BioPrint-LKM: An evidence-grounded large knowledge model for bioprinting knowledge retrieval and parameter initialization

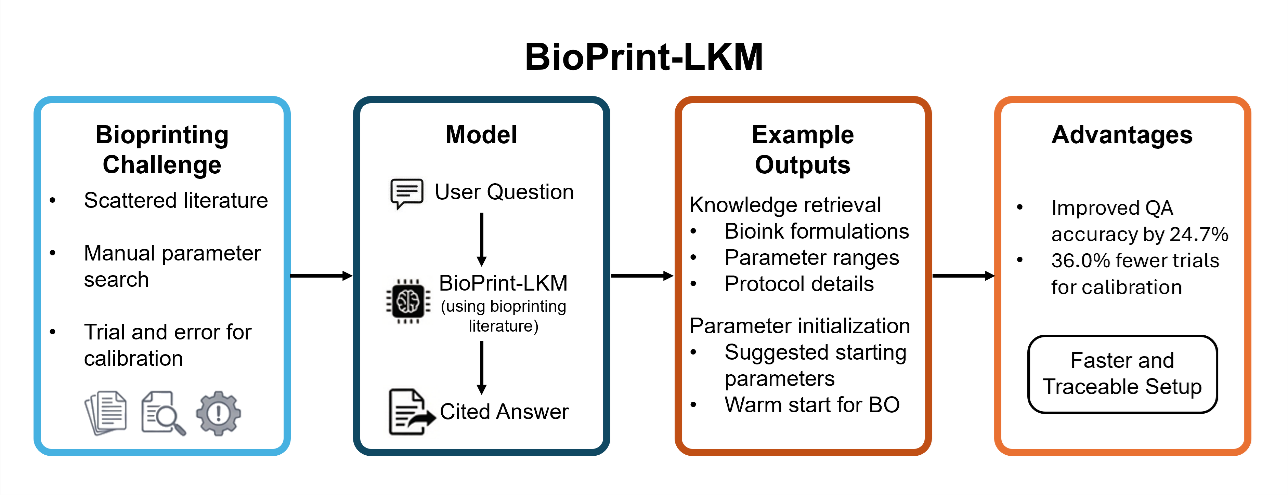
Bioprinting workflows often require repeated trial-and-error to achieve acceptable print quality, while relevant process knowledge and parameter ranges are dispersed across a rapidly growing literature. General-purpose language models can assist with scientific questions, but their output may be difficult to verify and can include unsupported claims. In this work, we present BioPrint-LKM, a bioprinting large knowledge model (LKM) implemented using a retrieval-augmented generation pipeline with citation grounding to provide traceable, evidence-based responses for bioprinting tasks. A curated knowledge base was constructed from 621 bioprinting papers, which were converted to text, segmented into passages, embedded into a vector index, and retrieved using exact nearest-neighbor similarity search. Retrieved passages were assembled into an augmented context and used to constrain generation under a domain prompt guideline that enforces source and page-level citation. The LKM was evaluated using 30 bioprinting-related questions and three large language model (LLM) backbones (GPT-4o, Claude-Sonnet-4.6, and Gemini-2.5-Flash). Across the tested 30-question benchmark, retrieval and citation grounding improved question-answer accuracy compared with LLM-only baselines by an average of 24.7%, particularly for queries requiring paper specific details such as component concentrations, mixing ratios, and instrument settings. A retrieval hyperparameter sweep further showed that top-k and passage length affect both evidence coverage and noise, with an intermediate passage length providing the best overall performance. Beyond knowledge retrieval, the LKM was applied to bioprinting process setup by generating initial parameter sets that were used to warm-start multi-objective Bayesian optimization toward target filament width and height. Compared with manual initialization, BioPrint-LKM-assisted initialization reduced the number of calibration trials by an average of 36.0% and supported downstream printing demonstrations, including conductive cell-laden hydrogel lines and an anatomical ear model. These results suggest that BioPrint-LKM, a citation grounded LKM, can serve as a practical lab-assistive tool to accelerate bioprinting setup and improve reproducibility.
- Mandrycky C, Wang Z, Kim K, Kim DH. 3D bioprinting for engineering complex tissues. Biotechnol Adv. 2016;34(4):422- 434. doi: 10.1016/j.biotechadv.2015.12.011
- Murphy SV, Atala A. 3D bioprinting of tissues and organs. Nat Biotechnol. 2014;32(8):773-785. doi: 10.1038/nbt.2958
- Groll J, Boland T, Blunk T, et al. Biofabrication: reappraising the definition of an evolving field. Biofabrication. 2016;8(1):013001. doi: 10.1088/1758-5090/8/1/013001
- Gungor-Ozkerim PS, Inci I, Zhang YS, Khademhosseini A, Dokmeci MR. Bioinks for 3D bioprinting: an overview. Biomater Sci. 2018;6(5):915-946. doi: 10.1039/c7bm00765e
- Sun W, Starly B, Daly AC, et al. The bioprinting roadmap. Biofabrication. 2020;12(2):022002. doi: 10.1088/1758-5090/ab5158
- Ng WL, Chua CK, Shen YF. Print Me An Organ! Why We Are Not There Yet. Prog Polym Sci. 2019;97:101145. doi: 10.1016/j.progpolymsci.2019.101145
- Lee JM, Ng WL, Yeong WY. Resolution and shape in bioprinting: Strategizing towards complex tissue and organ printing. Appl Phys Rev. 2019;6(1):011307. doi: 10.1063/1.5053909
- Ozbolat IT, Hospodiuk M. Current advances and future perspectives in extrusion-based bioprinting. Biomaterials. 2016;76:321-343. doi: 10.1016/j.biomaterials.2015.10.076
- Meng Z, He J, Li J, Su Y, Li D. Melt-based, solvent-free additive manufacturing of biodegradable polymeric scaffolds with designer microstructures for tailored mechanical/biological properties and clinical applications. Virtual Phys Prototyp. 2020;15(4):417-444. doi: 10.1080/17452759.2020.1808937
- Choe YE, Kim GH. A PCL/cellulose coil-shaped scaffold via a modified electrohydrodynamic jetting process. Virtual Phys Prototyp. 2020;15(4):403-416. doi: 10.1080/17452759.2020.1808269
- Li X, Liu B, Pei B, et al. Inkjet Bioprinting of Biomaterials. Chem Rev. 2020;120(19):10793-10833. doi: 10.1021/acs.chemrev.0c00008
- Weygant J, Entezari A, Koch F, et al. Droplet 3D cryobioprinting for fabrication of free-standing and volumetric structures. Aggregate. 2024;5(5):e599. doi: 10.1002/agt2.599
- Ng WL, Lee JM, Zhou M, et al. Vat polymerization-based bioprinting—process, materials, applications and regulatory challenges. Biofabrication. 2020;12(2):022001. doi: 10.1088/1758-5090/ab6034
- Lin H, Zhang D, Alexander PG, et al. Application of visible light-based projection stereolithography for live cell-scaffold fabrication with designed architecture. Biomaterials. 2013;34(2):331-339. doi: 10.1016/j.biomaterials.2012.09.048
- Lemma ED, Spagnolo B, De Vittorio M, Pisanello F. Studying Cell Mechanobiology in 3D: The Two-Photon Lithography Approach. Trends Biotechnol. 2019;37(4):358-372. doi: 10.1016/j.tibtech.2018.09.008
- Miri AK, Khalilpour A, Cecen B, Maharjan S, Shin SR, Khademhosseini A. Multiscale bioprinting of vascularized models. Biomaterials. 2019;198:204-216. doi: 10.1016/j.biomaterials.2018.08.006
- Lee A, Hudson AR, Shiwarski DJ, et al. 3D bioprinting of collagen to rebuild components of the human heart. Science. 2019;365(6452):482-487. doi: 10.1126/science.aav9051
- Parihar A, Parihar DS, Gaur K, Arya N, Choubey VK, Khan R. 3D bioprinting for drug development and screening: Recent trends towards personalized medicine. Hybrid Adv. 2024;7:100320. doi: 10.1016/j.hybadv.2024.100320
- Parihar A, Pandita V, Khan R. 3D printed human organoids: High throughput system for drug screening and testing in current COVID-19 pandemic. Biotechnol Bioeng. 2022;119(10):2669-2688. doi: 10.1002/bit.28166
- Grijalva Garces D, Strauß S, Gretzinger S, et al. On the reproducibility of extrusion-based bioprinting: round robin study on standardization in the field. Biofabrication. 2023;16(1):015002. doi: 10.1088/1758-5090/acfe3b
- Schwab A, Levato R, D’Este M, Piluso S, Eglin D, Malda J. Printability and Shape Fidelity of Bioinks in 3D Bioprinting. Chem Rev. 2020;120(19):11028-11055. doi: 10.1021/acs.chemrev.0c00084
- He Y, Yang F, Zhao H, Gao Q, Xia B, Fu J. Research on the printability of hydrogels in 3D bioprinting. Sci Rep. 2016;6(1):29977. doi: 10.1038/srep29977
- Compaired PM, García-Gareta E, Pérez MÁ. An experimental workflow for bioprinting optimization: Application to a custom-made biomaterial ink. Int J Bioprinting. 2025;11(3):397-415. doi: 10.36922/IJB025120094
- Tian S, Zhao H, Lewinski N. Key parameters and applications of extrusion-based bioprinting. Bioprinting. 2021;23:e00156. doi: 10.1016/j.bprint.2021.e00156
- Yu C, Jiang J. A Perspective on Using Machine Learning in 3D Bioprinting. Int J Bioprinting. 2020;6(1):253. doi: 10.18063/ijb.v6i1.253
- Shangguan P, Zhou H, Huang X, Su J, Yeong WY, Sing SL. Artificial intelligence-driven material development for additive manufacturing: A critical review. Int J AI Mater Des. 2025;2(2):1-26. doi: 10.36922/IJAMD025100007
- Zolfagharian A, Jin L, Ge Q, et al. Roadmap on Artificial Intelligence-Augmented Additive Manufacturing. Adv Intell Syst. 2026:e202500484. doi: 10.1002/aisy.202500484
- Sani AR, Zolfagharian A, Kouzani AZ. Artificial Intelligence- Augmented Additive Manufacturing: Insights on Closed-Loop 3D Printing. Adv Intell Syst. 2024;6(10):2400102. doi: 10.1002/aisy.202400102
- Huang X, Ng WL, Yeong WY. Predicting the number of printed cells during inkjet-based bioprinting process based on droplet velocity profile using machine learning approaches. J Intell Manuf. 2024;35(5):2349-2364. doi: 10.1007/s10845-023-02167-4
- Huang X, Wong YX, Goh GL, Gao X, Lee JM, Yeong WY. Machine learning-driven prediction of gel fraction in conductive gelatin methacryloyl hydrogels. Int J AI Mater Des. 2024;1(2):61-75. doi: 10.36922/ijamd.3807
- Shi X, Sun Y, Tian H, Abhilash PM, Luo X, Liu H. Material Extrusion Filament Width and Height Prediction via Design of Experiment and Machine Learning. Micromachines. 2023;14(11):2091. doi: 10.3390/mi14112091
- Ruberu K, Senadeera M, Rana S, et al. Coupling machine learning with 3D bioprinting to fast track optimisation of extrusion printing. Appl Mater Today. 2021;22:100914. doi: 10.1016/j.apmt.2020.100914
- Freeman S, Calabro S, Williams R, Jin S, Ye K. Bioink Formulation and Machine Learning-Empowered Bioprinting Optimization. Front Bioeng Biotechnol. 2022;10:913579. doi: 10.3389/fbioe.2022.913579
- Lewis P, Perez E, Piktus A, et al. Retrieval-Augmented Generation for Knowledge-Intensive NLP Tasks. In: Larochelle H, Ranzato M, Hadsell R, Balcan MF, Lin H, editors. Advances in Neural Information Processing Systems
- In: Proceedings of the 34th Conference on Neural Information Processing Systems (NeurIPS 2020); December 6–12, 2020; Online. Neural Information Processing Systems Foundation, Inc. (NeurIPS); 2020:9459-9474.
- Huang L, Yu W, Ma W, et al. A Survey on Hallucination in Large Language Models: Principles, Taxonomy, Challenges, and Open Questions. ACM Trans Inf Syst. 2025;43(2):42. doi: 10.1145/3703155
- Tshitoyan V, Dagdelen J, Weston L, et al. Unsupervised word embeddings capture latent knowledge from materials science literature. Nature. 2019;571(7763):95-98. doi: 10.1038/s41586-019-1335-8
- Lee J, Su H. A unified industrial large knowledge model framework in Industry 4.0 and smart manufacturing. Int J AI Mater Des. 2024;1(2):41-47. doi: 10.36922/ijamd.3681
- Lee J, Su H. Rethinking industrial artificial intelligence: A unified foundation framework. Int J AI Mater Des. 2025;2(2):56-68. doi: 10.36922/IJAMD025080006
- Lee J, Su H, Ji DY, Minami T. Engineering Artificial Intelligence: Framework, Challenges, and Future Direction. Mach Learn Eng. 2025;1(1):013001. doi: 10.1088/3049-4761/adce0d
- He K, Zhang X, Ren S, Sun J. Deep Residual Learning for Image Recognition. arXiv. Posted online December 10,
- doi: 10.48550/arXiv.1512.03385
- Sani AR, Zolfagharian A, Kouzani AZ. Automated defects detection in extrusion 3D printing using YOLO models. J Intell Manuf. 2026;37(1):351-371. doi: 10.1007/s10845-024-02543-8
- Sani AR, Kouzani AZ, Zolfagharian A. Real-time defect monitoring in material extrusion 3D printing using optimized YOLO models. Prog Addit Manuf. 2026;11(1):1115-1137. doi: 10.1007/s40964-025-01401-0
- Daulton S, Balandat M, Bakshy E. Differentiable Expected Hypervolume Improvement for Parallel Multi-Objective Bayesian Optimization. arXiv. Posted online June 9, 2020. doi: 10.48550/arXiv.2006.05078
- Lee J, Su H. Agentic AI for smart manufacturing. Manuf Lett. 2025;46:92-96. doi: 10.1016/j.mfglet.2025.10.013

