Induction and real-time ultrasonic monitoring of 3D cartilage-like tissue by a low shear stresses-based bioreactor

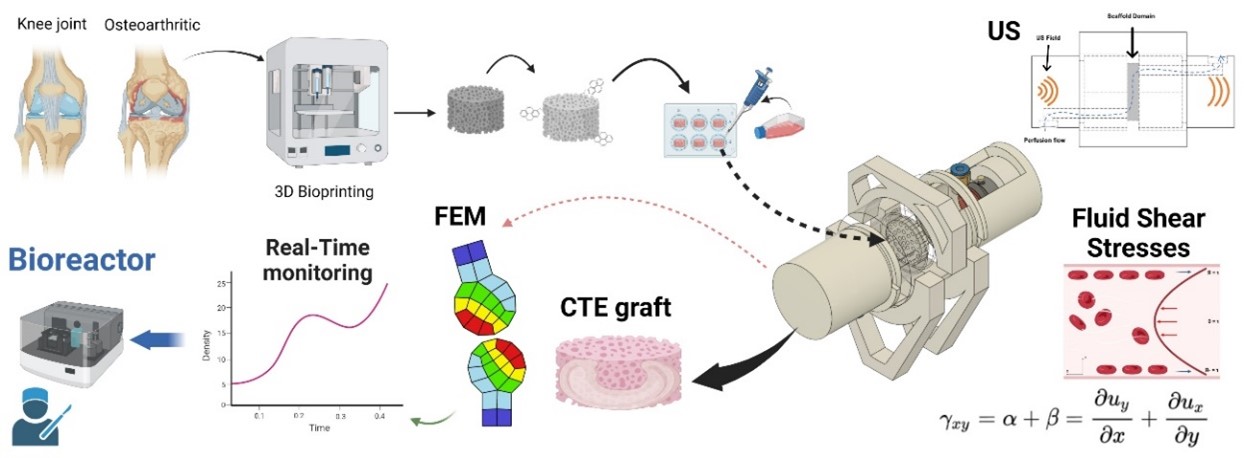
Osteoarthritis is a significant socioeconomic illness that mainly affects the articular cartilage, a tissue with a low capacity for self-healing, making it an ideal target for regenerative medicine and tissue engineering. Current interventions to treat cartilage injuries may not be completely effective. In this study, we have developed a novel bioreactor that creates viscous shear stress by flow perfusion. This bioreactor could induce ex vivo maturation of biomimetic 3D cartilage scaffolds, providing a potential solution to this problem. Infrapatellar fat pad mesenchymal stem cells (IPFP-MSCs) were used as a cellular source of the functionalized 3D scaffolds made of 1,4-butanediol thermoplastic polyurethane (bTPUe) modified with pyrene butyric acid (PBA). Our results indicate that our bioreactor induced chondrogenic differentiation, as confirmed by DNA quantification, extracellular matrix determination, and metabolic assay, without any conditioned medium. To control the biomechanical stimulation on IPFP-MSCs, a low-intensity ultrasonic transmission system has been developed and embedded in the bioreactor. Combined with a finite element model (FEM), tissue growth and differentiation can be deconvoluted in real-time from the recorded ultrasonic propagation and interaction across the graft. The FEM reconstructs this complex interaction. This is the first time a low-shear stress-based bioreactor has been reported to not only induce chondrogenic evolution but also monitor it in real time.
- Mason C, Dunnill P. A brief definition of regenerative medicine. Regenerative Med. 2007;3(1):1-5. doi: 10.2217/17460751.3.1.1
- Langer R, Vacanti JP, Vacanti CA, Atala A, Freed LE, Vunjak- Novakovic G. Tissue engineering: biomedical applications. Tissue Eng. 1995;1(2):151-161. doi: 10.1089/ten.1995.1.151
- Libby P. Inflammation in atherosclerosis. Nature. 2002;420(6917):868-874. doi: 10.1038/nature01323
- Chen S, Fu P, Wu H, Pei M. Meniscus, articular cartilage and nucleus pulposus : a comparative review of cartilage-like tissues in anatomy, development and function. Cell Tissue Res. 2017;370(1):53-70. doi: 10.1007/s00441-017-2613-0
- Houard X, Goldring MB, Berenbaum F. Homeostatic mechanisms in articular cartilage and role of inflammation in osteoarthritis. Curr Rheumatol Rep. 2013;15(11):375. doi: 10.1007/s11926-013-0375-6
- Heinegård D, Saxne T. The role of the cartilage matrix in osteoarthritis. Nat Rev Rheumatol. 2011;7(1):50-56. doi: 10.1038/nrrheum.2010.198
- Martínez R, Martínez C, Calvo R, Figueroa D. Osteoartritis (artrosis) de rodilla. Chilean J Orthop Traumatol. 2015;56(3):45-51. doi: 10.1016/j.rchot.2015.10.005
- Makris EA, Gomoll AH, Malizos KN, Hu JC, Athanasiou KA. Repair and tissue engineering techniques for articular cartilage. Nat Rev Rheumatol. 2015;11(1):21-34. doi: 10.1038/nrrheum.2014.157
- European Medicines Agency. Spherox. Spherox - EMEA/ H/C/002736 - II/0020; 2021. Accessed July 28, 2022. https://www.ema.europa.eu/en/medicines/human/EPAR/ spherox
- Vinatier C, Guicheux J. Cartilage tissue engineering: from biomaterials and stem cells to osteoarthritis treatments. Ann Phys Rehabil Med. 2016;59(3):139-144. doi: 10.1016/j.rehab.2016.03.002
- Legendre F, Ollitrault D, Hervieu M, et al. Enhanced hyaline cartilage matrix synthesis in collagen sponge scaffolds by using siRNA to stabilize chondrocytes phenotype cultured with bone morphogenetic protein-2 under hypoxia. Tissue Eng Part C Methods. 2013;19(7):550-567. doi: 10.1089/ten.TEC.2012.0508
- Bentley G, Biant LC, Vijayan S, Macmull S, Skinner JA, Carrington RWJ. Minimum ten-year results of a prospective randomised study of autologous chondrocyte implantation versus mosaicplasty for symptomatic articular cartilage lesions of the knee. J Bone Joint Surg Br. 2012;94(4):504-509. doi: 10.1302/0301-620X.94B4.27495
- Englund M, Guermazi A, Gale D, et al. Incidental meniscal findings on knee mri in middle-aged and elderly persons. N Engl J Med. 2008;359(11):1108-1115. doi: 10.1056/NEJMoa0800777
- Martel-Pelletier J. Pathophysiology of osteoarthritis. Osteoarthritis Cartilage. 1998;6(6):374-376. doi: 10.1053/joca.1998.0140
- Hodge WA, Fijan RS, Carlson KL, Burgess RG, Harris WH, Mann RW. Contact pressures in the human hip joint measured in vivo. Proc Natl Acad Sci USA. 1986;83(9): 2879-2883. doi: 10.1073/pnas.83.9.2879
- Park S, Krishnan R, Nicoll SB, Ateshian GA. Cartilage interstitial fluid load support in unconfined compression. J Biomech. 2003;36(12):1785-1796. doi: 10.1016/S0021-9290(03)00231-8
- Mow VC, Kuei SC, Lai WM, Armstrong CG. Biphasic creep and stress relaxation of articular cartilage in compression: theory and experiments. J Biomech Eng. 1980;102(1):73-84. doi: 10.1115/1.3138202
- Armstrong CG, Lai WM, Mow VC. An analysis of the unconfined compression of articular cartilage. J Biomech Eng. 1984;106(2):165-173. doi: 10.1115/1.3138475
- Tatsumura M, Sakane M, Ochiai N, Mizuno S. Off-loading of cyclic hydrostatic pressure promotes production of extracellular matrix by chondrocytes. Cells Tissues Organs. 2013;198(6):405-413. doi: 10.1159/000360156
- Tan AR, Dong EY, Andry JP, Bulinski JC, Ateshian GA, Hung CT. Coculture of engineered cartilage with primary chondrocytes induces expedited growth. Clin Orthop Relat Res. 2011;469(10): 2735-2743. doi: 10.1007/s11999-011-1772-7
- Sampat SR, O’Connell GD, Fong JV, Alegre-Aguarón E, Ateshian GA, Hung CT. Growth factor priming of synovium-derived stem cells for cartilage tissue engineering. Tissue Eng Part A. 2011;17(17-18):2259-2265. doi: 10.1089/ten.tea.2011.0155
- Rad MR, Eghbal MJ, Nadjmi N, et al. Polymeric scaffolds in tissue engineering: a literature review. J Biomed Mater Res B Appl Biomater. 2015;105(2):431-459. doi: 10.1002/jbm.b.33547
- Jang TS, Park SJ, Lee JE, et al. Topography-supported nanoarchitectonics of hybrid scaffold for systematically modulated bone regeneration and remodeling. Adv Funct Mater. 2022;32(51):2206863. doi: 10.1002/adfm.202206863
- Zhou X, Zhou G, Junka R, et al. Fabrication of polylactic acid (PLA)-based porous scaffold through the combination of traditional bio-fabrication and 3D printing technology for bone regeneration. Colloids Surf B Biointerfaces. 2021;197:111420. doi: 10.1016/j.colsurfb.2020.111420
- Chocarro-Wrona C, de Vicente J, Antich C, et al. Validation of the 1,4-butanediol thermoplastic polyurethane as a novel material for 3D bioprinting applications. Bioeng Transl Med. 2021;6(1):e10192. doi: 10.1002/btm2.10192
- Chen C, Bang S, Cho Y, et al. Research trends in biomimetic medical materials for tissue engineering: 3D bioprinting, surface modification, nano/micro-technology and clinical aspects in tissue engineering of cartilage and bone. Biomater Res. 2016;20(1):10. doi: 10.1186/s40824-016-0057-3
- Zhang H, Hollister S. Comparison of bone marrow stromal cell behaviors on poly(caprolactone) with or without surface modification: studies on cell adhesion, survival and proliferation. J Biomater Sci Polym Ed. 2009;20(14): 1975-1993. doi: 10.1163/156856208X396074
- Hinnemo M, Zhao J, Ahlberg P, et al. On monolayer formation of pyrenebutyric acid on graphene. Langmuir. 2017;33(15):3588-3593. doi: 10.1021/acs.langmuir.6b04237
- Douglas T, Haugen HJ. Coating of polyurethane scaffolds with collagen: Comparison of coating and cross-linking techniques. J Mater Sci Mater Med. 2008;19(8):2713-2719. doi: 10.1007/s10856-008-3393-6
- Vinardell T, Sheehy EJ, Buckley CT, Kelly DJ. A comparison of the functionality and in vivo phenotypic stability of cartilaginous tissues engineered from different stem cell sources. Tissue Eng Part A. 2012;18(11-12):1161-1170. doi: 10.1089/ten.TEA.2011.0544
- Bruder SP, Jaiswal N, Haynesworth SE. Growth kinetics, self‐renewal, and the osteogenic potential of purified human mesenchymal stem cells during extensive subcultivation and following cryopreservation. J Cell Biochem. 1997;64(2): 278-294. doi: 10.1002/(sici)1097-4644(199702)64:2<278::aid-jcb11>3.0.co;2-f
- Jimenez-Puerta GJ, Marchal JA, López-Ruiz E, Gálvez- Martín P. Role of mesenchymal stromal cells as therapeutic agents: potential mechanisms of action and implications in their clinical use. J Clin Med. 2020;9(2):445. doi: 10.3390/jcm9020445
- López-Ruiz E, Perán M, Cobo-Molinos J, et al. Chondrocytes extract from patients with osteoarthritis induces chondrogenesis in infrapatellar fat pad-derived stem cells. Osteoarthritis Cartilage. 2013;21(1):246-258. doi: 10.1016/j.joca.2012.10.007
- Ghosh S, Laha M, Mondal S, Sengupta S, Kaplan DL. In vitro model of mesenchymal condensation during chondrogenic development. Biomaterials. 2009;30(33):6530-6540. doi: 10.1016/j.biomaterials.2009.08.019
- Juhász T, Matta C, Somogyi C, et al. Mechanical loading stimulates chondrogenesis via the PKA/CREB-Sox9 and PP2A pathways in chicken micromass cultures. Cell Signal. 2014;26(3):468-482. doi: 10.1016/j.cellsig.2013.12.001
- Wang Y, Huang Z, Nayak PS, et al. Strain-induced differentiation of fetal type II epithelial cells is mediated via integrin α6β1-ADAM17/TACE signaling pathway. J Biol Chem. 2013;288(35):25646-25657. doi: 10.1074/jbc.M113.473777
- Ross TD, Coon BG, Yun S, et al. Integrins in mechanotransduction. Curr Opin Cell Biol. 2013;25(5):613-618. doi: 10.1016/j.ceb.2013.05.006
- Xu X, Liu S, Liu H, et al. Piezo channels: awesome mechanosensitive structures in cellular mechanotransduction and their role in bone. Int J Mol Sci. 2021;22(12):6429. doi: 10.3390/ijms22126429
- Agarwal P, Lee HP, Smeriglio P, et al. A dysfunctional TRPV4– GSK3β pathway prevents osteoarthritic chondrocytes from sensing changes in extracellular matrix viscoelasticity. Nat Biomed Eng. 2021;5(12):1472-1484. doi: 10.1038/s41551-021-00691-3
- Subramanian A, Budhiraja G, Sahu N. Chondrocyte primary cilium is mechanosensitive and responds to low-intensity-ultrasound by altering its length and orientation. Int J Biochem Cell Biol. 2017;91(Pt A):60-64. doi: 10.1016/j.biocel.2017.08.018
- Taheri S, Ghazali HS, Ghazali ZS, Bhattacharyya A, Noh I. Progress in biomechanical stimuli on the cell-encapsulated hydrogels for cartilage tissue regeneration. Biomater Res. 2023;27(1):22. doi: 10.1186/s40824-023-00358-x
- Tsimbouri PM, Childs PG, Pemberton GD, et al. Stimulation of 3D osteogenesis by mesenchymal stem cells using a nanovibrational bioreactor. Nat Biomed Eng. 2017;1(9): 758-770. doi: 10.1038/s41551-017-0127-4
- Bahmaee H, Owen R, Boyle L, et al. Design and evaluation of an osteogenesis-on-a-chip microfluidic device incorporating 3D cell culture. Front Bioeng Biotechnol. 2020;8(September):557111. doi: 10.3389/fbioe.2020.557111
- Callejas A, Melchor J, Faris IH, Rus G. Viscoelastic model characterization of human cervical tissue by torsional waves. J Mech Behav Biomed Mater. 2021;115:104261. doi: 10.1016/j.jmbbm.2020.104261
- Dominici M, Le Blanc K, Mueller I, et al. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy. 2006;8(4):315-317. doi: 10.1080/14653240600855905
- Martínez-Moreno D, Jiménez G, Chocarro-Wrona C, et al. Pore geometry influences growth and cell adhesion of infrapatellar mesenchymal stem cells in biofabricated 3D thermoplastic scaffolds useful for cartilage tissue engineering. Mater Sci Eng C Mater Biol Appl. 2021;122:111933. doi: 10.1016/j.msec.2021.111933
- Watanabe T, Sassa K. Velocity and amplitude of P-waves transmitted through fractured zones composed of multiple thin low-velocity layers. Int J Rock Mech Mining Sci Geomech Abstr. 1995;32(4):313-324.
- Tarrazó-Serrano D, Castiñeira-Ibáñez S, Sánchez-Aparisi E, Uris A, Rubio C. MRI compatible planar material acoustic lenses. Appl Sci. 2018;8(12):2634. doi: 10.3390/app8122634
- Poon C. Measuring the density and viscosity of culture media for optimized computational fluid dynamics analysis of in vitro devices. J Mech Behav Biomed Mater. 2022;126:105024. doi: 10.1016/j.jmbbm.2021.105024
- Schlichting H, Gersten K. Boundary-Layer Theory. Springer Berlin Heidelberg; 2017. https://books.google.es/books?id=bOUyDQAAQBAJ
- Bancroft GN, Sikavitsas VI, Van Den Dolder J, et al. Fluid flow increases mineralized matrix deposition in 3D perfusion culture of marrow stromal osteoblasts in a dose-dependent manner. Proc Natl Acad Sci U S A. 2002;99(20):12600-12605. doi: 10.1073/pnas.202296599
- Antoniou A, Evripidou N, Giannakou M, Constantinides G, Damianou C. Acoustical properties of 3D printed thermoplastics. J Acoust Soc Am. 2021;149(4):2854-2864. doi: 10.1121/10.0004772
- Hung KC, Tseng CS, Hsu SH. Synthesis and 3D Printing of biodegradable polyurethane elastomer by a water-based process for cartilage tissue engineering applications. Adv Healthc Mater. 2014;3(10):1578-1587. doi: 10.1002/adhm.201400018
- Wang C, Feng N, Chang F, et al. Injectable cholesterol-enhanced stereocomplex polylactide thermogel loading chondrocytes for optimized cartilage regeneration. Adv Healthc Mater. 2019;8(14):1-10. doi: 10.1002/adhm.201900312
- Wu J, Fu L, Yan Z, et al. Hierarchical porous ECM scaffolds incorporating GDF-5 fabricated by cryogenic 3D printing to promote articular cartilage regeneration. Biomater Res. 2023;27(1):7. doi: 10.1186/s40824-023-00349-y
- Lanir Y. Mechanisms of residual stress in soft tissues. J Biomech Eng. 2009;131(4):044506. doi: 10.1115/1.3049863
- Hotaling NA, Tang L, Irvine DJ, Babensee JE. Biomaterial strategies for immunomodulation. Annu Rev Biomed Eng. 2015;17:317-349. doi: 10.1146/annurev-bioeng-071813-104814
- Humphrey JD, Dufresne ER, Schwartz MA. Mechanotransduction and extracellular matrix homeostasis. Nat Rev Mol Cell Biol. 2014;15(12):802-812. doi: 10.1038/nrm3896
- Koo YW, Lim CS, Darai A, et al. Shape-memory collagen scaffold combined with hyaluronic acid for repairing intervertebral disc. Biomater Res. 2023;27(1):26. doi: 10.1186/s40824-023-00368-9
- Gonalves A, Costa P, Rodrigues MT, Dias IR, Reis RL, Gomes ME. Effect of flow perfusion conditions in the chondrogenic differentiation of bone marrow stromal cells cultured onto starch based biodegradable scaffolds. Acta Biomater. 2011;7(4):1644-1652. doi: 10.1016/j.actbio.2010.11.044
- Bernhard JC, Hulphers E, Rieder B, et al. Perfusion enhances hypertrophic chondrocyte matrix deposition, but not the bone formation. Tissue Eng Part A. 2018;24(11- 12):1022-1033. doi: 10.1089/ten.tea.2017.0356
- Muhammad H, Rais Y, Miosge N, Ornan EM. The primary cilium as a dual sensor of mechanochemical signals in chondrocytes. Cell Mol Life Sci. 2012;69(13):2101-2107. doi: 10.1007/s00018-011-0911-3
- Wann AKT, Zuo N, Haycraft CJ, et al. Primary cilia mediate mechanotransduction through control of ATP‐induced Ca2+ signaling in compressed chondrocytes. FASEB J. 2012;26(4):1663-1671.
- Knight AKTWMM. Primary cilia elongation in response to interleukin-1 mediates the inflammatory response. Cell Mol Life Sci. 2012;69(17):2967-2977. doi: 10.1007/s00018-012-0980-y
- Kock LM, Malda J, Dhert WJA, Ito K, Gawlitta D. Flow-perfusion interferes with chondrogenic and hypertrophic matrix production by mesenchymal stem cells. J Biomech. 2014;47(9):2122-2129. doi: 10.1016/j.jbiomech.2013.11.006
- Son B, Kim HD, Kim M, et al. Physical stimuli-induced chondrogenic differentiation of mesenchymal stem cells using magnetic nanoparticles. Adv Healthc Mater. 2015;4(9):1339-1347. doi: 10.1002/adhm.201400835
- Molladavoodi S, Robichaud M, Wulff D, Gorbet M. Corneal epithelial cells exposed to shear stress show altered cytoskeleton and migratory behaviour. PLoS One. 2017;12(6):1-16. doi: 10.1371/journal.pone.0178981
- Wang X, Lin Q, Zhang T, et al. Low-intensity pulsed ultrasound promotes chondrogenesis of mesenchymal stem cells via regulation of autophagy. Stem Cell Res Ther. 2019;10(1):41. doi: 10.1186/s13287-019-1142-z
- Allen JS, Roy RA, Church CC. On the role of shear viscosity in mediating inertial cavitation from short-pulse, megahertz-frequency ultrasound. IEEE Trans Ultrason Ferroelectr Freq Control. 1997;44(4):743-751. doi: 10.1109/58.655189
- Van de Walle AB, Moore MC, McFetridge PS. Sequential adaptation of perfusion and transport conditions significantly improves vascular construct recellularization and biomechanics. J Tissue Eng Regen Med. 2020;14(3):510-520. doi: 10.1002/term.3015
- Provin C, Takano K, Sakai Y, Fujii T, Shirakashi R. A method for the design of 3D scaffolds for high-density cell attachment and determination of optimum perfusion culture conditions. J Biomech. 2008;41(7):1436-1449. doi: 10.1016/j.jbiomech.2008.02.025
- Stott NS, Jiang TX, Chuong CM. Successive formative stages of precartilaginous mesenchymal condensations in vitro: modulation of cell adhesion by Wnt-7A and BMP-2. J Cell Physiol. 1999;180(3):314-324. doi: 10.1002/(SICI)1097-4652(199909)180:3<314::AID-JCP2>3.0.CO;2-Y
- Tschaikowsky M, Brander S, Barth V, et al. The articular cartilage surface is impaired by a loss of thick collagen fibers and formation of type I collagen in early osteoarthritis. Acta Biomater. 2022;146: 274-283. doi: 10.1016/j.actbio.2022.04.036
- Li Y, Toole BP, Dealy CN, Kosher RA. Hyaluronan in limb morphogenesis. Dev Biol. 2007;305(2):411-420.
- Akiyama H, Lyons JP, Mori-Akiyama Y, et al. Interactions between Sox9 and β-catenin control chondrocyte differentiation. Genes Dev. 2004;18(9):1072-1087. doi: 10.1101/gad.1171104
- Koo MA, Kang JK, Lee MH, et al. Stimulated migration and penetration of vascular endothelial cells into poly (L-lactic acid) scaffolds under flow conditions. Biomater Res. 2014;18(1):7. doi: 10.1186/2055-7124-18-7
- Martínez-Moreno D, Venegas-Bustos D, Guillermo R, Gálvez-Martín P, Jiménez G, Marchal JA. Chondro-inductive b-TPUe-based functionalized scaffolds for application in cartilage tissue engineering. Adv Healthc Mater. 2022;11(19):e2200251. doi: 10.1002/adhm.202200251
- Zhao Q, Eberspaecher H, Lefebvre V, De Crombrugghe B. Parallel expression of Sox9 and Col2a1 in cells undergoing chondrogenesis. Dev Dyn. 1997;209(4): 377-386. doi: 10.1002/(SICI)1097-0177(199708)209:4<377::AID-AJA5>3.0.CO;2-F
- Flöter M, Bittar CK, Zabeu JL, Carneiro AC. Review of comparative studies between bone densitometry and quantitative ultrasound of the calcaneus in osteoporosis. Acta Reumatol Port. 2011;36(4):327-335.
- Zimmermann R, Fiabane L, Gasteuil Y, Volk R, Pinton JF. Characterizing flows with an instrumented particle measuring Lagrangian accelerations. New J Phys. 2013;15(1):15018. doi: 10.1088/1367-2630/15/1/015018
- Kanungo BP, Gibson LJ. Density–property relationships in mineralized collagen–glycosaminoglycan scaffolds. Acta Biomater. 2009;5(4):1006-1018. doi: 10.1016/j.actbio.2008.11.029

