentiation. Notably, extensive neurite outgrowth was observed even in 4% gelatin scaffolds without MSPs, although survival declined at day 20. This suggests that additional approaches could be explored to sustain long-term MN differentiation and survival under these conditions.
The choice of bioink and its processing parameters is critical for successful 3D bioprinting of spinal cord tissue. Hydrogels, including gelatin-based bioinks, are particularly attractive for this purpose due to their biocompatibility and modifiable mechanical properties, which can accommodate various cell types and tissue requirements.30–32 Gelatinbased hydrogels mimic natural extracellular matrix and support cellular functions such as proliferation, differentiation, and synaptic connectivity.15,26,33
Crosslinking is essential for obtaining reproducible, stable, and biocompatible gelatin-based scaffolds. Physical, enzymatic, and chemical methods can be used to achieve this.34,35 Physical methods include electron beam irradiation, γ-irradiation,36 and dehydrothermal treatment.37 Enzymatic methods involve the use of transglutaminase to covalently crosslink glutamine and lysine residues.38,39 Chemical methods40–44 offer greater control over the physical properties of hydrogels but may introduce cytotoxicity risks due to residual crosslinking agents.45 Natural crosslinkers, such as genipin, citric acid, and tannic acid, offer lower toxicity alternatives.46 Photo-crosslinkable gelatin derivatives, such as methacryloyl gelatin (GelMA), allow cell encapsulation during crosslinking but may face challenges related to photoinitiator toxicity and UV light penetration.47,48 However, the potential cytotoxicity of photoinitiator, in the context of homogenous light depth penetration and application of UV light, might affect the homogeneity of the hydrogel properties and cell viability, prompting investigations of visible light-based crosslinking strategies.49,50 Recent developments in bio-orthogonal crosslinking strategies using functionalized gelatin derivatives offer promising, cell-friendly alternatives for hydrogel fabrication51–56 and can be tested in the future for incorporation of other cell types, to achieve optimal MN differentiation and functional spinal cord organoid.
The findings from this study demonstrate the critical role of bioink composition, scaffold properties, and differentiation factor delivery in promoting motor neuron survival, differentiation, and neurite outgrowth in 3D-printed scaffolds. By leveraging optimized bioink formulations and integrating MSP-based delivery systems, we bridge the gap between traditional in vitro differentiation approaches and advanced 3D bioprinting methods. These advancements not only improve our ability to replicate neural tissue conditions but also lay the groundwork for generating multicellular disease-specific models. By applying this protocol to iPSC-derived neural progenitors from patients with MN diseases such as amyotrophic lateral sclerosis (ALS), we can take the next step toward creating 3D-printed neural tissues for disease modeling, therapeutic screening, and regenerative medicine.57
5. Conclusion
In this study, we developed a 3D printing bioink and protocol that supports neuron viability and promote neurite outgrowth within a 300 µm scaffold layer, maintaining structural stability for at least 1 month in culture. We demonstrated that optimized bioink formulations, including reduced gelatin concentration and precise thermal regulation during printing, significantly enhance 3D MN integration. Additionally, we highlighted the importance of pre-printing culture conditions, showing that neural progenitors predifferentiated as monolayers exhibited long-term MN survival, while those predifferentiated as neurospheres did not.
Our findings emphasize the crucial role of scaffold stiffness in neural differentiation, with rheological analyses confirming that a 4% gelatin bioink tuned to 0.5 kPa provides an optimal environment for MN maturation. Moreover, incorporating MSPs loaded with differentiation factors facilitated sustained MN differentiation and enhanced neurite outgrowth, bridging the gap between traditional in vitro differentiation methods and 3D bioprinting approaches.
This study underscores the potential of bioengineered 3D-printed scaffolds to mimic in vivo conditions, providing a stable, biocompatible environment for MN differentiation and survival. The integration of optimized bioink formulations and controlled differentiation factor delivery paves the way for the development of advanced multicellular neural tissue models. By applying this protocol to iPSC-derived neural progenitors from patients with MN diseases such as ALS, we move closer to creating 3D-printed neural tissues for disease modeling, therapeutic screening, and regenerative medicine.
1.Pașca SP, Arlotta P, Bateup HS, et al. A nomenclature consensus for nervous system organoids and assembloids. Nature. 2022;609(7929):907-910.
doi: 10.1038/s41586-022-05219-6
2.Birtele M, Lancaster M, Quadrato G. Modelling human brain development and disease with organoids. Nat Rev Mol Cell Biol. 2024;1-24.
doi: 10.1038/s41580-024-00804-1.
3.Yan Y, Li X, Gao Y, et al. 3D bioprinting of human neural tissues with functional connectivity. Cell Stem Cell. 2024;31(2):260-274.
doi: 10.1016/j.stem.2023.12.009
4.Willerth SM. Bioprinting functional neural networks. Cell Stem Cell. 2024;31(2):151-152.
doi: 10.1016/j.stem.2023.12.014
5.Bock M, Hong SJ, Zhang S, et al. Morphogenetic designs, and disease models in central nervous system organoids. Int J Mol Sci. 2024;25(14):7750.
doi: 10.3390/ijms25147750
6.Ma L, Zhang Z, Mu Y, Liu B, Zhou H, Wang D. The application of biomaterial‐based spinal cord tissue engineering. Macromol Biosci. 2024:e2400444.
7.Huang MS, Christakopoulos F, Roth JG, Heilshorn SC. Organoid bioprinting: from cells to functional tissues. Nat Rev Bioeng. 2024;3:1-17.
doi: 10.1038/s44222-024-00268-0
8.Rodriguez-Palomo A, Lutz-Bueno V, Guizar-Sicairos M, Kádár R, Andersson M, Liebi M. Nanostructure and anisotropy of 3D printed lyotropic liquid crystals studied by scattering and birefringence imaging. Addit Manuf. 2021;47:102289.
doi: 10.1016/j.addma.2021.102289
9.Rodriguez‐Palomo A, Lutz‐Bueno V, Cao X, Kádár R, Andersson M, Liebi M. In situ visualization of the structural evolution and alignment of lyotropic liquid crystals in confined flow. Small. 2021;17(7):2006229.
10.Rau DA, Williams CB, Bortner MJ. Rheology and printability: a survey of critical relationships for direct ink write materials design. Prog Mater Sci. 2023;140:101188.
doi: 10.1016/j.pmatsci.2023.101188
11.Pașca SP, Arlotta P, Bateup HS, et al. A framework for neural organoids, assembloids and transplantation studies. Nature. 2024:1–3.
doi: 10.1038/s41586-024-08487-6
12.Scarian E, Bordoni M, Fantini V, et al. Patients’ stem cells differentiation in a 3D environment as a promising experimental tool for the study of amyotrophic lateral sclerosis. Int J Mol Sci. 2022;23(10):5344.
doi: 10.3390/ijms23105344
13.Han Y, King M, Tikhomirov E, et al. Towards 3D bioprinted spinal cord organoids. Int J Mol Sci. 2022; 23(10):5788.
doi: 10.3390/ijms23105788
14.Young AT, White OC, Daniele MA. Rheological properties of coordinated physical gelation and chemical crosslinking in gelatin methacryloyl (GelMA) hydrogels. Macromol Biosci. 2020;20(12):e2000183.
15.Asim S, Tabish TA, Liaqat U, Ozbolat IT, Rizwan M. Advances in gelatin bioinks to optimize bioprinted cell functions. Adv Healthc Mater. 2023;12(17):e2203148.
16.Yuan X, Zhu Z, Xia P, et al. Tough gelatin hydrogel for tissue engineering. Adv Sci (Weinh). 2023;10(24):e2301665.
17.Elosegui-Artola A, Gupta A, Najibi AJ, et al. Matrix viscoelasticity controls spatiotemporal tissue organization. Nat Mater. 2023;22(1):117-127.
doi: 10.1038/s41563-022-01400-4
18.Chen X, Liu C, McDaniel G, et al. Viscoelasticity of hyaluronic acid hydrogels regulates human pluripotent stem cell‐derived spinal cord organoid patterning and vascularization. Adv Healthc Mater. 2024;13(32):e2402199.
19.Garcia-Bennett AE, König N, Abrahamsson N, et al. In vitro generation of motor neuron precursors from mouse embryonic stem cells using mesoporous nanoparticles. Nanomedicine. 2014;9(16):2457-2466.
doi: 10.2217/nnm.14.23
20.Garcia-Bennett AE, Kozhevnikova M, König N, et al. Delivery of differentiation factors by mesoporous silica particles assists advanced differentiation of transplanted murine embryonic stem cells. Stem Cells Transl Med. 2013;2(11):906-915.
21.Aldskogius H, Berens C, Kanaykina N, et al. Regulation of boundary cap neural crest stem cell differentiation after transplantation. Stem Cells. 2009;27(7):1592-1603.
doi: 10.1002/stem.77
22.Rønn LC, Ralets I, Hartz BP, et al. A simple procedure for quantification of neurite outgrowth based on stereological principles. J Neurosci Methods. 2000;100(1-2):25-32.
doi: 10.1016/s0165-0270(00)00228-4
23.Normand V, Muller S, Ravey JC, Parker A. Gelation kinetics of gelatin: a master curve and network modeling. Macromolecules. 2000;33(3):1063-1071.
doi: 10.1021/ma9909455
24.Thompson R, Chan C. Signal transduction of the physical environment in the neural differentiation of stem cells. Technology (Singap World Sci). 2016;4(1):1-8.
doi: 10.1142/S2339547816400070
25.Tringides CM, Boulingre M, Khalil A, Lungjangwa T, Jaenisch R, Mooney DJ. Tunable conductive hydrogel scaffolds for neural cell differentiation. Adv Healthc Mater. 2023;12(7):e2202221.
26.Layrolle P, Payoux P, Chavanas S. Message in a scaffold: natural biomaterials for three-dimensional (3D) bioprinting of human brain organoids. Biomolecules. 2022;13(1):25.
doi: 10.3390/biom13010025
27.Zhang W, Chu G, Wang H, Chen S, Li B, Han F. Effects of matrix stiffness on the differentiation of multipotent stem cells. Curr Stem Cell Res Ther. 2020;15(5):449-461.
doi: 10.2174/1574888X15666200408114632
28.Castillo Ransanz L, Van Altena PF, Heine VM, Accardo A. Engineered cell culture microenvironments for mechanobiology studies of brain neural cells. Front Bioeng Biotechnol. 2022;10:1096054.
doi: 10.3389/fbioe.2022.1096054
29.Roth JG, Huang MS, Navarro RS, Akram JT, LeSavage BL, Heilshorn SC. Tunable hydrogel viscoelasticity modulates human neural maturation. Sci Adv. 2023;9(42):eadh8313.
30.Cruz EM, Machado LS, Zamproni LN, et al. A gelatin methacrylate-based hydrogel as a potential bioink for 3D bioprinting and neuronal differentiation. Pharmaceutics. 2023;15(2):627.
doi: 10.3390/pharmaceutics15020627
31.Kwokdinata C, Ramanujam V, Chen J, et al. Encapsulation of human spinal cord progenitor cells in hyaluronan-gelatin hydrogel for spinal cord injury treatment. ACS Appl Mater Interfaces. 2023;15(44):50679-50692.
32.Wang P, Qian L, Liang H, et al. A polyvinyl alcohol/ acrylamide hydrogel with enhanced mechanical properties promotes full-thickness skin defect healing by regulating immunomodulation and angiogenesis through paracrine secretion. Engineering. 2024;37:138-151.
doi: 10.1016/j.eng.2024.02.005
33.Lukin I, Erezuma I, Maeso L, et al. Progress in gelatin as biomaterial for tissue engineering. Pharmaceutics. 2022;14(6):1177.
doi: 10.3390/pharmaceutics14061177
34.Campiglio CE, Contessi Negrini N, Farè S, Draghi L. Crosslinking strategies for electrospun gelatin scaffolds. Materials (Basel). 2019;12(15):2476.
doi: 10.3390/ma12152476
35.Van Vlierberghe S. Crosslinking strategies for porous gelatin scaffolds. J Mater Sci. 2016;51(9):4349-4357.
doi: 10.1007/s10853-016-9747-4
36.Cataldo F, Ursini O, Lilla E, Angelini G. Radiation-induced crosslinking of collagen gelatin into a stable hydrogel. J Radioanal Nucl Chem. 2008;275(1):125-131.
doi: 10.1007/s10967-007-7003-8
37.Omata K, Matsuno T, Asano K, Hashimoto Y, Tabata Y, Satoh T. Enhanced bone regeneration by gelatin–β‐tricalcium phosphate composites enabling controlled release of bFGF. J Tissue Eng Regen Med. 2014;8(8):604-611.
doi: 10.1002/term.1553
38.Dinh TN, Hou S, Park S, Shalek BA, Jeong KJ. Gelatin hydrogel combined with polydopamine coating to enhance tissue integration of medical implants. ACS Biomater Sci Eng. 2018;4(10):3471-3477.
doi: 10.1021/acsbiomaterials.8b00886
39.Wen C, Lu L, Li X. Mechanically robust gelatin–a lginate IPN hydrogels by a combination of enzymatic and ionic crosslinking approaches. Macromol Mater Eng. 2014;299(4):504-513.
40.Campiglio CE, Ponzini S, De Stefano P, Ortoleva G, Vignati L, Draghi L. Cross-linking optimization for electrospun gelatin: challenge of preserving fiber topography. Polymers (Basel). 2020;12(11):2472.
41.Esteves C, Santos GM, Alves C, et al. Effect of film thickness in gelatin hybrid gels for artificial olfaction. Materials Today Bio. 2019;1:100002.
doi: 10.1016/j.mtbio.2019.100002
42.Negrini NC, Bonnetier M, Giatsidis G, Orgill DP, Farè S, Marelli B. Tissue-mimicking gelatin scaffolds by alginate sacrificial templates for adipose tissue engineering. Acta Biomater. 2019;87:61-75.
doi: 10.1016/j.actbio.2019.01.018
43.Negrini NC, Celikkin N, Tarsini P, Farè S, Święszkowski W. Three-dimensional printing of chemically crosslinked gelatin hydrogels for adipose tissue engineering. Biofabrication. 2020;12(2):025001.
doi: 10.1088/1758-5090/ab56f9.
44.Sarem M, Arya N, Heizmann M, et al. Interplay between stiffness and degradation of architectured gelatin hydrogels leads to differential modulation of chondrogenesis in vitro and in vivo. Acta Biomater. 2018;69:83-94.
doi: 10.1016/j.actbio.2018.01.025
45.Yang G, Xiao Z, Long H, et al. Assessment of the characteristics and biocompatibility of gelatin sponge scaffolds prepared by various crosslinking methods. Sci Rep. 2018;8(1):1616.
doi: 10.1038/s41598-018-20006-y
46.Sapuła P, Bialik-Wąs K, Malarz K. Are natural compounds a promising alternative to synthetic cross-linking agents in the preparation of hydrogels? Pharmaceutics. 2023;15(1):253.
doi: 10.3390/pharmaceutics15010253
47.Kerscher P, Kaczmarek JA, Head SE, et al. Direct production of human cardiac tissues by pluripotent stem cell encapsulation in gelatin methacryloyl. ACS Biomater Sci Eng. 2017;3(8):1499-1509.
doi: 10.1021/acsbiomaterials.6b00226
48.Van Hoorick J, Tytgat L, Dobos A, et al. (Photo-) crosslinkable gelatin derivatives for biofabrication applications. Acta Biomater. 2019;97:46-73.
doi: 10.1016/j.actbio.2019.07.035
49.Lim WL, Chowdhury SR, Ng MH, Law JX. Physicochemical properties and biocompatibility of electrospun polycaprolactone/gelatin nanofibers. Int J Environ Res Public Health. 2021;18(9):4764.
50.Nguyen AK, Goering PL, Elespuru RK, Sarkar Das S, Narayan RJ. The photoinitiator lithium phenyl (2,4,6-Trimethylbenzoyl) phosphinate with exposure to 405 nm light is cytotoxic to mammalian cells but not mutagenic in bacterial reverse mutation assays. Polymers. 2020;12(7):1489.
51.Sletten EM, Bertozzi CR. From mechanism to mouse: a tale of two bioorthogonal reactions. Acc Chem Res. 2011;44(9):666-676.
doi: 10.1021/ar200148z
52.Arkenberg MR, Nguyen HD, Lin CC. Recent advances in bio-orthogonal and dynamic crosslinking of biomimetic hydrogels. J Mater Chem B. 2020;8(35):7835-7855.
doi: 10.1039/d0tb01429j.
53.Koshy ST, Zhang DK, Grolman JM, Stafford AG, Mooney DJ. Injectable nanocomposite cryogels for versatile protein drug delivery. Acta Biomater. 2018;65:36-43.
doi: 10.1016/j.actbio.2017.11.024
54.Contessi Negrini N, Angelova Volponi A, Sharpe PT, Celiz AD. Tunable cross-linking and adhesion of gelatin hydrogels via bioorthogonal click chemistry. ACS Biomater Sci Eng. 2021;7(9):4330-4346.
55.Tytgat L, Markovic M, Qazi TH, et al. Photo-crosslinkable recombinant collagen mimics for tissue engineering applications. J Mater Chem B. 2019;7(19):3100-3108.
doi: 10.1039/C8TB03308K
56.Lin Y, Du H, Roos Y, Miao S. Transglutaminase crosslinked fish gelatins for emulsion stabilization: from conventional to Pickering emulsions. Food Hydrocolloids. 2023; 144:108979.
doi: 10.1016/j.foodhyd.2023.108979
57.Buchner F, Dokuzluoglu Z, Grass T, Rodriguez-Muela N. Spinal cord organoids to study motor neuron development and disease. Life (Basel). 2023;13(6):1254.
doi: 10.3390/life13061254

