Optimizing printability and mechanical properties of poly(3-hydroxybutyrate) biocomposite blends and their biological response to Saos-2 cells

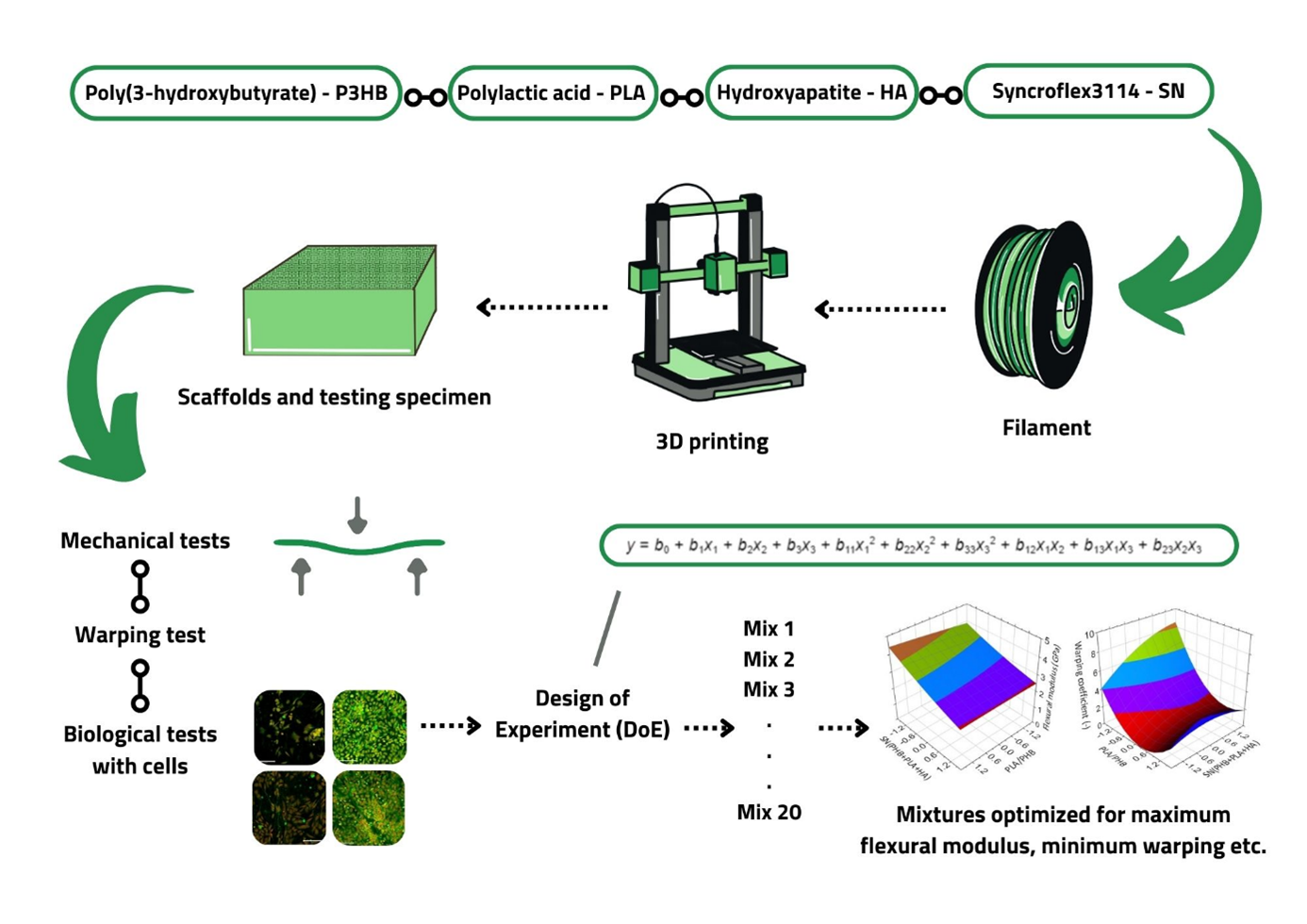
Bone tissue engineering requires scaffolds with three-dimensional (3D) structures that facilitate vascularization and new tissue growth. 3D printing, especially through fused deposition modeling (FDM), has emerged as an effective method for creating complex structures with high reproducibility. Early research in this area demonstrated the potential of poly(ε-caprolactone) (PCL) and poly(L-lactide) (PLLA) scaffolds for bone regeneration. Recently, polylactide (PLA) and polyhydroxyalkanoates (PHAs) have garnered attention for their biocompatibility and ability to support cell proliferation. Among PHAs, poly(3-hydroxybutyrate) (PHB) shows promise due to its intrinsic biocompatibility and resorbability, making it a candidate for FDM-based scaffold fabrication. In the presented study, we aim to develop and optimize a biocompatible PHB-based composite material for bone tissue engineering, incorporating PLA, hydroxyapatite, and the plasticizer Syncroflex 3114 to enhance mechanical properties and printability. This composite was processed into filaments for 3D printing and characterized through thermal, mechanical, and biological evaluations. Using a design of experiment approach, we investigated factors such as temperature performance, warping, degradation, and strength to determine the optimal composition for use in tissue engineering. Four optimal mixture compositions fulfilling the optimization criteria of having the most suitable properties for bone tissue engineering, namely the best printability and maximum mechanical properties, were obtained. The mixtures were optimized specifically for minimum warping coefficient (0.5); maximum flexural strength (66.9 MPa); maximum compression modulus (2.4 GPa); and maximum compression modulus (2.3 GPa) with a warping coefficient of no more than 1 at the same time. In conclusion, the study shows a new possible way to effectively develop and test 3D-printed PHB-based scaffolds with specifically optimized material properties.
- Abbasi N, Hamlet S, Love RM, Nguyen N.-T. Porous scaffolds for bone regeneration. J Sci Adv Mater Devices. 2020;5(1):1-9. doi: 10.1016/j.jsamd.2020.01.007
- Ngo TD, Kashani A, Imbalzano G, Nguyen KTQ, Hui D. Additive manufacturing (3D printing): a review of materials, methods, applications and challenges. Compos B Eng. 2018;143:172-196. doi: 10.1016/j.compositesb.2018.02.012
- Zhang Q, Zhou J, Zhi P, et al. 3D printing method for bone tissue engineering scaffold. Med Nov Technol Devices. 2023;17:100205. doi: 10.1016/j.medntd.2022.100205
- Bose S, Vahabzadeh S, Bandyopadhyay A. Bone tissue engineering using 3D printing. Mater Today. 2013;16(12):496-504. doi: 10.1016/j.mattod.2013.11.017
- Chiulan I, Frone AN, Brandabur C, Panaitescu DM. Recent advances in 3D printing of aliphatic polyesters. Bioeng Basel Switz 2017;5(1):E2. doi: 10.3390/bioengineering5010002
- Shunmugasundaram M, Baig M, Madala A. A review of bio-degradable materials for fused deposition modeling machine. Mater Today Proc. 2020;27:1596-1600. doi: 10.1016/j.matpr.2020.03.267
- Mehrpouya M, Vahabi H, Barletta M, Laheurte P, Langlois V. Additive manufacturing of polyhydroxyalkanoates (PHAs) biopolymers: materials, printing techniques, and applications. Mater Sci Eng C Mater Biol Appl. 2021;127:112216. doi: 10.1016/j.msec.2021.112216
- Amrita, Manoj A, Panda RC. Biodegradable filament for 3D printing process: a review. Eng Sci. 2022;18(7):11-19. doi: 10.30919/es8d616
- Rigotti D, Pegoretti A. Additive manufacturing with biodegradable polymers. In: Biodegradable Polymers, Blends and Composites. Woodhead Publishing; 2022:611-679. doi: 10.1016/B978-0-12-823791-5.00026-0.
- Hutmacher DW, Schantz T, Zein I, Ng KW, Teoh SH, Tan KC. Mechanical properties and cell cultural response of polycaprolactone scaffolds designed and fabricated via fused deposition modeling. J Biomed Mater Res. 2001;55(2):203-216. doi: 10.1002/1097-4636(200105)55:2<203::AID-JBM1007>3.0.CO;2-7
- Xiong Z, Yan Y, Zhang R, Sun L. Fabrication of porous poly(l-Lactic Acid) scaffolds for bone tissue engineering via precise extrusion. Scr Mater. 2001;45(7):773-779. doi: 10.1016/S1359-6462(01)01094-6.
- Zein I, Hutmacher DW, Tan KC, Teoh SH. Fused deposition modeling of novel scaffold architectures for tissue engineering applications. Biomaterials. 2002;23(4):1169-1185. doi: 10.1016/S0142-9612(01)00232-0
- Cao T, Ho K-H, Teoh S-H. Scaffold design and in vitro study of osteochondral coculture in a three-dimensional porous polycaprolactone scaffold fabricated by fused deposition modeling. Tissue Eng. 2003;9(Suppl 1):S103-S112. doi: 10.1089/10763270360697012
- Park S, Kim G, Jeon YC, Koh Y, Kim W. 3D polycaprolactone scaffolds with controlled pore structure using a rapid prototyping system. J Mater Sci Mater Med. 2009;20(1):229-234. doi: 10.1007/s10856-008-3573-4
- Domingos M, Chiellini F, Gloria A, Ambrosio L, Bartolo P, Chiellini E. Effect of process parameters on the morphological and mechanical properties of 3D bioextruded poly(Ε‐caprolactone) scaffolds. Rapid Prototyp J. 2012;18(1): 56-67. doi: 10.1108/13552541211193502
- Korpela J, Kokkari A, Korhonen H, Malin M, Närhi T, Seppälä J. Biodegradable and bioactive porous scaffold structures prepared using fused deposition modeling. J Biomed Mater Res B Appl Biomater. 2013;101B(4):610-619. doi: 10.1002/jbm.b.32863
- Rai B, Teoh SH, Ho KH, et al. The effect of rhBMP-2 on canine osteoblasts seeded onto 3D bioactive polycaprolactone scaffolds. Biomaterials. 2004;25(24):5499-5506. doi: 10.1016/j.biomaterials.2004.01.007
- Rai B, Lin JL, Lim ZXH, Guldberg RE, Hutmacher DW, Cool SM. Differences between in vitro viability and differentiation and in vivo bone-forming efficacy of human mesenchymal stem cells cultured on PCL–TCP scaffolds. Biomaterials. 2010;31(31):7960-7970. doi: 10.1016/j.biomaterials.2010.07.001
- Teo EY, Ong S-Y, Chong MSK, et al. Polycaprolactone-based fused deposition modeled mesh for delivery of antibacterial agents to infected wounds. Biomaterials. 2011;32(1):279–287. doi: 10.1016/j.biomaterials.2010.08.089
- Konopnicki S, Sharaf B, Resnick C., et al. Tissue-engineered bone with 3-dimensionally printed β-tricalcium phosphate and polycaprolactone scaffolds and early implantation: an in vivo pilot study in a porcine mandible model. J Oral Maxillofac Surg. 2015;73(5):1016.e1-1016.e11. doi: 10.1016/j.joms.2015.01.021
- Dávila JL, Freitas MS, Inforçatti Neto P, Silveira ZC, Silva JVL, d’Ávila MA. Fabrication of PCL/β-TCP scaffolds by 3d mini-screw extrusion printing. J Appl Polym Sci. 2016;133(15):1-9. doi: 10.1002/app.43031
- Sparks DS, Wiper J, Lloyd T, et al. Protocol for the BONE-RECON trial: a single-arm feasibility trial for critical sized lower limb BONE defect RECONstruction using the mPCL-TCP scaffold system with autologous vascularised corticoperiosteal tissue transfer. BMJ Open. 2023;13(5):e056440. doi: 10.1136/bmjopen-2021-056440
- Next-level tissue regeneration. Biomendex. https:// biomendex.com/(accessed 2024-09-19).
- Chen X, Chen G, Wang G, Zhu P, Gao C. Recent progress on 3D-printed polylactic acid and its applications in bone repair. Adv Eng Mater. 2020;22(4):1901065. doi: 10.1002/adem.201901065
- Gregor A, Filová E, Novák M, et al. Designing of PLA scaffolds for bone tissue replacement fabricated by ordinary commercial 3D printer. J Biol Eng. 2017;11:31. doi: 10.1186/s13036-017-0074-3
- Wurm MC, Möst T, Bergauer B, et al. In-vitro evaluation of polylactic acid (PLA) manufactured by fused deposition modeling. J Biol Eng. 2017;11(1):29. doi: 10.1186/s13036-017-0073-4
- Germain L, Fuentes CA, van Vuure AW, Rieux Ad, Dupont- Gllain C. 3D-printed biodegradable gyroid scaffolds for tissue engineering applications. Mater Des. 2018;151:113-122. doi: 10.1016/j.matdes.2018.04.037
- Alizadeh-Osgouei M, Li Y, Vahid A, Ataee A, Wen C. High strength porous PLA gyroid scaffolds manufactured via fused deposition modeling for tissue-engineering applications. Smart Mater Med. 2021;2:15-25. doi: 10.1016/j.smaim.2020.10.003
- Eryildiz M. Fabrication of drug-loaded 3D-printed bone scaffolds with radial gradient porosity. J Mater Eng Perform. 2023;32(9):4249-4257. doi: 10.1007/s11665-022-07490-0
- Schoen AH. Infinite Periodic Minimal Surfaces Without Self-Intersections. National Aeronautics and Space Administration; 1970.
- Xiong Z, Yan Y, Wang S, Zhang R, Zhnag C. Fabrication of porous scaffolds for bone tissue engineering via low-temperature deposition. Scr Mater. 2002;46(11): 771-776. doi: 10.1016/S1359-6462(02)00071-4
- Drummer D, Cifuentes‐Cuéllar S, Rietzel D. Suitability of PLA/TCP for fused deposition modeling. Rapid Prototyp J. 2012;18(6):500-507. doi: 10.1108/13552541211272045
- Elhattab K, Bhaduri SB, Sikder P. Influence of fused deposition modelling nozzle temperature on the rheology and mechanical properties of 3D printed β-tricalcium phosphate (TCP)/polylactic acid (PLA) composite. Polymers. 2022;14(6):1222. doi: 10.3390/polym14061222
- Distler T, Fournier N, Grünewald A, et al. Polymer-bioactive glass composite filaments for 3D scaffold manufacturing by fused deposition modeling: fabrication and characterization. Front Bioeng Biotechnol. 2020;8:552. doi: 10.3389/fbioe.2020.00552
- Matsuo A, Chiba H, Takahashi H, Toyoda J, Abukawa H. Clinical application of a custom-made bioresorbable raw particulate hydroxyapatite/poly-l-lactide mesh tray for mandibular reconstruction. Odontology. 2010;98(1): 85-88. doi: 10.1007/s10266-009-0111-x
- Senatov FS, Niaza KV, Stepashkin AA, Kaloshkin SD. Low-cycle fatigue behavior of 3d-printed PLA-based porous scaffolds. Compos Part B Eng. 2016;97:193-200. doi: 10.1016/j.compositesb.2016.04.067
- Esposito Corcione C, Gervaso F, Scalera F, Montagna F, Sannino A, Maffezzoli A. The feasibility of printing polylactic acid–nanohydroxyapatite composites using a low-cost fused deposition modeling 3D printer. J Appl Polym Sci. 2017;134(13);44656. doi: 10.1002/app.44656
- Esposito Corcione C, Scalera F, Gervaso F, Montagna F, Sannino A, Maffezzoli A. One-step solvent-free process for the fabrication of high loaded PLA/HA composite filament for 3D printing. J Therm Anal Calorim. 2018;134(1): 575-582. doi: 10.1007/s10973-018-7155-5
- Esmaeili S, Akbari Aghdam H, Motififard M, et al. A porous polymeric–hydroxyapatite scaffold used for femur fractures treatment: fabrication, analysis, and simulation. Eur J Orthop Surg Traumatol. 2020;30(1):123-131. doi: 10.1007/s00590-019-02530-3
- Dubinenko G, Zinoviev A, Bolbasov E, et al. Highly filled poly(l-lactic acid)/hydroxyapatite composite for 3D printing of personalized bone tissue engineering scaffolds. J Appl Polym Sci. 2021;138(2):49662. doi: 10.1002/app.49662
- Alves AA, Siqueira EC, Barros MPS, Silva PEC, Houllou LM. Polyhydroxyalkanoates: a review of microbial production and technology application. Int J Environ Sci Technol. 2023;20(3):3409-3420. doi: 10.1007/s13762-022-04213-9
- Kosorn W, Sakulsumbat M, Uppanan P, et al. PCL/PHBV blended three dimensional scaffolds fabricated by fused deposition modeling and responses of chondrocytes to the scaffolds. J Biomed Mater Res B Appl Biomater. 2017;105(5):1141-1150. doi: 10.1002/jbm.b.33658
- Ausejo JG, Rydz J, Musiol M, et al. A comparative study of three-dimensional printing directions: the degradation and toxicological profile of a PLA/PHA blend. Polym Degrad Stab. 2018;152:191-207. doi: 10.1016/j.polymdegradstab.2018.04.024
- Findrik Balogová A, Hudák R, Tóth T, et al. Determination of geometrical and viscoelastic properties of PLA/PHB samples made by additive manufacturing for urethral substitution. J Biotechnol. 2018;284:123-130. doi: 10.1016/j.jbiotec.2018.08.019
- Ecker JV, Burzic I, Haider A, Hild S, Rennhofer H. Improving the impact strength of PLA and its blends with PHA in fused layer modelling. Polym Test. 2019;78:105929. doi: 10.1016/j.polymertesting.2019.105929
- Rydz J, Włodarczyk J, Gonzalez Ausejo J, et al. Three-dimensional printed PLA and PLA/PHA dumbbell-shaped specimens: material defects and their impact on degradation behavior. Materials. 2020;13(8):2005. doi: 10.3390/ma13082005
- Findrik Balogová A, Trebuňová M, Ižaríková G, et al. In vitro degradation of specimens produced from PLA/PHB by additive manufacturing in simulated conditions. Polymers. 2021;13(10):1542. doi: 10.3390/polym13101542
- Gielisch M, Heimes D, Thiem DGE, et al. Steam-sterilized and degradable fused filament fabrication-printed polylactide/ polyhydroxyalkanoate surgical guides for dental implants: are they accurate enough for static navigation? Int J Bioprint. 2022;9(2):655. doi: 10.18063/ijb.v9i2.655
- Kohan M, Lancoš S, Schnitzer M, Živčák J, Hudák R. Analysis of PLA/PHB biopolymer material with admixture of hydroxyapatite and tricalcium phosphate for clinical use. Polymers. 2022;14(24):5357. doi: 10.3390/polym14245357
- Giubilini A, Siqueira G, Clemens FJ, et al. 3D-printing nanocel lul o s e-poly(3-hydroxybutyr ate-Co-3- hydroxyhexanoate) biodegradable composites by fused deposition modeling. ACS Sustain Chem Eng. 2020;8(27):10292-10302. doi: 10.1021/acssuschemeng.0c03385
- Kovalcik A, Sangroniz L, Kalina M, et al. Properties of scaffolds prepared by fused deposition modeling of poly(hydroxyalkanoates). Int J Biol Macromol. 2020;161:364-376. doi: 10.1016/j.ijbiomac.2020.06.022
- Dong X, Premaratne ID, Sariibrahimoglu K, Limem S, Scott J, Gadjiko M. 3D-printed poly-4-hydroxybutyrate bioabsorbable scaffolds for nipple reconstruction. Acta Biomater. 2022;143:333-343. doi: 10.1016/j.actbio.2022.02.040
- Volpini V, Giubilini A, Corsi L, Nobili A, Bondioli F. Characterization of biocompatible scaffolds manufactured by fused filament fabrication of poly(3-Hydroxybutyrate- Co-3-Hydroxyhexanoate). R Soc Open Sci. 2022;9(4): 211485. doi: 10.1098/rsos.211485
- Ivorra-Martinez J, Peydro MÁ, Gomez-Caturla J, Sanchez- Nacher L, Boronat T, Balart R. The effects of processing parameters on mechanical properties of 3D-printed polyhydroxyalkanoates parts. Virtual Phys Prototyp. 2023;18(1):e2164734. doi: 10.1080/17452759.2022.2164734
- Aráoz B, Karakaya E, Wusener AG, et al. 3D printed poly(Hydroxybutyrate-Co-Hydroxyvalerate)—45S5 bioactive glass composite resorbable scaffolds suitable for bone regeneration. J Mater Res. 2021;36:4000-4012. doi: 10.1557/s43578-021-00272-9
- Ye X, Zhang Y, Liu T, et al. Beta-tricalcium phosphate enhanced mechanical and biological properties of 3D-printed polyhydroxyalkanoates scaffold for bone tissue engineering. Int J Biol Macromol. 2022;209:1553-1561. doi: 10.1016/j.ijbiomac.2022.04.056
- Chen G-Q, Wu Q. The application of polyhydroxyalkanoates as tissue engineering materials. Biomaterials. 2005;26(33):6565-6578. doi: 10.1016/j.biomaterials.2005.04.036
- Lim J, You M, Li J, Li Z. Emerging bone tissue engineering via polyhydroxyalkanoate (PHA)-based scaffolds. Mater Sci Eng C Mater Biol Appl. 2017;79:917-929. doi: 10.1016/j.msec.2017.05.132
- Butt FI, Muhammad N, Hamid A, Moniruzzaman M, Sharif F. Recent progress in the utilization of biosynthesized polyhydroxyalkanoates for biomedical applications - review. Int J Biol Macromol. 2018;120(Pt A): 1294-1305. doi: 10.1016/j.ijbiomac.2018.09.002
- Gregory DA, Taylor CS, Fricker AT, et al. Polyhydroxyalkanoates and their advances for biomedical applications. Trends Mol Med. 2022;28(4):331-342. doi: 10.1016/j.molmed.2022.01.007
- Pereira TF, Oliveira MF, Maia IA, et al. 3D printing of poly(3-Hydroxybutyrate) porous structures using selective laser sintering. Macromol Symp. 2012;319(1):64-73. doi: 10.1002/masy.201100237
- Melčová V, Svoradová K, Menčík P, et al. FDM 3D printed composites for bone tissue engineering based on plasticized Poly(3-Hydroxybutyrate)/Poly(d,l-Lactide) Blends. Polymers. 2020;12(12):2806. doi: 10.3390/polym12122806
- Krobot Š, Melčová V, Menčík P, et al. Poly(3- Hydroxybutyrate) (PHB) and polycaprolactone (PCL) based blends for tissue engineering and bone medical applications processed by FDM 3D printing. Polymers. 2023;15(10):2404. doi: 10.3390/polym15102404
- Kanabenja W, Passarapark K, Subchokpool T, et al. 3D printing filaments from plasticized polyhydroxybutyrate/ polylactic acid blends reinforced with hydroxyapatite. Addit Manuf. 2022;59:103130. doi: 10.1016/j.addma.2022.103130
- Moroni S, Khorshid S, Aluigi A, Tiboni M, Casettari L. Poly(3-Hydroxybutyrate): a potential biodegradable excipient for direct 3D printing of pharmaceuticals. Int J Pharm. 2022;623:121960. doi: 10.1016/j.ijpharm.2022.121960
- Kontárová S, Přikryl R, Melčová V, et al. Printability, mechanical and thermal properties of poly(3-Hydroxybutyrate)- Poly(Lactic Acid)-plasticizer blends for three-dimensional (3D) printing. Materials. 2020;13(21):4736. doi: 10.3390/ma13214736
- Armillotta A, Bellotti M, Cavallaro M. Warpage of FDM parts: experimental tests and analytic model. Robot Comput- Integr Manuf. 2018;50:140-152. doi: 10.1016/j.rcim.2017.09.007
- Antony Samy A, Golbang A, Harkin-Jones E, Archer E, Dahale M, McIlhagger A. Influence of ambient temperature on part distortion: a simulation study on amorphous and semi-crystalline polymer. Polymers. 2022;14(5):879. doi: 10.3390/polym14050879
- Samy AA, Golbang A, Harkin-Jones E, Archer E, Tormey D, McIlhagger A. Finite element analysis of residual stress and warpage in a 3D printed semi-crystalline polymer: effect of ambient temperature and nozzle speed. J Manuf Process. 2021;70:389-399. doi: 10.1016/j.jmapro.2021.08.054
- Wang X, Xu S, Zhou S, et al. Topological design and additive manufacturing of porous metals for bone scaffolds and orthopaedic implants: a review. Biomaterials. 2016;83:127-141. doi: 10.1016/j.biomaterials.2016.01.012
- Haba Y, Skripitz R, Lindner T, et al. Bone mineral densities and mechanical properties of retrieved femoral bone samples in relation to bone mineral densities measured in the respective patients. ScientificWorldJournal 2012;2012:242403. doi: 10.1100/2012/242403
- Rohlmann A, Zilch H, Bergmann G, Kolbel R. Material properties of femoral cancellous bone in axial loading. Arch. Orthop Trauma Surg. 1980;97(2); 95-102. doi: 10.1007/BF00450930
- Uchiyama T, Tanizawa T, Muramatsu H, Endo N, Takahashi HE, Hara T. Three-dimensional microstructural analysis of human trabecular bone in relation to its mechanical properties. Bone. 1999;25(4):487-491. doi: 10.1016/S8756-3282(99)00188-X
- Gonzalez Ausejo J, Rydz J, Musioł M, et al. Three-dimensional printing of PLA and PLA/PHA dumbbell-shaped specimens of crisscross and transverse patterns as promising materials in emerging application areas: prediction study. Polym Degrad Stab. 2018;156:100-110. doi: 10.1016/j.polymdegradstab.2018.08.008
- Ayatollahi MR, Nemati Giv A, Razavi N, Khoramishad, H. Mechanical properties of adhesively single lap-bonded joints reinforced with multi- walled carbon nanotubes and silica nanoparticles. J Adhes. 2017; 93(11): 896-913. doi: 10.1080/00218464.2016.1187069
- Zilch H, Rohlmann A, Bergmann G, Kölbel R. Material properties of femoral cancellous bone in axial loading. Part II: time dependent properties. Arch Orthop Trauma Surg (1978). 1980;97(4):257-262. doi: 10.1007/BF00380706
- Song Y, Li Y, Song W, Yee K, Lee K-Y, Tagarielli VL. Measurements of the mechanical response of unidirectional 3D-printed PLA. Mater Des. 2017;123:154-164. doi: 10.1016/j.matdes.2017.03.051
- Guo R, Lu S, Page JM, et al. Fabrication of 3D scaffolds with precisely controlled substrate modulus and pore size by templated-fused deposition modeling to direct osteogenic differentiation. Adv Healthc Mater. 2015;4(12): 1826-1832. doi: 10.1002/adhm.201500099

