Integrative pathophysiological and psychological strategies for Alzheimer’s and dementia-related disease management

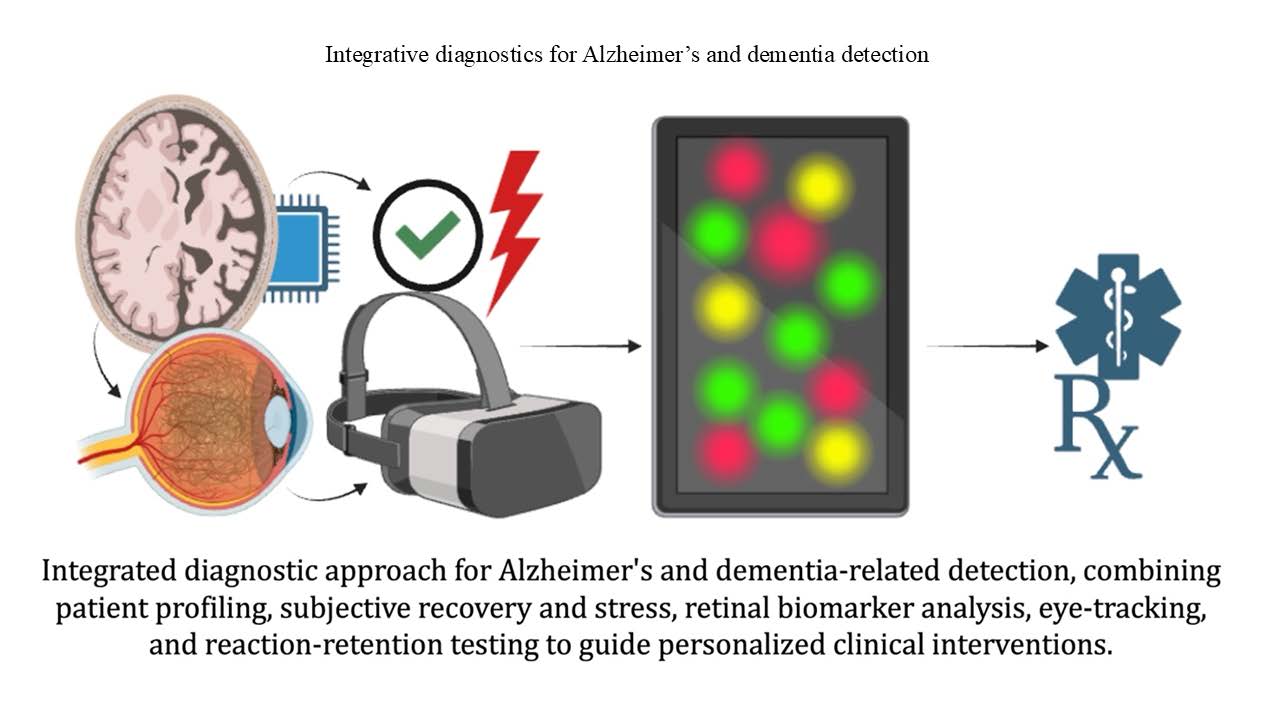
As global demographics shift toward an aging population, the urgency for innovative diagnostic tools to address Alzheimer’s and dementia-related diseases (ADR) has never been greater. Current methods often miss the opportunity to capture the nuanced interplay between cognitive decline and emotional health, leaving critical diagnostic gaps. This paper introduces an integrated approach combining advanced retinal imaging through eye-tracking, reaction-retention testing, and the short recovery-stress scale (SRSS) to provide a holistic evaluation and monitoring system of ADR progression. Retinal imaging through eye-tracking captures neurodegenerative biomarkers such as saccadic movements and fixation patterns and offers real-time insights into cognitive processing. Reaction-retention tests directly measure short-term memory and processing speed while the SRSS evaluates emotional states influencing cognitive health and physical capabilities. These modalities deliver a comprehensive cognitive-emotional profile, enabling earlier detection and personalized interventions. By synthesizing data across physiological and psychological domains, this approach addresses the multifaceted nature of ADR, providing clinicians with actionable insights into disease management. These integrative tools also hold promise for scalable applications in routine clinical and home settings, paving the way for enhanced monitoring, tailored therapies, and improved patient outcomes. The proposed framework represents a potential paradigm shift in ADR diagnostics, offering potential extensions to other neurodegenerative conditions, including Parkinson’s disease and traumatic brain injuries.
- Garcia-Cordero I, Migeot J, Fittipaldi S, et al. Metacognition of emotion recognition across neurodegenerative diseases. Cortex. 2021;137:93-107. doi: 10.1016/j.cortex.2020.12.023
- Sharafi SM, Sylvestre JP, Chevrefils C, et al. Vascular retinal biomarkers improves the detection of the likely cerebral amyloid status from hyperspectral retinal images. Alzheimers Dement (N Y). 2019;5:610-617. doi: 10.1016/j.trci.2019.09.006
- Imaoka Y, Flury A, De Bruin ED. Assessing saccadic eye movements with head-mounted display virtual reality technology. Front Psychiatry. 2020;11:572938. doi: 10.3389/fpsyt.2020.572938
- Babu Henry Samuel I, Wang C, Burke SE, Kluger B, Ding M. Compensatory neural responses to cognitive fatigue in young and older adults. Front Neural Circuits. 2019;13:12. doi: 10.3389/fncir.2019.00012
- Ratnayaka JA, Serpell LC, Lotery AJ. Dementia of the eye: The role of amyloid beta in retinal degeneration. Eye (Lond). 2015;29:1013-1026. doi: 10.1038/eye.2015.100
- Liu X, Liu Y, Liu Q. Fluorescent sensing platforms for detecting and imaging the biomarkers of Alzheimer’s disease. Biosensors (Basel). 2023;13:515. doi: 10.3390/bios13050515
- Kim HM, Han JW, Park YJ, Bae JB, Woo SJ, Kim KW. Association between retinal layer thickness and cognitive decline in older adults. JAMA Ophthalmol. 2022;140:683-690. doi: 10.1001/jamaophthalmol.2022.1563
- Torm MEW, Sander B, Hornum M, Krohn P, Birn H, Larsen M. Characterization of hyperreflective dots by structural and angiographic optical coherence tomography in patients with diabetic retinopathy and healthy subjects. J Clin Med. 2022;11:6646. doi: 10.3390/jcm11226646
- Oyama A, Takeda S, Ito Y, et al. Novel method for rapid assessment of cognitive impairment using high-performance eye-tracking technology. Sci Rep. 2019;9:12932. doi: 10.1038/s41598-019-49275-x
- Liao C, Xu J, Chen Y, Ip NY. Retinal dysfunction in Alzheimer’s disease and implications for biomarkers. Biomolecules. 2021;11:1215. doi: 10.3390/biom11081215
- Lee JY, Martin-Bastida A, Murueta-Goyena A, et al. Multimodal brain and retinal imaging of dopaminergic degeneration in Parkinson disease. Nat Rev Neurol. 2022;18:203-220. doi: 10.1038/s41582-022-00618-9
- Outeiro TF, Koss DJ, Erskine D, et al. Dementia with lewy bodies: An update and outlook. Mol Neurodegener. 2019;14:5. doi: 10.1186/s13024-019-0306-8
- Keady J, Jones L, Ward R, et al. Introducing the bio-psycho-social-physical model of dementia through a collective case study design. J Clin Nurs. 2013;22:2768-2777. doi: 10.1111/j.1365-2702.2012.04292.x
- Jenner LA, Farran EK, Welham A, Jones C, Moss J. The use of eye-tracking technology as a tool to evaluate social cognition in people with an intellectual disability: A systematic review and meta-analysis. J Neurodev Disord. 2023;15:42. doi: 10.1186/s11689-023-09506-9
- Mahanama B, Jayawardana Y, Rengarajan S, et al. Eye movement and pupil measures: A review. Front Comput Sci. 2022;3:733531. doi: 10.3389/fcomp.2021.733531
- Medina LD, Woo E, Rodriguez-Agudelo Y, et al. Reaction time and response inhibition in autosomal dominant Alzheimer’s disease. Brain Cogn. 2021;147:105656. doi: 10.1016/j.bandc.2020.105656
- Perkins AR, Travis SK, Mizuguchi S, et al. Convergent validity of the short recovery and stress scale in collegiate weightlifters. Int J Exerc Sci. 2022;15:1457-1471. doi: 10.70252/DAEO2007
- Travis SK, Mizuguchi S, Stone MH, Sands WA, Bazyler CD. Preparing for a national weightlifting championship: A case series. J Strength Cond Res. 2020;34:1842-1850. doi: 10.1519/JSC.0000000000003312
- Kölling S, Ferrauti A, Meyer T, Pfeiffer M, Kellmann M. Modification and applicability of questionnaires to assess the recovery-stress state among adolescent and child athletes. Front Physiol. 2019;10:1414. doi: 10.3389/fphys.2019.01414
- Lu S, Tan Z, Kong S, Zhang D. Cost-effective gaze tracking system based on polymer fiber specklegrams. Opt Lett. 2024;49:5027-5030. doi: 10.1364/OL.531946
- Sinha P, Barocas JA. Cost-effectiveness of aducanumab to prevent Alzheimer’s disease progression at current list price. Alzheimer’s Dement (NY). 2022;8:e12256. doi: 10.1002/trc2.12256
- Kellmann M, Kölling S. Recovery and Stress in Sport: A Manual for Testing and Assessment. 1st ed. London, New York: Routledge; 2019.

