A 3D-bioprinted neuroinflammatory co-culture model for in vitro study of Parkinson’s disease pathology

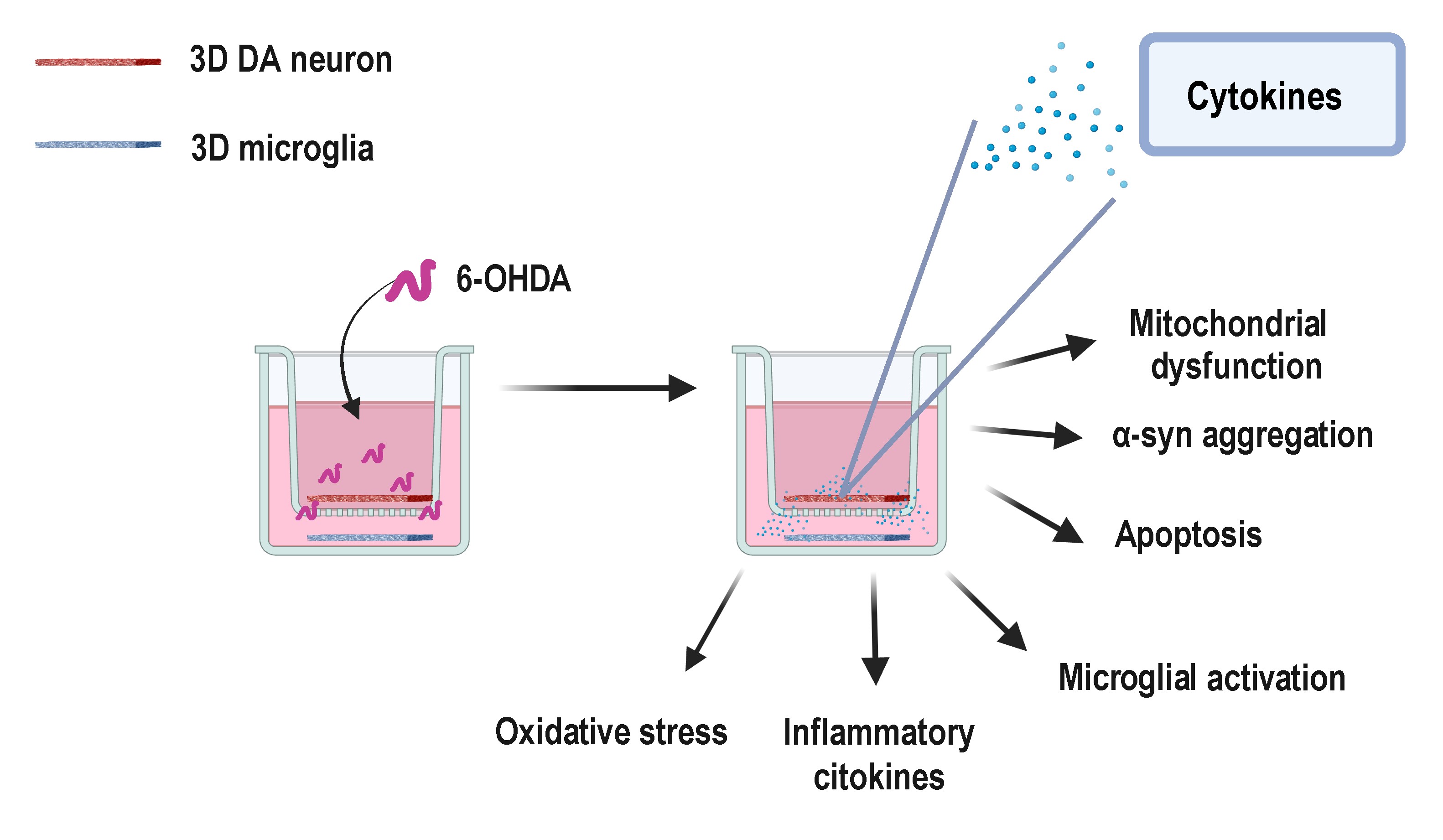
Parkinson’s disease (PD), a common neurodegenerative disorder, is characterized by dopaminergic (DA) neuron apoptosis, mitochondrial dysfunction, and aggregation of α-synuclein (α-syn). In this study, we developed a three-dimensional (3D)-bioprinted neuroinflammatory co-culture model to simulate key pathological features of PD in vitro. Traditional 2D cell culture models fail to accurately replicate the complex microenvironment of PD, leading to the development of the 3D-bioprinted model. Utilizing a polyethylene glycol-hyaluronic acid methacryloyl hydrogel matrix, this model supports the co-culture of DA neurons and microglia, enabling a more accurate representation of PD pathology. Experimental results demonstrated that, under 6-hydroxydopamine induction, the 3D model successfully mimicked neuroinflammatory responses associated with PD, including M1 polarization of microglia and increased secretion of pro-inflammatory factors. Compared to traditional 2D models, DA neurons in the 3D model exhibited greater resistance to oxidative stress and neurotoxic challenges, with significantly slower rates of apoptosis. Additionally, the 3D model displayed key PD-specific pathological features, such as altered mitochondrial membrane potential, elevated reactive oxygen species levels, and overexpression of α-syn. This 3D-bioprinted PD model provides a closer-to-physiological platform for investigating the pathogenesis of PD and holds potential for use in drug screening. However, further optimization is required to enhance the model’s complexity and long-term stability, including incorporating peripheral immune cells to better simulate the progression of chronic neuroinflammation.
- Dorsey ER, Bloem BR. The Parkinson pandemic-a call to action. JAMA Neurol. 2018;75(1):9-10. doi: 10.1001/jamaneurol.2017.3299
- Barbosa AF, Voos MC, Chen J, et al. Cognitive or cognitive-motor executive function tasks? Evaluating verbal fluency measures in people with Parkinson’s disease. Biomed Res Int. 2017;2017:7893975. doi: 10.1155/2017/7893975
- Surmeier DJ, Obeso JA, Halliday GM. Selective neuronal vulnerability in Parkinson disease. Nat Rev Neurosci. 2017;18(2):101-113. doi: 10.1038/nrn.2016.178
- Stefanis L. α-Synuclein in Parkinson’s disease. Cold Spring Harb Perspect Med. 2012;2(2):a009399. doi: 10.1101/cshperspect.a009399
- Luk KC. Oxidative stress and α-synuclein conspire in vulnerable neurons to promote Parkinson’s disease progression. J Clin Invest. 2019;129(9):3530-3531. doi: 10.1172/jci130351
- Wong YC, Krainc D. α-synuclein toxicity in neurodegeneration: mechanism and therapeutic strategies. Nat Med. 2017;23(2):1-13. doi: 10.1038/nm.4269
- Polymeropoulos MH, Lavedan C, Leroy E, et al. Mutation in the alpha-synuclein gene identified in families with Parkinson’s disease. Science. 1997;276(5321):2045-2047. doi: 10.1126/science.276.5321.2045
- Pajares M, Rojo AI, Manda G, Manda G, Boscá L, Cuadrado A. Inflammation in Parkinson’s disease: mechanisms and therapeutic implications. Cells. 2020;9(7):1687. doi: 10.3390/cells9071687
- Yang R, Li DD, Li XX, Yang XX, Gao HM, Zhang F. Dihydroquercetin alleviates dopamine neuron loss via regulating TREM2 activation. Int J Biol Macromol. 2024;269(Pt 2):132179. doi: 10.1016/j.ijbiomac.2024.132179
- Yu Z, Yang L, Yang Y, et al. Epothilone B benefits nigral dopaminergic neurons by attenuating microglia activation in the 6-hydroxydopamine lesion mouse model of Parkinson’s disease. Front Cell Neurosci. 2018;12:324. doi: 10.3389/fncel.2018.00324
- Jha MK, Lee WH, Suk K. Functional polarization of neuroglia: implications in neuroinflammation and neurological disorders. Biochem Pharmacol. 2016;103:1-16. doi: 10.1016/j.bcp.2015.11.003
- Stępkowski TM, Wasyk I, Grzelak A, Kruszewski M. 6-OHDA-induced changes in Parkinson’s disease-related gene expression are not affected by the overexpression of PGAM5 in in vitro differentiated embryonic mesencephalic cells. Cell Mol Neurobiol. 2015;35(8):1137-1147. doi: 10.1007/s10571-015-0207-5
- Yan JQ, Sun JC, Zhai MM, Cheng LN, Bai XL, Feng CL. Lovastatin induces neuroprotection by inhibiting inflammatory cytokines in 6-hydroxydopamine treated microglia cells. Int J Clin Exp Med. 2015;8(6): 9030-9037.
- Zhang P, Li Y, Han X, Xing Q, Zhao L. Dexmedetomidine regulates 6-hydroxydopamine-induced microglial polarization. Neurochem Res. 2017;42(5):1524-1532. doi: 10.1007/s11064-017-2209-9
- Kwon HS, Koh SH. Neuroinflammation in neurodegenerative disorders: the roles of microglia and astrocytes. Transl Neurodegener. 2020;9(1):42. doi: 10.1186/s40035-020-00221-2
- da Silva DJ, Borges AF, Souza PO, et al. Decreased toll-like receptor 2 and toll-like receptor 7/8-induced cytokines in Parkinson’s disease patients. Neuroimmunomodulation 2016;23(1):58-66. doi: 10.1159/000443238
- Freeman L, Guo H, David CN, Brickey WJ, Jha S, Ting JP. NLR members NLRC4 and NLRP3 mediate sterile inflammasome activation in microglia and astrocytes. J Exp Med. 2017;214(5):1351-1370. doi: 10.1084/jem.20150237
- Liu W, Zhong Z, Hu N, et al. Coaxial extrusion bioprinting of 3D microfibrous constructs with cell-favorable gelatin methacryloyl microenvironments. Biofabrication. 2018;10(2):024102. doi: 10.1088/1758-5090/aa9d44
- Mota F, Braga L, Rocha L, Cabral B. 3D and 4D bioprinted human model patenting and the future of drug development. Nat Biotechnol. 2020;38(6):689-694. doi: 10.1038/s41587-020-0540-1
- Kang HW, Lee SJ, Ko IK, Kengla C, Yoo JJ, Atala A. A 3D bioprinting system to produce human-scale tissue constructs with structural integrity. Nat Biotechnol. 2016; 34(3):312-319. doi: 10.1038/nbt.3413
- Zhang Y, Chen H, Long X, Xu T. Three-dimensional-engineered bioprinted in vitro human neural stem cell self-assembling culture model constructs of Alzheimer’s disease. Bioact Mater. 2022;11:192-205. doi: 10.1016/j.bioactmat.2021.09.023
- Rueda-Gensini L, Serna JA, Rubio D, et al. Three-dimensional neuroimmune co-culture system for modeling Parkinson’s disease microenvironmentsin vitro. Biofabrication. 2023;15(4). doi: 10.1088/1758-5090/ace21b
- Chlebanowska P, Tejchman A, Sułkowski M, Skrzypek K, Majka M. Use of 3D organoids as a model to study idiopathic form of Parkinson’s disease. Int J Mol Sci. 2020;21(3):694. doi: 10.3390/ijms21030694
- Kim J, Koo BK, Knoblich JA. Human organoids: model systems for human biology and medicine. Nat Rev Mol Cell Biol. 2020;21(10):571-584. doi: 10.1038/s41580-020-0259-3
- Rick CE, Ebert A, Virag T, Bohn MC, Surmeier DJ. Differentiated dopaminergic MN9D cells only partially recapitulate the electrophysiological properties of midbrain dopaminergic neurons. Dev Neurosci. 2006;28(6):528-537. doi: 10.1159/000095115
- Jensen G, Holloway JL, Stabenfeldt SE. Hyaluronic acid biomaterials for central nervous system regenerative medicine. Cells. 2020;9(9):2113. doi: 10.3390/cells9092113
- Bao B, Zeng Q, Li K, et al. Rapid fabrication of physically robust hydrogels. Nat Mater. 2023;22(10):1253-1260. doi: 10.1038/s41563-023-01648-4
- Daubner SC, Le T, Wang S. Tyrosine hydroxylase and regulation of dopamine synthesis. Arch Biochem Biophys. 2011;508(1):1-12. doi: 10.1016/j.abb.2010.12.017
- Simunovic F, Yi M, Wang Y, et al. Gene expression profiling of substantia nigra dopamine neurons: further insights into Parkinson’s disease pathology. Brain 2009; 132(Pt 7):1795-1809. doi: 10.1093/brain/awn323
- De Camilli P, Cameron R, Greengard P. Synapsin I (protein I), a nerve terminal-specific phosphoprotein. I. Its general distribution in synapses of the central and peripheral nervous system demonstrated by immunofluorescence in frozen and plastic sections. J Cell Biol. 1983;96(5):1337-1354. doi: 10.1083/jcb.96.5.1337
- Hopperton KE, Mohammad D, Trépanier MO, Giuliano V, Bazinet RP. Markers of microglia in post-mortem brain samples from patients with Alzheimer’s disease: a systematic review. Mol Psychiatry 2018;23(2):177-198. doi: 10.1038/mp.2017.246
- Akolawala Q, Rovituso M, Versteeg HH, Rondon AMR, Accardo A. Evaluation of proton-induced DNA damage in 3D-engineered glioblastoma microenvironments. ACS Appl Mater Interfaces. 2022;14(18):20778-20789. doi: 10.1021/acsami.2c03706
- Wu Y, Xiang Y, Fang J, et al. The influence of the stiffness of GelMA substrate on the outgrowth of PC12 cells. Biosci Rep. 2019;39(1):BSR20181748. doi: 10.1042/BSR20181748
- Xia C, Chen P, Mei S, et al. Photo-crosslinked HAMA hydrogel with cordycepin encapsulated chitosan microspheres for osteoarthritis treatment. Oncotarget. 2017;8(2):2835-2849. doi: 10.18632/oncotarget.13748
- Jin P, Pang A, Yang R, Guo X, He J, Zhai J. Study on mechanical properties of polyurethane cross-linked P(E-co-T)/PEG blended polyether elastomer. Polymers (Basel). 2022;14(24):5419. doi: 10.3390/polym14245419
- Monzel AS, Smits LM, Hemmer K, et al. Derivation of human midbrain-specific organoids from neuroepithelial stem cells. Stem Cell Rep. 2017;8(5):1144-1154. doi: 10.1016/j.stemcr.2017.03.010
- Abdelrahman S, Alsanie WF, Khan ZN, et al. A Parkinson’s disease model composed of 3D bioprinted dopaminergic neurons within a biomimetic peptide scaffold. Biofabrication. 2022;14(4). doi: 10.1088/1758-5090/ac7eec
- Fiore NJ, Ganat YM, Devkota K, et al. Bioengineered models of Parkinson’s disease using patient-derived dopaminergic neurons exhibit distinct biological profiles in a 3D microenvironment. Cell Mol Life Sci. 2022;79(2):78. doi: 10.1007/s00018-021-04047-7
- Gendelman HE. Neural immunity: friend or foe? J Neurovirol. 2002;8(6):474-479. doi: 10.1080/13550280290168631
- Streit WJ, Mrak RE, Griffin WS. Microglia and neuroinflammation: a pathological perspective. J Neuroinflammation. 2004;1(1):14. doi: 10.1186/1742-2094-1-14
- Garden GA. Epigenetics and the modulation of neuroinflammation. Neurotherapeutics. 2013;10(4):782-788. doi: 10.1007/s13311-013-0207-4
- Kwak TH, Kang JH, Hali S, et al. Generation of homogeneous midbrain organoids with in vivo-like cellular composition facilitates neurotoxin-based Parkinson’s disease modeling. Stem Cells. 2020;38(6),727-740. doi: 10.1002/stem.3163

