3D bioprinting of the glioblastoma microenvironment for preclinical assessment of CDK4/6 inhibition

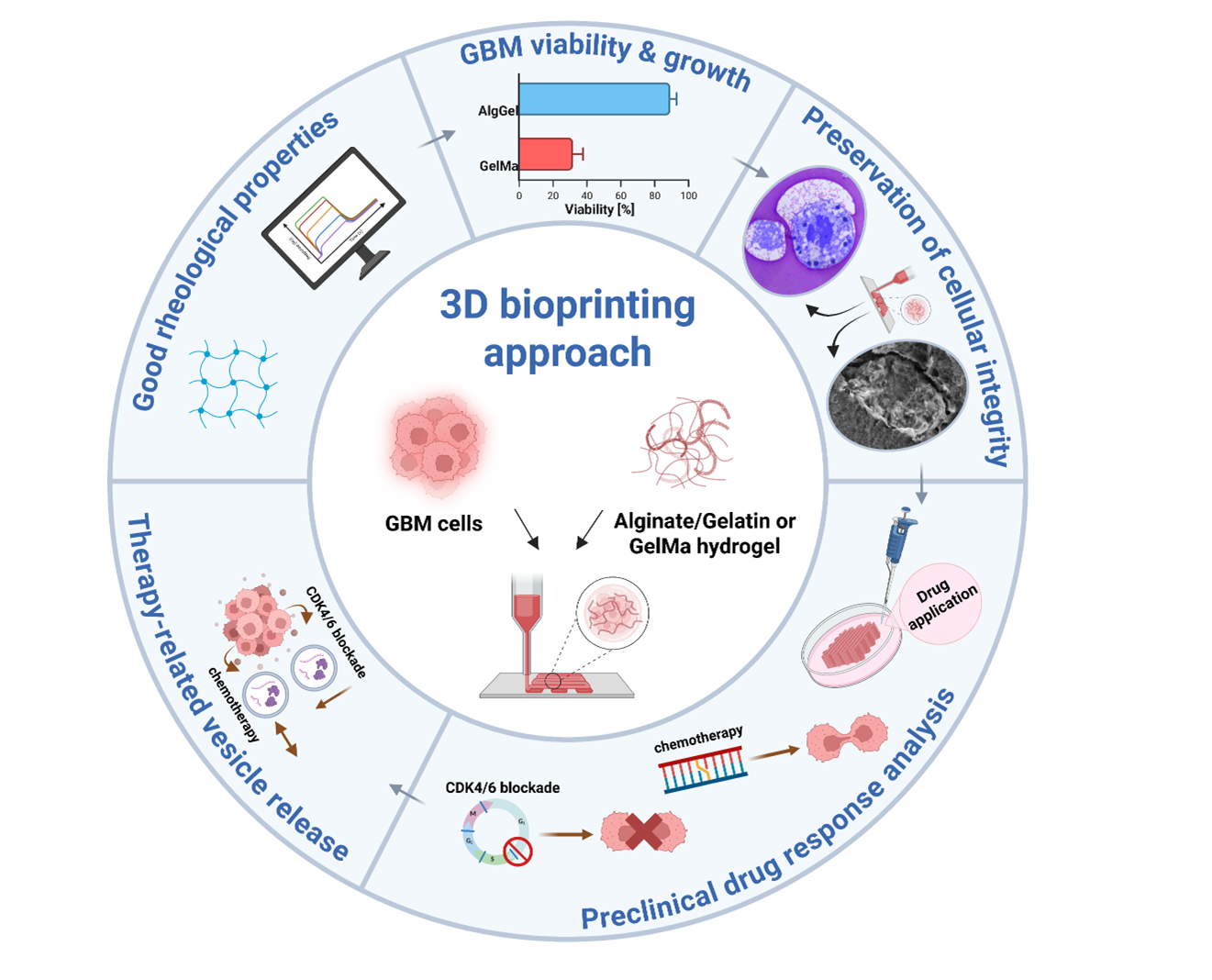
Glioblastoma (GBM) is an aggressive, World Health Organization grade 4 brain tumor with a poor prognosis, largely due to its complex, treatment-resistant microenvironment. To better model this environment for preclinical testing, we developed a three-dimensional (3D) biomimetic bioprinting platform using patient-derived GBM cells. Two hydrogels, alginate/gelatin (AlgGel; 1.5%/7.5%) and gelatin methacryloyl (10%), were evaluated for biocompatibility. GBM cells (GBM06, GBM14, and GBM15), transduced with iRFP-680 for viability tracking, were embedded in the hydrogels and printed. Tumor growth and viability were monitored for 28 days using fluorescence microscopy, complemented by electron microscopy (EM) for structural analysis. Drug response testing included temozolomide (TMZ; 10 μM) and the cyclin-dependent kinases 4/6 inhibitor abemaciclib (1 μM). Cell viability and extracellular vesicle (EV) release were quantified. Efficacy was further assessed in a co-culture with astrocytes. The AlgGel hydrogel supported superior long-term viability and growth. EM analysis of AlgGel scaffolds revealed preserved cellular architecture and adherence to the bioprinted extracellular matrix. Drug response assays confirmed findings previously observed in 2D and 3D cultures. Two cycles of abemaciclib reduced GBM cell viability in AlgGel scaffolds, accompanied by a significant decrease in EV secretion. TMZ, in contrast, did not significantly affect cell viability. The reduction in viability remained pronounced in co-culture with astrocytes, without compromising astrocyte viability. In this study, we present a 3D biomimetic bioprinting model that successfully mimics key aspects of the GBM microenvironment. This model demonstrates strong potential as a preclinical drug screening tool, enabling improved mechanistic insight into cell–matrix interactions that govern nutrient/metabolite diffusion and therapeutic responses.
- Ostrom QT, Cioffi G, Waite K, Kruchko C, Barnholtz-Sloan JS. CBTRUS statistical report: primary brain and other central nervous system tumors diagnosed in the United States in 2014–2018. Neuro Oncol. 2021;23(Suppl 3):iii1. doi: 10.1093/NEUONC/NOAB200
- Weller M, Wen PY, Chang SM, et al. Glioma. Nat Rev Dis Primers. 2024;10(1):33. doi: 10.1038/S41572-024-00516-Y
- Hughes JP, Rees SS, Kalindjian SB, Philpott KL. Principles of early drug discovery. Br J Pharmacol. 2011;162(6):1239-1249. doi: 10.1111/J.1476-5381.2010.01127.X
- Knight E, Przyborski S. Advances in 3D cell culture technologies enabling tissue-like structures to be created in vitro. J Anat. 2015;227(6):746-756. doi: 10.1111/JOA.12257
- Costa EC, Moreira AF, de Melo-Diogo D, Gaspar VM, Carvalho MP, Correia IJ. 3D tumor spheroids: an overview on the tools and techniques used for their analysis. Biotechnol Adv. 2016;34(8):1427-1441. doi: 10.1016/J.BIOTECHADV.2016.11.002
- Ravi M, Paramesh V, Kaviya SR, Anuradha E, Paul Solomon FD. 3D cell culture systems: advantages and applications. J Cell Physiol. 2015;230(1):16-26. doi: 10.1002/JCP.24683
- Jensen C, Teng Y. Is it time to start transitioning from 2D to 3D cell culture? Front Mol Biosci. 2020;7:3. doi: 10.3389/FMOLB.2020.00033
- Ozbolat IT, Peng W, Ozbolat V. Application areas of 3D bioprinting. Drug Discov Today. 2016;21(8):1257-1271. doi: 10.1016/J.DRUDIS.2016.04.006
- Gungor-Ozkerim PS, Inci I, Zhang YS, Khademhosseini A, Dokmeci MR. Bioinks for 3D bioprinting: an overview. Biomater Sci. 2018;6(5):915-946. doi: 10.1039/C7BM00765E
- Han S, Kim S, Chen Z, et al. 3D bioprinted vascularized tumour for drug testing. Int J Mol Sci. 2020;21(8):2993. doi: 10.3390/IJMS21082993
- Bo T, Pascucci E, Capuani S, et al. 3D bioprinted mesenchymal stem cell laden scaffold enhances subcutaneous vascularization for delivery of cell therapy. Biomed Microdevices. 2024;26(3):29. doi: 10.1007/S10544-024-00713-2
- Forkel H, Grabarczyk P, Depke M, et al. BCL11B depletion induces the development of highly cytotoxic innate T cells out of IL-15 stimulated peripheral blood αβ CD8+ T cells. Oncoimmunology. 2022;11(1):2148850. doi: 10.1080/2162402X.2022.2148850
- Troschke-Meurer S, Zumpe M, Meißner L, et al. Chemotherapeutics used for high-risk neuroblastoma therapy improve the efficacy of anti-GD2 antibody dinutuximab beta in preclinical spheroid models. Cancers (Basel). 2023;15(3):904. doi: 10.3390/CANCERS15030904
- Riess C, del Moral K, Fiebig A, et al. Implementation of a combined CDK inhibition and arginine-deprivation approach to target arginine-auxotrophic glioblastoma multiforme cells. Cell Death Dis. 2022;13(6):555. doi: 10.1038/s41419-022-05006-1
- Kayser A, Wolff A, Berlin P, et al. Selective but not pan-CDK inhibition abrogates 5-FU-driven tissue factor upregulation in colon cancer. Sci Rep. 2024;14(1):1-12. doi: 10.1038/s41598-024-61076-5
- Amorim PA, d’Ávila MA, Anand R, Moldenaers P, Van Puyvelde P, Bloemen V. Insights on shear rheology of inks for extrusion-based 3D bioprinting. Bioprinting. 2021;22:e00129. doi: 10.1016/J.BPRINT.2021.E00129
- Naghieh S, Chen X. Printability–a key issue in extrusion-based bioprinting. J Pharm Anal. 2021;11(5):564-579. doi: 10.1016/J.JPHA.2021.02.001
- Habib MA, Khoda B. Rheological analysis of bio-ink for 3D bio-printing processes. J Manuf Process. 2022;76:708-718. doi: 10.1016/J.JMAPRO.2022.02.048
- Hull SM, Brunel LG, Heilshorn SC. 3D bioprinting of cell-laden hydrogels for improved biological functionality. Adv Mater. 2022;34(2):2103691. doi: 10.1002/ADMA.202103691
- Gao T, Gillispie GJ, Copus JS, et al. Optimization of gelatin-alginate composite bioink printability using rheological parameters: a systematic approach. Biofabrication. 2018;10(3):034106. doi: 10.1088/1758-5090/AACDC7
- Riess C, Koczan D, Schneider B, et al. Cyclin-dependent kinase inhibitors exert distinct effects on patient-derived 2D and 3D glioblastoma cell culture models. Cell Death Discov. 2021;7(1):54. doi: 10.1038/s41420-021-00423-1
- Freitag T, Kaps P, Ramtke J, et al. Combined inhibition of EZH2 and CDK4/6 perturbs endoplasmic reticulum-mitochondrial homeostasis and increases antitumor activity against glioblastoma. NPJ Precis Oncol. 2024;8(1):156. doi: 10.1038/S41698-024-00653-3
- Da Ros M, De Gregorio V, Iorio AL, et al. Glioblastoma chemoresistance: the double play by microenvironment and blood-brain barrier. Int J Mol Sci. 2018;19(10):2879. doi: 10.3390/IJMS19102879
- Zhang X, Ding K, Wang J, Li X, Zhao P. Chemoresistance caused by the microenvironment of glioblastoma and the corresponding solutions. Biomed Pharmacother. 2019;109:39-46. doi: 10.1016/J.BIOPHA.2018.10.063
- Perrin SL, Samuel MS, Koszyca B, et al. Glioblastoma heterogeneity and the tumour microenvironment: implications for preclinical research and development of new treatments. Biochem Soc Trans. 2019;47(2):625-638. doi: 10.1042/BST20180444
- Giuseppe M Di, Law N, Webb B, et al. Mechanical behaviour of alginate-gelatin hydrogels for 3D bioprinting. J Mech Behav Biomed Mater. 2018;79:150-157. doi: 10.1016/J.JMBBM.2017.12.018
- Liu P, Shen H, Zhi Y, et al. 3D bioprinting and in vitro study of bilayered membranous construct with human cells-laden alginate/gelatin composite hydrogels. Colloids Surf B Biointerfaces. 2019;181:1026-1034. doi: 10.1016/J.COLSURFB.2019.06.069
- Yin J, Yan M, Wang Y, Fu J, Suo H. 3D bioprinting of low-concentration cell-laden gelatin methacrylate (GelMA) bioinks with a two-step cross-linking strategy. ACS Appl Mater Interfaces. 2018;10(8):6849-6857. doi: 10.1021/ACSAMI.7B16059
- Cuvellier M, Ezan F, Oliveira H, et al. 3D culture of HepaRG cells in GelMa and its application to bioprinting of a multicellular hepatic model. Biomaterials. 2021;269:120611. doi: 10.1016/J.BIOMATERIALS.2020.120611
- Nair K, Gandhi M, Khalil S, et al. Characterization of cell viability during bioprinting processes. Biotechnol J. 2009;4(8):1168-1177. doi: 10.1002/BIOT.200900004
- Boularaoui S, Al Hussein G, Khan KA, Christoforou N, Stefanini C. An overview of extrusion-based bioprinting with a focus on induced shear stress and its effect on cell viability. Bioprinting. 2020;20:e00093. doi: 10.1016/J.BPRINT.2020.E00093
- Adhikari J, Roy A, Das A, et al. Effects of processing parameters of 3D bioprinting on the cellular activity of bioinks. Macromol Biosci. 2021;21(1):2000179. doi: 10.1002/MABI.202000179
- Davidenko N, Schuster CF, Bax DV, et al. Evaluation of cell binding to collagen and gelatin: a study of theeffect of 2D and 3D architecture and surface chemistry. J Mater Sci Mater Med. 2016;27(10):148. doi: 10.1007/S10856-016-5763-9
- Su K, Wang C. Recent advances in the use of gelatin in biomedical research. Biotechnol Lett. 2015;37(11):2139-2145. doi: 10.1007/S10529-015-1907-0
- Streulli CH, Akhtar N. Signal co-operation between integrins and other receptor systems. Biochem J. 2009;418(3): 491-506. doi: 10.1042/BJ20081948
- Jeanes AI, Wang P, Moreno-Layseca P, et al. Specific β-containing integrins exert differential control on proliferation and two-dimensional collective cell migration in mammary epithelial cells. J Biol Chem. 2012;287(29):24103-24112. doi: 10.1074/JBC.M112.360834
- Moreno-Layseca P, Streuli CH. Signalling pathways linking integrins with cell cycle progression. Matrix Biol. 2014;34:144-153. doi: 10.1016/J.MATBIO.2013.10.011
- Tang M, Xie Q, Gimple RC, et al. Three-dimensional bioprinted glioblastoma microenvironments model cellular dependencies and immune interactions. Cell Res. 2020;30(10):833-853. doi: 10.1038/s41422-020-0338-1
- Li DM, Chen QD, Wei GN, et al. Hypoxia-induced miR-137 inhibition increased glioblastoma multiforme growth and chemoresistance through LRP6. Front Oncol. 2021;10:611699. doi: 10.3389/FONC.2020.611699
- Blaeser A, Filipa Duarte Campos D, Puster U, et al. Controlling shear stress in 3D bioprinting is a key factor to balance printing resolution and stem cell integrity. Adv Healthc Mater. 2016;5(3):326-333. doi: 10.1002/ADHM.201500677
- Hanahan D, Weinberg RAA. Hallmarks of cancer: the next generation. Cell. 2011;144(5):646-674. doi: 10.1016/j.cell.2011.02.013
- Koo H, Choi SW, Cho HJ, et al. Ethnic delineation of primary glioblastoma genome. Cancer Med. 2020;9(19):7352. doi: 10.1002/CAM4.3370
- Zhao R, Choi BY, Lee MH, Bode AM, Dong Z. Implications of genetic and epigenetic alterations of CDKN2A (p16INK4a) in cancer. EBioMedicine. 2016;8:30-39. doi: 10.1016/J.EBIOM.2016.04.017
- Fontoura JC, Viezzer C, dos Santos FG, et al. Comparison of 2D and 3D cell culture models for cell growth, gene expression and drug resistance. Mater Sci Eng C. 2020;107:110264. doi: 10.1016/J.MSEC.2019.110264
- Mulcahy LA, Pink RC, Carter DRF. Routes and mechanisms of extracellular vesicle uptake. J Extracell Vesicles. 2014;3(1):24641. doi: 10.3402/JEV.V3.24641
- Raposo G, Stoorvogel W. Extracellular vesicles: exosomes, microvesicles, and friends. J Cell Biol. 2013;200(4):373-383. doi: 10.1083/JCB.201211138
- Nejo T, Krishna S, Yamamichi A, et al. Glioma-neuronal circuit remodeling induces regional immunosuppression. Nat Commun. 2025;16(1):4770. doi: 10.1038/s41467-025-60074-z
- Brandao M, Simon T, Critchley G, Giamas G. Astrocytes, the rising stars of the glioblastoma microenvironment. Glia. 2019;67(5):779-790. doi: 10.1002/glia.23520
- Mega A, Hartmark Nilsen M, Leiss LW, et al. Astrocytes enhance glioblastoma growth. Glia. 2020;68(2):316-327. doi: 10.1002/glia.23718

