A multimodal framework for early detection, risk assessment, and biomarker-guided therapy in prostate cancer: Considerations for sub-Saharan African populations

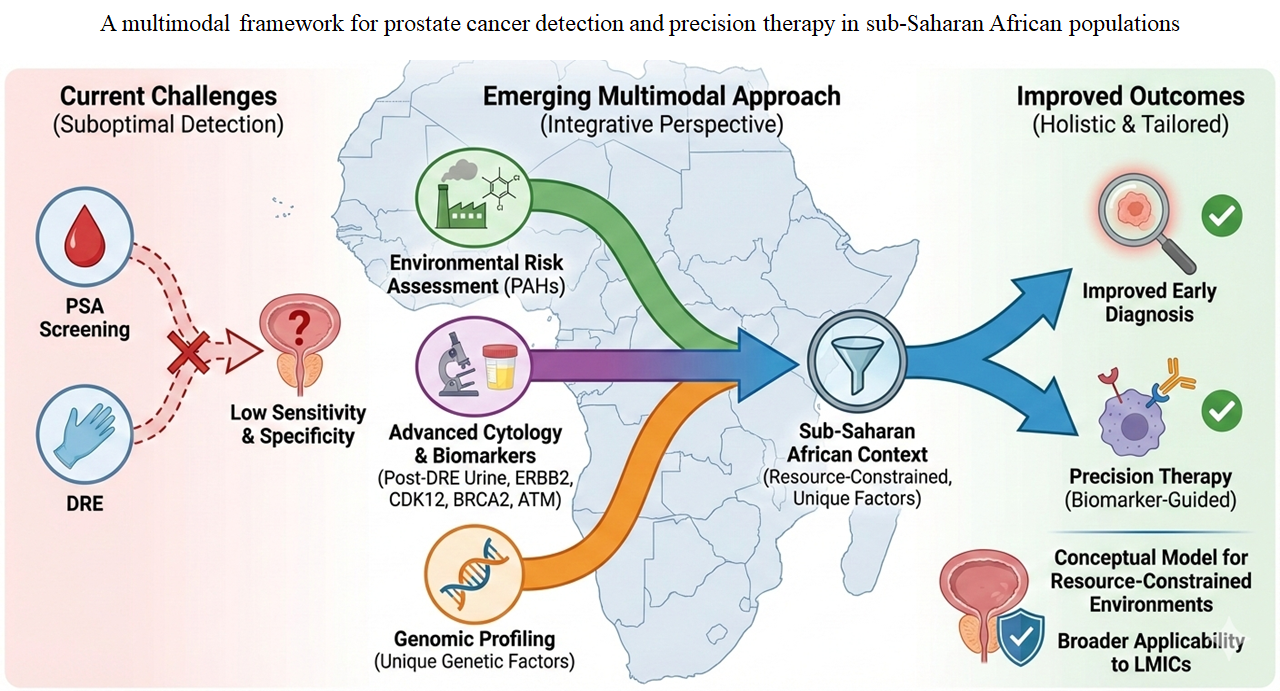
Prostate cancer remains a major global public health concern, with rising incidence rates worldwide, especially in sub-Saharan Africa. Traditional screening modalities, which rely solely on prostate-specific antigen concentration measurement and digital rectal examination (DRE), have shown poor sensitivity and specificity. Hence, this review critically evaluates emerging multimodal diagnostic strategies, including environmental risk assessment, new cytological and immunohistochemical biomarkers, and genomic profiling, to improve early detection and enable precision therapy for prostate cancer. We examine current information on polycyclic aromatic hydrocarbons as environmental risk indicators, the optimization of post-DRE urine cytology, cytological examination of expressed prostate secretions, and the clinical usefulness of biomarkers such as ERBB2, CDK12, BRCA2, and ATM. Specific attention is paid to the African background, where unique genetic and environmental factors necessitate the development of tailored diagnostic algorithms. This integrative approach provides a theoretical framework for developing comprehensive diagnostic pathways that can enhance early detection rates and deliver individualized treatment in resource-constrained environments. Although the target population of this review is sub-Saharan Africans, the principles and frameworks presented have broader applicability to other low- and middle-income nations facing similar challenges.
- Ferlay J, Colombet M, Soerjomataram I, et al. Cancer statistics for the year 2020: An overview. Int J Cancer. 2021;149(4):778-789. doi: 10.1002/ijc.33588
- Okwor CJ, Nnakenyi ID, Agbo EO, Nweke M. Sensitivity and specificity of prostate-specific antigen and its surrogates towards the detection of prostate cancer in sub-Saharan Africa: a systematic review with meta-analysis. Afr J Urol. 2023;29(1):41. doi: 10.1186/s12301-023-00372-4
- Iheanacho CO, Enechukwu OH. Epidemiology of prostate cancer in Nigeria: a mixed methods systematic review. Cancer Causes Control. 2025;36(1):1-12. doi: 10.1007/s10552-024-01917-w
- Mottet N, Van Den Bergh RCN, Briers E, et al. EAU-EANMESTRO-ESUR-SIOG Guidelines on Prostate Cancer—2020 Update. Part 1: Screening, Diagnosis, and Local Treatment with Curative Intent. Eur Urol. 2021;79(2):243-262. doi: 10.1016/j.eururo.2020.09.042
- Sequeira JP, Salta S, Freitas R, et al. Biomarkers for Pre-Treatment Risk Stratification of Prostate Cancer Patients: A Systematic Review. Cancers. 2024;16(7):1363. doi: 10.3390/cancers16071363
- Butler EN, Umar A, Heckman-Stoddard BM, Kundrod KA, Signorello LB, Castle PE. Redefining precision cancer prevention to promote health equity. Trends Cancer. 2022;8(4):295-302. doi: 10.1016/j.trecan.2022.01.009
- Peng B, Dong Q, Li F, Wang T, Qiu X, Zhu T. A Systematic Review of Polycyclic Aromatic Hydrocarbon Derivatives: Occurrences, Levels, Biotransformation, Exposure Biomarkers, and Toxicity. Environ Sci Technol. 2023;57(41):15314-15335. doi: 10.1021/acs.est.3c03170
- Zhang D, Xu X, Wei Y, et al. Prognostic Role of DNA Damage Response Genes Mutations and their Association With the Sensitivity of Olaparib in Prostate Cancer Patients. Cancer Control. 2022;29. doi: 10.1177/10732748221129451
- Feng Y, Li Z, Li W. Polycyclic Aromatic Hydrocarbons (PAHs): Environmental Persistence and Human Health Risks. Nat Prod Commun. 2025;20(1). doi: 10.1177/1934578X241311451
- Ephraim-Emmanuel BC, Ordinioha B. Exposure and Public Health Effects of Polycyclic Aromatic Hydrocarbon Compounds in Sub-Saharan Africa: A Systematic Review. Int J Toxicol. 2021;40(3):250-269. doi: 10.1177/10915818211002487
- Moorthy B, Chu C, Carlin DJ. Polycyclic Aromatic Hydrocarbons: From Metabolism to Lung Cancer. Toxicol Sci. 2015;145(1):5-15. doi: 10.1093/toxsci/kfv040
- Volesky-Avellaneda KD, Morais S, Walter SD, et al. Cancers Attributable to Infections in the US in 2017: A Meta-Analysis. JAMA Oncol. 2023;9(12):1678. doi: 10.1001/jamaoncol.2023.4273
- Moradpour Z, Barik A, Jorjani G, et al. Prostate cancer incidence and mortality linked to metalworking fluid exposure: a systematic review and meta-analysis. Front Oncol. 2025;14:1491159. doi: 10.3389/fonc.2024.1491159
- Monllor-Tormos A, Garcia-Vigara A, Morgan O, et al. Mediterranean diet for cancer prevention and survivorship. Maturitas. 2023;178:107841. doi: 10.1016/j.maturitas.2023.107841
- Langmann F, Ibsen DB, Johnston LW, Perez-Cornago A, Dahm CC. Legumes as a Substitute for Red and Processed Meat, Poultry or Fish, and the Risk of Non-Alcoholic Fatty Liver Disease in a Large Cohort. J Hum Nutr Diet. 2025;38(1):e70004. doi: 10.1111/jhn.70004
- Montano L, Baldini GM, Piscopo M, et al. Polycyclic Aromatic Hydrocarbons (PAHs) in the Environment: Occupational Exposure, Health Risks and Fertility Implications. Toxics. 2025;13(3):151. doi: 10.3390/toxics13030151
- Nsonwu-Anyanwu AC, Helal M, Khaled A, et al. Urinary Biomonitoring and Cancer Risk Assessment of Polycyclic Aromatic Hydrocarbon Exposure in Relation to Water Intake in Calabar, Nigeria. Expo Health. 2025;17(3):875-886. doi: 10.1007/s12403-025-00704-5
- Amachree D, Eli AA, Lazarus OT, Franca OA. Impact of Anthropogenic Pressures on the Physico-Chemical Characteristics and Quality of Surface Water from Amadi Creek, Port Harcourt, Rivers State, Nigeria: Implication to Water Quality and Ecological Health. BJES. 2025;13(2):1-20. doi: 10.37745/bjes.2013/vol13n3120
- Shaffer RM, Lee AL, Nachman R, Christensen K, Bateson TF. A Perspective from US Environmental Protection Agency (EPA) Scientists: How Your Epidemiologic Analyses Can Inform the Human Health Risk Assessment Process. Environ Health Perspect. 2025;133(3-4):045001. doi: 10.1289/EHP15203
- Styszko K, Pamuła J, Pac A, Sochacka-Tatara E. Biomarkers for polycyclic aromatic hydrocarbons in human excreta: recent advances in analytical techniques—a review. Environ Geochem Health. 2023;45(10):7099-7113. doi: 10.1007/s10653-023-01699-1
- Xu J, Liu Y, Zhang Q, et al. DNA damage, serum metabolomic alteration and carcinogenic risk associated with low-level air pollution. Environ Pollut. 2022;297:118763. doi: 10.1016/j.envpol.2021.118763
- Young AS, Mullins CE, Sehgal N, et al. The need for a cancer exposome atlas: a scoping review. JNCI Cancer Spectr. 2025;9(1):pkae122. doi: 10.1093/jncics/pkae122
- Gomella PT, Leong JY, Gomella LG, et al. Non-DRE Voided Urine Test to Diagnose Prostate Cancer: Updated Results. Diagnostics. 2025;15(5):607. doi: 10.3390/diagnostics15050607
- Van Neste L, Hendriks RJ, Dijkstra S, et al. Detection of High-grade Prostate Cancer Using a Urinary Molecular Biomarker–Based Risk Score. Eur Urol. 2016;70(5):740-748. doi: 10.1016/j.eururo.2016.04.012
- Hashim MA, Arshia A, Rehman S, Chandra A. Pitfalls in Urinary Tract Cytopathology. Acta Cytol. 2024;68(3):250-259. doi: 10.1159/000537737
- Abe H, Kawahara A, Akiba J, Yamaguchi R. Advances in diagnostic liquid‐based cytology. Cytopathology. 2024;35(6):682-694. doi: 10.1111/cyt.13405
- McKiernan J, Noerholm M, Tadigotla V, et al. A urine-based Exosomal gene expression test stratifies risk of high-grade prostate Cancer in men with prior negative prostate biopsy undergoing repeat biopsy. BMC Urol. 2020;20(1):138. doi: 10.1186/s12894-020-00712-4
- Kemesiene J, Nicolau C, Cholstauskas G, et al. Usefulness of urinary biomarker-based risk score and multiparametric MRI for clinically significant prostate cancer detection in biopsy-naive patients. Abdom Radiol. 2025;50(8):3816-3826. doi: 10.1007/s00261-024-04727-5
- Hendriks RJ, Van Der Leest MMG, Israel B, et al. Clinical use of the SelectMDx urinary-biomarker test with or without mpMRI in prostate cancer diagnosis: a prospective, multicenter study in biopsy-naive men. Prostate CancerProstatic Dis. 2021;24(4):1110-1119. doi: 10.1038/s41391-021-00367-8
- Prakash P, Verma S, Gupta S. Influence of microbiome in intraprostatic inflammation and prostate cancer. Prostate. 2024;84(13):1179-1188. doi: 10.1002/pros.24756
- Koshrovski-Michael S, Ajamil DR, Dey P, et al. Two-in-one nanoparticle platform induces a strong therapeutic effect of targeted therapies in P-selectin–expressing cancers. Sci Adv. 2024;10(50):eadr4762. doi: 10.1126/sciadv.adr4762
- Allison DB, VandenBussche CJ. Diagnostic challenges in urinary cytology: Practical insights from The Paris System for Reporting Urinary Cytology. Cancer Cytopathol. 2025;133(8):e70033. doi: 10.1002/cncy.70033
- Epstein JI, Amin MB, Fine SW, et al. The 2019 Genitourinary Pathology Society (GUPS) White Paper on Contemporary Grading of Prostate Cancer. Arch Pathol Lab Med. 2021;145(4):461-493. doi: 10.5858/arpa.2020-0015-RA
- Erdogdu IH, Tuna B, Yorukoglu K, Montironi R. Biomarkers in Urinary Tract and Male Genital System Carcinomas. In: Sarioglu S, Sagol O, Aysal A, eds. Biomarkers in Carcinoma of Unknown Primary. Springer; 2022:237-273. doi: 10.1007/978-3-030-84432-5_9
- McNeal JE. The zonal anatomy of the prostate. Prostate. 1981;2(1):35-49. doi: 10.1002/pros.2990020105
- Hao S, Discacciati A, Eklund M, et al. Cost-effectiveness of Prostate Cancer Screening Using Magnetic Resonance Imaging or Standard Biopsy Based on the STHLM3-MRI Study. JAMA Oncol. 2023;9(1):88. doi: 10.1001/jamaoncol.2022.5252
- Patel J, Klopper J, Cottrill EE. Molecular diagnostics in the evaluation of thyroid nodules: Current use and prospective opportunities. Front Endocrinol. 2023;14:1101410. doi: 10.3389/fendo.2023.1101410
- Sun RL, Liao PY, Liao YT, et al. Real-world therapeutic strategies and survival outcomes in advanced HER2-mutant non-small cell lung cancer. J Chin Med Assoc. 2025;88(4):307-315. doi: 10.1097/JCMA.0000000000001212
- Kim SW, Yu H, Kim Y, et al. HER2 overexpression predicts pathological T2 stage and improved survival in de novo muscle-invasive bladder cancer after immediate radical cystectomy: a retrospective cohort study. Int J Surg. 2024;110(2):847-858. doi: 10.1097/JS9.0000000000000859
- Narita Y, Mizuno T, Ishizuka Y, et al. Clinicopathological and prognostic significance of HER2-low expression in advanced gastric cancer: a retrospective observational study. Oncologist. 2025;30(7):oyae328. doi: 10.1093/oncolo/oyae328
- Quaquarini E, Grillo F, Gervaso L, et al. Prognostic and Predictive Roles of HER2 Status in Non-Breast and Non-Gastroesophageal Carcinomas. Cancers. 2024;16(18):3145. doi: 10.3390/cancers16183145
- Wolff AC, Somerfield MR, Dowsett M, et al. Human Epidermal Growth Factor Receptor 2 Testing in Breast Cancer. Arch Pathol Lab Med. 2023;147(9):993-1000. doi: 10.5858/arpa.2023-0950-SA
- Tilki D, Van Den Bergh RCN, Briers E, et al. EAU-EANMESTRO-ESUR-ISUP-SIOG Guidelines on Prostate Cancer. Part II—2024 Update: Treatment of Relapsing and Metastatic Prostate Cancer. Eur Urol. 2024;86(2):164-182. doi: 10.1016/j.eururo.2024.04.010
- Mateo J, Steuten L, Aftimos P, et al. Delivering precision oncology to patients with cancer. Nat Med. 2022;28(4):658-665. doi: 10.1038/s41591-022-01717-2
- Magnuson B, Bedi K, Narayanan IV, et al. CDK12 regulates co-transcriptional splicing and RNA turnover in human cells. iScience. 2022;25(9):105030. doi: 10.1016/j.isci.2022.105030
- Nguyen CB, Reimers MA, Perera C, et al. Evaluating Immune Checkpoint Blockade in Metastatic Castration-Resistant Prostate Cancers with Deleterious CDK12 Alterations in the Phase 2 IMPACT Trial. Clin Cancer Res. 2024;30(15):3200-3210. doi: 10.1158/1078-0432.CCR-24-0400
- Viswanathan SR, Ha G, Hoff AM, et al. Structural Alterations Driving Castration-Resistant Prostate Cancer Revealed by Linked-Read Genome Sequencing. Cell. 2018;174(2):433-447.e19. doi: 10.1016/j.cell.2018.05.036
- Bang S, Won D, Shin S, et al. Circulating Tumor DNA Analysis on Metastatic Prostate Cancer with Disease Progression. Cancers. 2023;15(15):3998. doi: 10.3390/cancers15153998
- Antonarakis ES, Isaacsson Velho P, Fu W, et al. CDK12- Altered Prostate Cancer: Clinical Features and Therapeutic Outcomes to Standard Systemic Therapies, Poly (ADPRibose) Polymerase Inhibitors, and PD-1 Inhibitors. JCO Precis Oncol. 2020;(4):370-381. doi: 10.1200/PO.19.00399
- Pritchard CC, Mateo J, Walsh MF, et al. Inherited DNARepair Gene Mutations in Men with Metastatic Prostate Cancer. N Engl J Med. 2016;375(5):443-453. doi: 10.1056/NEJMoa1603144
- Olmos D, Lorente D, Alameda D, et al. Treatment patterns and outcomes in metastatic castration-resistant prostate cancer patients with and without somatic or germline alterations in homologous recombination repair genes. Ann Oncol. 2024;35(5):458-472. doi: 10.1016/j.annonc.2024.01.011
- Marino F, Totaro A, Gandi C, et al. Germline mutations in prostate cancer: a systematic review of the evidence for personalized medicine. Prostate Cancer Prostatic Dis. 2023;26(4):655-664. doi: 10.1038/s41391-022-00609-3
- Lee S, Park I, Ahn B, et al. Evaluation of intraductal carcinoma and invasive cribriform carcinoma as predictors of genetic mutations in systemic treatment-naive prostate cancer patients. BMC Cancer. 2025;25(1):1736. doi: 10.1186/s12885-025-15001-2
- De Bono J, Mateo J, Fizazi K, et al. Olaparib for Metastatic Castration-Resistant Prostate Cancer. N Engl J Med. 2020;382(22):2091-2102. doi: 10.1056/NEJMoa1911440
- Rajwa P, Quhal F, Pradere B, et al. Prostate cancer risk, screening and management in patients with germline BRCA1/2 mutations. Nat Rev Urol. 2023;20(4):205-216. doi: 10.1038/s41585-022-00680-4
- Spratt DE, Srinivas S, Adra N, et al. Prostate Cancer, Version 3.2026, NCCN Clinical Practice Guidelines In Oncology. J Natl Compr Canc Netw. 2025;23(11):469-493. doi: 10.6004/jnccn.2025.0052
- Sibomana O. Genetic Diversity Landscape in African Population: A Review of Implications for Personalized and Precision Medicine. PGPM. 2024;17:487-496. doi: 10.2147/PGPM.S485452
- Fan X, Xie N, Chen J, et al. Multiparametric MRI and Machine Learning Based Radiomic Models for Preoperative Prediction of Multiple Biological Characteristics in Prostate Cancer. Front Oncol. 2022;12:839621. doi: 10.3389/fonc.2022.839621
- Visser WCH, De Jong H, Steyaert S, Melchers WJG, Mulders PFA, Schalken JA. Clinical use of the mRNA urinary biomarker SelectMDx test for prostate cancer. Prostate Cancer Prostatic Dis. 2022;25(3):583-589. doi: 10.1038/s41391-022-00562-1
- Alobid S. Targeting the PI3K/AKT/mTOR signaling pathway in prostate cancer: Molecular dysregulation, therapeutic advances, and future directions. Saudi Pharm J. 2026;34(1):2. doi: 10.1007/s44446-025-00056-w
- Choudhury AD. PTEN‐PI3K pathway alterations in advanced prostate cancer and clinical implications. Prostate. 2022;82(S1). doi: 10.1002/pros.24372
- Cheng HH, Sokolova AO, Schaeffer EM, Small EJ, Higano CS. Germline and Somatic Mutations in Prostate Cancer for the Clinician. J Natl Compr Canc Netw. 2019;17(5):515-521. doi: 10.6004/jnccn.2019.7307
- Hussain M, Mateo J, Fizazi K, et al. Survival with Olaparib in Metastatic Castration-Resistant Prostate Cancer. N Engl J Med. 2020;383(24):2345-2357. doi: 10.1056/NEJMoa2022485
- Barnes DR, Silvestri V, Leslie G, et al. Breast and prostate cancer risks for male BRCA1 and BRCA2 pathogenic variant carriers using polygenic risk scores. J Natl Cancer Inst. 2022;114(1):109-122. doi: 10.1093/jnci/djab147
- Mateo J, De Bono JS, Fizazi K, et al. Olaparib for the Treatment of Patients With Metastatic Castration-Resistant Prostate Cancer and Alterations in BRCA1 and/or BRCA2 in the PROfound Trial. J Clin Oncol. 2024;42(5):571-583. doi: 10.1200/JCO.23.00339
- Castroviejo-Bermejo M, Cruz C, Llop-Guevara A, et al. A RAD51 assay feasible in routine tumor samples calls PARP inhibitor response beyond BRCA mutation. EMBO Mol Med. 2018;10(12):e9172. doi: 10.15252/emmm.201809172
- Abida W, Armenia J, Gopalan A, et al. Prospective Genomic Profiling of Prostate Cancer Across Disease States Reveals Germline and Somatic Alterations That May Affect Clinical Decision Making. JCO Precis Oncol. 2017;(1):1-16. doi: 10.1200/PO.17.00029
- Guo H, Zhang J, Qin C, et al. Biomarker-Targeted Therapies in Non–Small Cell Lung Cancer: Current Status and Perspectives. Cells. 2022;11(20):3200. doi: 10.3390/cells11203200
- Stopsack KH, Nandakumar S, Wibmer AG, et al. Oncogenic Genomic Alterations, Clinical Phenotypes, and Outcomes in Metastatic Castration-Sensitive Prostate Cancer. Clin Cancer Res. 2020;26(13):3230-3238. doi: 10.1158/1078-0432.CCR-20-0168
- Gong J, Kim DM, Freeman MR, et al. Genetic and biological drivers of prostate cancer disparities in Black men. Nat Rev Urol. 2024;21(5):274-289. doi: 10.1038/s41585-023-00828-w
- Ge T, Gu X, Jia R, et al. Crosstalk between metabolic reprogramming and epigenetics in cancer: updates on mechanisms and therapeutic opportunities. Cancer Commun. 2022;42(11):1049-1082. doi: 10.1002/cac2.12374
- Baboudjian M, Breda A, Rajwa P, et al. Active Surveillance for Intermediate-risk Prostate Cancer: A Systematic Review, Meta-analysis, and Metaregression. Eur Urol Oncol. 2022;5(6):617-627. doi: 10.1016/j.euo.2022.07.004
- Kang DW, Field CJ, Patel D, et al. Effects of high-intensity interval training on cardiometabolic biomarkers in patients with prostate cancer undergoing active surveillance: a randomized controlled trial. Prostate Cancer Prostatic Dis. 2025;28(2):469-474. doi: 10.1038/s41391-024-00867-3
- Sartor O, De Bono J, Chi KN, et al. Lutetium-177–PSMA-617 for Metastatic Castration-Resistant Prostate Cancer. N Engl J Med. 2021;385(12):1091-1103. doi: 10.1056/NEJMoa2107322
- Sathianathen NJ, Konety BR, Crook J, Saad F, Lawrentschuk N. Landmarks in prostate cancer. Nat Rev Urol. 2018;15(10):627-642. doi: 10.1038/s41585-018-0060-7
- Schweizer MT, Sivakumar S, Tukachinsky H, et al. Concordance of DNA Repair Gene Mutations in Paired Primary Prostate Cancer Samples and Metastatic Tissue or Cell-Free DNA. JAMA Oncol. 2021;7(9):1378. doi: 10.1001/jamaoncol.2021.2350
- Fernandez A, Artola M, Leon S, et al. Cancer Vulnerabilities Through Targeting the ATR/Chk1 and ATM/Chk2 Axes in the Context of DNA Damage. Cells. 2025;14(10):748. doi: 10.3390/cells14100748
- Warner E. Genomic and clinical features of metastatic prostate cancer. Published online 2022. doi: 10.14288/1.0421268
- Garcia MEG, Kirsch DG, Reitman ZJ. Targeting the ATM Kinase to Enhance the Efficacy of Radiotherapy and Outcomes for Cancer Patients. Semin Radiat Oncol. 2022;32(1):3-14. doi: 10.1016/j.semradonc.2021.09.008
- Middleton MR, Dean E, Evans TRJ, et al. Phase 1 study of the ATR inhibitor berzosertib (formerly M6620, VX-970) combined with gemcitabine ± cisplatin in patients with advanced solid tumours. Br J Cancer. 2021;125(4):510-519. doi: 10.1038/s41416-021-01405-x
- Saw PE, Song E. Clinical RNA Sequencing: From Molecular Basis to Clinics. In: RNA Therapeutics in Human Diseases. Springer; 2025:687-707. doi: 10.1007/978-981-96-3041-7_29
- Stark R, Grzelak M, Hadfield J. RNA sequencing: the teenage years. Nat Rev Genet. 2019;20(11):631-656. doi: 10.1038/s41576-019-0150-2
- Lakritz S, Nicklawsky A, Alami V, et al. Activity of Platinum Chemotherapy in Men With Prostate Cancer With and Without DNA Damage Repair Mutations. Clin Genitourin Cancer. 2025;23(1):102293. doi: 10.1016/j.clgc.2024.102293
- Taylor SC, Laperriere G, Germain H. Droplet Digital PCR versus qPCR for gene expression analysis with low abundant targets: from variable nonsense to publication quality data. Sci Rep. 2017;7(1):2409. doi: 10.1038/s41598-017-02217-x
- Feng DC, Zhu WZ, Wang J, et al. The implications of single-cell RNA-seq analysis in prostate cancer: unraveling tumor heterogeneity, therapeutic implications and pathways towards personalized therapy. Mil Med Res. 2024;11(1):21. doi: 10.1186/s40779-024-00526-7
- Ashinze P, Unwaba W, Adeyemo B, et al. Precision Medicine in Africa: Current State and Strategies for development. Precis Nanomed. 2025;8(2). doi: 10.33218/001c.137477
- Jalloh M, Cassell A, Niang L, Rebbeck T. Global viewpoints: updates on prostate cancer in SUB‐SAHARAN Africa. BJU Int. 2024;133(1):6-13. doi: 10.1111/bju.16178
- Popejoy AB, Fullerton SM. Genomics is failing on diversity. Nature. 2016;538(7624):161-164. doi: 10.1038/538161a
- Manrai AK, Funke BH, Rehm HL, et al. Genetic Misdiagnoses and the Potential for Health Disparities. N Engl J Med. 2016;375(7):655-665. doi: 10.1056/NEJMsa1507092
- Mulder N, Abimiku A, Adebamowo SN, et al. H3Africa: current perspectives. PGPM. 2018;11:59-66. doi: 10.2147/PGPM.S141546
- Sekhoacha M, Riet K, Motloung P, Gumenku L, Adegoke A, Mashele S. Prostate Cancer Review: Genetics, Diagnosis, Treatment Options, and Alternative Approaches. Molecules. 2022;27(17):5730. doi: 10.3390/molecules27175730
- Darst BF, Shen J, Madduri RK, et al. Evaluating approaches for constructing polygenic risk scores for prostate cancer in men of African and European ancestry. Am J Hum Genet. 2023;110(7):1200-1206. doi: 10.1016/j.ajhg.2023.05.010
- Hoffmann TJ, Graff RE, Madduri RK, et al. Genome-wide association study of prostate-specific antigen levels in 392,522 men identifies new loci and improves prediction across ancestry groups. Nat Genet. 2025;57(2):334-344. doi: 10.1038/s41588-024-02068-z
- Bhambhvani HP, Zamora A, Shkolyar E, et al. Development of robust artificial neural networks for prediction of 5-year survival in bladder cancer. Urol Oncol. 2021;39(3):193.e7-193.e12. doi: 10.1016/j.urolonc.2020.05.009
- Marcus L, Fashoyin-Aje LA, Donoghue M, et al. FDA Approval Summary: Pembrolizumab for the Treatment of Tumor Mutational Burden–High Solid Tumors. Clin Cancer Res. 2021;27(17):4685-4689. doi: 10.1158/1078-0432.CCR-21-0327
- Shore N, Armstrong AJ, Barata P, et al. Implementing and Optimizing Universal Germline Genetic Testing for Patients With Prostate Cancer in Clinical Practice. Urology. 2025;199:1-10. doi: 10.1016/j.urology.2025.01.070
- Onyije FM, Hosseini B, Togawa K, Schuz J, Olsson A. Cancer Incidence and Mortality among Petroleum Industry Workers and Residents Living in Oil Producing Communities: A Systematic Review and Meta-Analysis. Int J Environ Res Public Health. 2021;18(8):4343. doi: 10.3390/ijerph18084343
- Ezejiofor TIN. Morbidity and mortality patterns of Nigerian petroleum oil refining and distribution industry. Mary Slessor J Med. 2010;10(1):10.2.
- Ofuru V, Ekeke N, Obiorah C. Genitourinary malignancies in Port Harcourt, Nigeria. Port Harcourt Med J. 2017;11(1):10. doi: 10.4103/phmj.phmj_9_17
- Papageorgiou GI, Skouteris N, Kosma A, Markouizou A, Charalampakis N. Is the Patient Actually Failing on Enzalutamide? A Case Report and Issues to Consider in Enzalutamide-Resistant Oligoprogressive Metastatic Castrate-Resistant Prostate Cancer. RRCT. 2023;18(2):146-155. doi: 10.2174/1574887118666230412112136
- Sokolova A, Marshall CH, Lozano R, et al. Treatment response comparisons between ATM and BRCA2 germline carriers for mCRPC. J Clin Oncol. 2020;38(6_suppl):63. doi: 10.1200/JCO.2020.38.6_suppl.63
- Jiang B, Bi Y, Chen Y, Bi J, Deng J, Zhang G. Case Report: Two cases of chemotherapy refractory aggressive variant prostate cancer with extreme durable response to PARP inhibitor. Front Oncol. 2025;15:1533627. doi: 10.3389/fonc.2025.1533627
- Steenbock O, Paffenholz P, Rieger C, et al. Next generation sequencing (NGS)-based molecular panel analysis for metastatic prostate cancer: how often can we detect druggable mutations? NGS for metastatic adenocarcinoma of the prostate. Urologie. 2025;64(3):256-268. doi: 10.1007/s00120-024-02493-2
- Dall’Era M, Mitsiades N, Parikh M, et al. Neoadjuvant niraparib in men with DNA repair gene deficient clinically localized prostate cancer: Clinical and molecular results from a phase 2 investigator-initiated trial. J Clin Oncol. 2025;43(16_suppl):5104. doi: 10.1200/JCO.2025.43.16_suppl.5104
- Dillon KM, Bekele RT, Sztupinszki Z, et al. PALB2 or BARD1 loss confers homologous recombination deficiency and PARP inhibitor sensitivity in prostate cancer. npj Precis Oncol. 2022;6(1):49. doi: 10.1038/s41698-022-00291-7
- Ogunniyi TJ, Fatokun BS, Isah KO, Abdulbaki A, Emiola AR, Batisani K. Current Status of Cancer Diagnosis and Treatment in Nigeria. Health Sci Rep. 2025;8(6):e70877. doi: 10.1002/hsr2.70877
- Bajinka O, Touray M, Barrow A, Ouedraogo SY, Jallow L. Cancer education and capacity in sub-Saharan Africa-where do we stand? Health Educ Res. 2025;40(6):cyaf062. doi: 10.1093/her/cyaf062
- Abiye T, Owhotemu OM, Agusomu TD, Raimi MO. Extractive Economies, Invisible Burdens: Reframing Environmental Degradation in the Niger Delta as a Public Health Emergency. Preprint posted online February 26, 2026. doi: 10.20944/preprints202602.1686.v1
- Gilfillan E, Araujo M, Kromberg J, Wessels T, Fourie K. The evolving role of genetic counselors in South Africa: A decade of growth. J Genet Couns. 2025;34(4):e70097. doi: 10.1002/jgc4.70097
- Munung NS, Nnodu OE, Moru PO, et al. Looking ahead: ethical and social challenges of somatic gene therapy for sickle cell disease in Africa. Gene Ther. 2024;31(5-6):202-208. doi: 10.1038/s41434-023-00429-7
- Twesigomwe D, Mazhindu TA, Nagy M, Agesa G, Scholefield J, Masimirembwa C. Pharmacogenomics in Africa: A Potential Catalyst for Precision Medicine in Genetically Diverse Populations. Annu Rev Genomics Hum Genet. 2025;26(1):321-349. doi: 10.1146/annurev-genom-121323-104008

