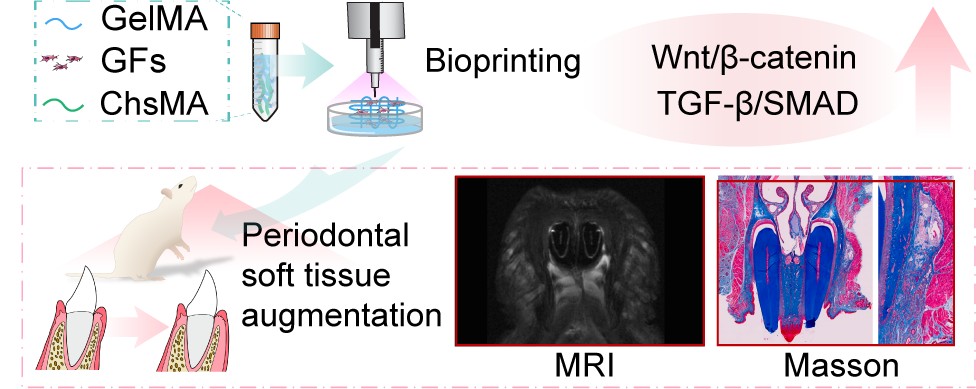
3D-bioprinted active gingival hydrogel for periodontal soft tissue regeneration via TGF-β/ Smad and Wnt/β-Catenin-mediated extracellular matrix remodeling

Soft tissue management is essential in periodontal, orthodontic, and implant therapies, yet autologous grafts remain limited by donor-site morbidity, inconsistent tissue quality, and restricted availability. To address these challenges, we developed an active gingival hydrogel (AGH) composed of gelatin methacrylate, chondroitin sulfate methacrylate, and gingival fibroblasts, which was fabricated into cell-laden hydrogels using extrusion-based 3D bioprinting. The AGH exhibited excellent rheological performance, print fidelity, and interconnected porous microstructures that supported nutrient diffusion and cell migration. Gingival fibroblasts cocultured with the AGH showed robust adhesion, proliferation, and collagen matrix deposition, accompanied by significant upregulation of fibronectin and type I collagen. Mechanistic studies revealed that these effects were mediated through activation of the Wnt/β-catenin and transforming growth factor-β/Smad signaling pathways, which synergistically regulate extracellular matrix remodeling and epithelial keratinization. In vivo experiments demonstrated that AGH implantation significantly enhanced gingival thickness, collagen density, and neovascularization while reducing inflammatory infiltration, as verified by magnetic resonance imaging as well as histological and immunohistochemical analyses. Furthermore, coculture with gingival epithelial cells promoted upregulation of Krt10 and Krt14, indicating improved epithelial differentiation. Collectively, this study establishes a 3D-bioprinted active gingival hydrogel as a biomimetic and functional substitute for autologous grafts, offering a promising strategy for periodontal and peri-implant soft tissue regeneration.
- Malpartida-Carrillo V, Tinedo-Lopez PL, Guerrero ME, Amaya-Pajares SP, Özcan M, Rösing CK. Periodontal phenotype: a review of historical and current classifications evaluating different methods and characteristics. J Esthet Restor Dent. 2021;33(3):432-445. doi: 10.1111/jerd.12661
- Ramanauskaite A, Sader R. Esthetic complications in implant dentistry. Periodontol 2000. 2022;88(1): 73-85. doi: 10.1111/prd.12412
- Stefanini M, Barootchi S, Sangiorgi M, et al. Do soft tissue augmentation techniques provide stable and favorable peri-implant conditions in the medium and long term? A systematic review. Clin Oral Implants Res. 2023;34 Suppl 26:28-42. doi: 10.1111/clr.14150
- Thoma DS, Benić GI, Zwahlen M, Hämmerle CH, Jung RE. A systematic review assessing soft tissue augmentation techniques. Clin Oral Implants Res. 2009;20 Suppl 4: 146-165. doi: 10.1111/j.1600-0501.2009.01784.x
- Edel A. Clinical evaluation of free connective tissue grafts used to increase the width of keratinised gingiva. J Clin Periodontol. 1974;1(4):185-196. doi: 10.1111/j.1600-051x.1974.tb01257.x
- Hürzeler MB, Weng D. A single-incision technique to harvest subepithelial connective tissue grafts from the palate. Int J Periodontics Restorative Dent. 1999;19(3): 279-287.
- Bertl K, Melchard M, Pandis N, Müller-Kern M, Stavropoulos A. Soft tissue substitutes in non-root coverage procedures: a systematic review and meta-analysis. Clin Oral Investig. 2017;21(2):505-518. doi: 10.1007/s00784-016-2044-4
- Song YW, Kim S, Waller T, et al. Soft tissue substitutes to increase gingival thickness: histologic and volumetric analyses in dogs. J Clin Periodontol. 2019;46(1):96-104. doi: 10.1111/jcpe.13034
- Valles C, Vilarrasa J, Barallat L, Pascual A, Nart J. Efficacy of soft tissue augmentation procedures on tissue thickening around dental implants: a systematic review and meta-analysis. Clin Oral Implants Res. 2022;33 Suppl 23:72-99. doi: 10.1111/clr.13920
- Montero E, Molina A, Matesanz P, Monje A, Sanz-Sánchez I, Herrera D. Efficacy of soft tissue substitutes, in comparison with autogenous grafts, in surgical procedures aiming to increase the peri-implant keratinized mucosa: a systematic review. Clin Oral Implants Res. 2022;33 Suppl 23:32-46. doi: 10.1111/clr.13751
- Rotundo R, Pancrazi GL, Grassi A, Ceresoli L, Di Domenico GL, Bonafede V. Soft tissue substitutes in periodontal and peri-implant soft tissue augmentation: a systematic review. Materials (Basel). 2024;17(5) doi: 10.3390/ma17051221
- Alfonso García SL, Parada-Sanchez MT, Arboleda Toro D. The phenotype of gingival fibroblasts and their potential use in advanced therapies. Eur J Cell Biol. 2020;99(7): 151123. doi: 10.1016/j.ejcb.2020.151123
- Chiquet M, Katsaros C, Kletsas D. Multiple functions of gingival and mucoperiosteal fibroblasts in oral wound healing and repair. Periodontol 2000. 2015;68(1):21-40. doi: 10.1111/prd.12076
- Squier CA, Kammeyer GA. The role of connective tissue in the maintenance of epithelial differentiation in the adult. Cell Tissue Res. 1983;230(3):615-630. doi: 10.1007/bf00216205
- Hsieh PC, Jin YT, Chang CW, Huang CC, Liao SC, Yuan K. Elastin in oral connective tissue modulates the keratinization of overlying epithelium. J Clin Periodontol. 2010;37(8):705-711. doi: 10.1111/j.1600-051X.2010.01542.x
- Chuhuaicura P, Rodríguez-Niklitschek C, Oporto GH, Salazar LA. Distinct molecular mechanisms in oral mucosal wound healing: translational insights and future directions. Int J Mol Sci. 2025;26(21) doi: 10.3390/ijms262110660
- Gumede DB, Abrahamse H, Houreld NN. Targeting Wnt/β- catenin signaling and its interplay with TGF-β and Notch signaling pathways for the treatment of chronic wounds. Cell Commun Signal. 2024;22(1):244. doi: 10.1186/s12964-024-01623-9
- Das S, Jegadeesan JT, Basu B. Gelatin methacryloyl (GelMA)-based biomaterial inks: process science for 3D/4D printing and current status. Biomacromolecules. 2024;25(4):2156-2221. doi: 10.1021/acs.biomac.3c01271
- Zhang X, Huang C, Huang K, et al. Living and injectable porous hydrogel microspheres promoting inflammation modulation and extracellular matrix remodeling for intervertebral disc regeneration. ACS Appl Mater Interfaces. 2025;17(42):57953-57966. doi: 10.1021/acsami.5c13982
- Wei Long N, Cian V, Boyang H, Wai Yee Y, Paulo B. Advanced bioprinting strategies for fabrication of biomimetic tissues and organs. Int J Extreme Manuf. 2025;7(6):062006. doi: 10.1088/2631-7990/adeee0
- Kim JJ, Cho D-W. Advanced strategies in 3D bioprinting for vascular tissue engineering and disease modelling using smart bioinks. Virtual Phys Prototyping. 2024;19(1):e2395470. doi: 10.1080/17452759.2024.2395470
- Schwab A, Levato R, D’Este M, Piluso S, Eglin D, Malda J. Printability and shape fidelity of bioinks in 3D bioprinting. Chem Rev. 2020;120(19):11028-11055. doi: 10.1021/acs.chemrev.0c00084
- Osi AR, Zhang H, Chen J, et al. Three- dimensional-printable thermo/photo-cross-linked methacrylated chitosan-gelatin hydrogel composites for tissue engineering. ACS Appl Mater Interfaces. 2021;13(19):22902-22913. doi: 10.1021/acsami.1c01321
- Choi JJE, Zwirner J, Ramani RS, et al. Mechanical properties of human oral mucosa tissues are site dependent: a combined biomechanical, histological and ultrastructural approach. Clin Exp Dent Res. 2020;6(6):602-611. doi: 10.1002/cre2.305
- Yue K, Trujillo-de Santiago G, Alvarez MM, Tamayol A, Annabi N, Khademhosseini A. Synthesis, properties, and biomedical applications of gelatin methacryloyl (GelMA) hydrogels. Biomaterials. 2015;73:254-271. doi: 10.1016/j.biomaterials.2015.08.045
- Guan S, Zhang XL, Lin XM, Liu TQ, Ma XH, Cui ZF. Chitosan/gelatin porous scaffolds containing hyaluronic acid and heparan sulfate for neural tissue engineering. J Biomater Sci Polym Ed. 2013;24(8):999-1014.doi: 10.1080/09205063.2012.731374
- Gong H, Agustin J, Wootton D, Zhou JG. Biomimetic design and fabrication of porous chitosan–gelatin liver scaffolds with hierarchical channel network. J Mater Sci Mater Med. 2014;25(1):113-120. doi: 10.1007/s10856-013-5061-8
- Yin J, Yan M, Wang Y, Fu J, Suo H. 3D Bioprinting of low-concentration cell-laden gelatin methacrylate (GelMA) bioinks with a two-step cross-linking strategy. ACS Appl Mater Interfaces. 2018;10(8):6849-6857. doi: 10.1021/acsami.7b16059
- Peña OA, Martin P. Cellular and molecular mechanisms of skin wound healing. Nat Rev Mol Cell Biol. 2024;25(8):599-616. doi: 10.1038/s41580-024-00715-1
- Wei K, Nguyen HN, Brenner MB. Fibroblast pathology in inflammatory diseases. J Clin Invest. 2021;131(20). doi: 10.1172/jci149538
- Hu J, Li J, Jiang J, et al. Design of synthetic collagens that assemble into supramolecular banded fibers as a functional biomaterial testbed. Nat Commun. 2022;13(1):6761. doi: 10.1038/s41467-022-34127-6
- Mascharak S, Talbott HE, Januszyk M, et al. Multi-omic analysis reveals divergent molecular events in scarring and regenerative wound healing. Cell Stem Cell. 2022;29(2):315-327.e6. doi: 10.1016/j.stem.2021.12.011
- Zeng Y, Wang T, Liu Y, et al. Wnt and Smad signaling pathways synergistically regulated the osteogenic differentiation of fibroblasts in ankylosing spondylitis. Tissue Cell. 2022;77:101852. doi: 10.1016/j.tice.2022.101852
- Sharma S, Thakur SL, Joshi SK, Kulkarni SS. Measurement of gingival thickness using digital vernier caliper and ultrasonographic method: a comparative study. J Investig Clin Dent. 2014;5(2):138-143. doi: 10.1111/jicd.12026
- Wang J, Cha S, Zhao Q, Bai D. Methods to assess tooth gingival thickness and diagnose gingival phenotypes: a systematic review. J Esthet Restor Dent. 2022;34(4):620-632. doi: 10.1111/jerd.12900
- Heil A, Schwindling FS, Jelinek C, et al. Determination of the palatal masticatory mucosa thickness by dental MRI: a prospective study analysing age and gender effects. Dentomaxillofac Radiol. 2018;47(2):20170282. doi: 10.1259/dmfr.20170282
- Schwarz L, Unger E, Gahleitner A, Rausch-Fan X, Jonke E. A novel approach for gingiva thickness measurements around lower anterior teeth by means of dental magnetic resonance imaging. Clin Oral Investig. 2023;28(1):18. doi: 10.1007/s00784-023-05459-4
- Wang Z, Wang L, Li T, et al. 3D bioprinting in cardiac tissue engineering. Theranostics. 2021;11(16):7948-7969. doi: 10.7150/thno.61621
- Liu P, Li Q, Yang Q, et al. Evaluation of the effect of 3D-bioprinted gingival fibroblast-encapsulated ADM scaffolds on keratinized gingival augmentation. J Periodontal Res. 2023;58(3):564-574. doi: 10.1111/jre.13126
- Dai Y, Wang P, Mishra A, et al. 3D bioprinting and artificial intelligence-assisted biofabrication of personalized oral soft tissue constructs. Adv Healthc Mater. 2024:e2402727. doi: 10.1002/adhm.202402727

