Exosome immobilization of 3D-printed polycaprolactone scaffolds for bone tissue engineering

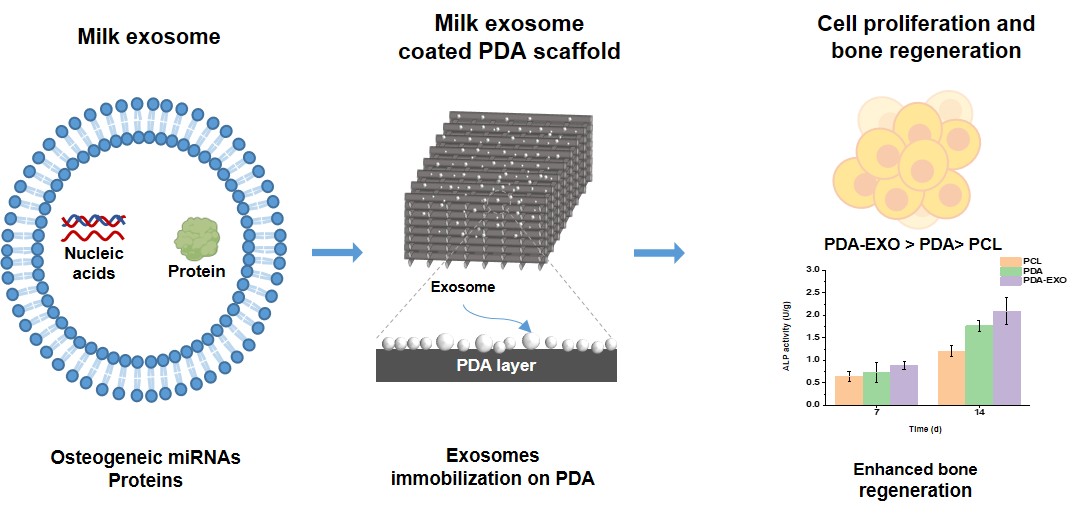
Biodegradable polymers are widely used in bone tissue engineering to repair bone defects by providing biocompatible scaffolds with good mechanical support. Among them, three-dimensional (3D)-printed polycaprolactone (PCL) is commonly used due to its biocompatibility and compressive stability. However, its hydrophobicity and lack of osteogenic cues limit cell attachment and osteogenic differentiation. To address these limitations, 3D-printed PCL scaffolds were coated with polydopamine (PDA) to increase hydrophilicity, and milk-derived exosomes (EXOs) were immobilized on the surface to promote cell proliferation and induce osteogenic differentiation, thereby producing PDA–EXO scaffolds. EXOs represent a cell-free alternative for delivering growth factors and microRNA cargo that provide osteogenic cues for bone regeneration. PDA–EXO scaffolds demonstrated greater cell viability and proliferation compared to PCL and PDA scaffolds due to the synergistic effects of the PDA coating and the EXOs. The PDA–EXO scaffolds also led to better osteogenic differentiation compared to the other scaffolds. Taken together, these findings indicate that PDA enhanced surface hydrophilicity and that milk derived EXOs provided osteo inductive signals, thereby synergistically increasing cell proliferation and osteogenic differentiation while maintaining the scaffold’s mechanical properties. PDA–EXO functionalization, therefore, represents a practical, cell-free strategy to enhance PCL scaffolds for bone tissue engineering.
- Roszkowski S. Therapeutic potential of mesenchymal stem cell-derived exosomes for regenerative medicine applications. Clin Exp Med. 2024;24(1):46. doi: 10.1007/s10238-023-01282-z
- Oliveira MC, Arntz OJ, Blaney Davidson EN, et al. Milk extracellular vesicles accelerate osteoblastogenesis but impair bone matrix formation. J Nutr Biochem. 2016;30:74-84. doi: 10.1016/j.jnutbio.2015.11.017
- Jang H, Kim H, Kim EH, et al. Post-insertion technique to introduce targeting moieties in milk exosomes for targeted drug delivery. Biomater Res. 2023;27(1):124. doi: 10.1186/s40824-023-00456-w
- Go G, Jeon J, Lee G, Lee JH, Lee SH. Bovine milk extracellular vesicles induce the proliferation and differentiation of osteoblasts and promote osteogenesis in rats. J Food Biochem. 2021;45(4):e13705. doi: 10.1111/jfbc.13705
- Zhao P, Xiao L, Peng J, Qian YQ, Huang CC. Exosomes derived from bone marrow mesenchymal stem cells improve osteoporosis through promoting osteoblast proliferation via MAPK pathway. Eur Rev Med Pharmacol Sci. 2018;22(12):3962-3970. doi: 10.26355/eurrev_201806_15280
- Preethi Soundarya S, Haritha Menon A, Viji Chandran S, Selvamurugan N. Bone tissue engineering: Scaffold preparation using chitosan and other biomaterials with different design and fabrication techniques. Int J Biol Macromol. 2018;119:1228-1239. doi: 10.1016/j.ijbiomac.2018.08.056
- Chia HN, Wu BM. Recent advances in 3D printing of biomaterials. J Biol Eng. 2015;9(1):4. doi: 10.1186/s13036-015-0001-4
- Karanth D, Song K, Martin ML, et al. Towards resorbable 3D-printed scaffolds for craniofacial bone regeneration. Orthod Craniofac Res. 2023;26(suppl 1):188-195. doi: 10.1111/ocr.12645
- Lee SS, Du X, Kim I, Ferguson SJ. Scaffolds for bone-tissue engineering. Matter. 2022;5(9):2722-2759. doi: 10.1016/j.matt.2022.06.003
- Li L, Li J, Guo J, et al. 3D molecularly functionalized cell-free biomimetic scaffolds for osteochondral regeneration. Adv Funct Mater. 2019;29(6):1807356. doi: 10.1002/adfm.201807356
- Deng L, Liu Y, Wu Q, et al. Exosomes to exosome functionalized scaffolds: a novel approach to stimulate bone regeneration. Stem Cell Res Ther. 2024;15(1):407. doi: 10.1186/s13287-024-04024-4
- Xu H, Chai Q, Xu X, et al. Exosome-functionalized Ti6Al4V scaffolds promoting osseointegration by modulating endogenous osteogenesis and osteoimmunity. ACS Appl Mater Interfaces. 2022;14(41):46161-46175. doi: 10.1021/acsami.2c11102
- Shenoda BB, Ajit SK. Modulation of immune responses by exosomes derived from antigen-presenting cells. Clin Med Insights Pathol. 2016;9(suppl 1):1-8. doi: 10.4137/CPath.S39925
- Hajiali F, Tajbakhsh S, Shojaei A. Fabrication and properties of polycaprolactone composites containing calcium phosphate-based ceramics and bioactive glasses in bone tissue engineering: A Review. Polym Rev. 2018;58(1):164-207. doi: 10.1080/15583724.2017.1332640
- Ebrahimi Z, Irani S, Ardeshirylajimi A, Seyedjafari E. Enhanced osteogenic differentiation of stem cells by 3D printed PCL scaffolds coated with collagen and hydroxyapatite. Sci Rep. 2022;12(1):12359. doi: 10.1038/s41598-022-15602-y
- Tolabi H, Bakhtiary N, Sayadi S, et al. A critical review on polydopamine surface-modified scaffolds in musculoskeletal regeneration. Front Bioeng Biotechnol. 2022;10:1008360. doi: 10.3389/fbioe.2022.1008360
- Tsai WB, Chen WT, Chien HW, Kuo WH, Wang MJ. Poly(dopamine) coating of scaffolds for articular cartilage tissue engineering. Acta Biomater. 2011;7(12):4187-4194. doi: 10.1016/j.actbio.2011.07.024
- Lee DJ, Tseng HC, Wong SW, Wang Z, Deng M, Ko CC. Dopaminergic effects on in vitro osteogenesis. Bone Res. 2015;3(1):15020. doi: 10.1038/boneres.2015.20
- Wang J, Cui Y, Zhang B, et al. Polydopamine-modified functional materials promote bone regeneration. Mater Des. 2024;238:112655. doi: 10.1016/j.matdes.2024.112655
- Zhang P, He M, Zeng Y. Ultrasensitive microfluidic analysis of circulating exosomes using a nanostructured graphene oxide/polydopamine coating. Lab Chip. 2016;16(16):3033-3042. doi: 10.1039/C6LC00279J
- Li W, Liu Y, Zhang P, et al. Tissue-engineered bone immobilized with human adipose stem cells-derived exosomes promotes bone regeneration. ACS Appl Mater Interfaces. 2018;10(6):5240-5254. doi: 10.1021/acsami.7b17620
- Gao Y, Yuan Z, Yuan X, et al. Bioinspired porous microspheres for sustained hypoxic exosomes release and vascularized bone regeneration. Bioact Mater. 2022;14:377-388. doi: 10.1016/j.bioactmat.2022.01.041
- Ahn G, Kim YH, Ahn JY. Multifaceted effects of milk exosomes (Mi-Exo) as a modulator of scar-free wound healing. Nanoscale Adv. 2021;3(2):528-537. doi: 10.1039/D0NA00665C
- Kalluri R, LeBleu VS. The biology, function, and biomedical applications of exosomes. Science. 2020;367(6478):eaau6977. doi: 10.1126/science.aau6977
- Kalra H, Adda CG, Liem M, et al. Comparative proteomics evaluation of plasma exosome isolation techniques and assessment of the stability of exosomes in normal human blood plasma. Proteomics. 2013;13(22):3354-3364. doi: 10.1002/pmic.201300282
- Deng F, Miller J. A review on protein markers of exosome from different bio-resources and the antibodies used for characterization. J Histotechnol. 2019;42(4):226-239. doi: 10.1080/01478885.2019.1646984
- Sims NA. The JAK1/STAT3/SOCS3 axis in bone development, physiology, and pathology. Exp Mol Med. 2020;52(8):1185-1197. doi: 10.1038/s12276-020-0445-6
- Asagiri M, Takayanagi H. The molecular understanding of osteoclast differentiation. Bone. 2007;40(2):251-264. doi: 10.1016/j.bone.2006.09.023
- Kitaura H, Kimura K, Ishida M, Kohara H, Yoshimatsu M, Takano-Yamamoto T. Immunological reaction in TNF-α- mediated osteoclast formation and bone resorption in vitro and in vivo. Clin Dev Immunol. 2013;2013:181849. doi: 10.1155/2013/181849
- Wang L, You X, Zhang L, Zhang C, Zou W. Mechanical regulation of bone remodeling. Bone Res. 2022;10(1):16. doi: 10.1038/s41413-022-00190-4
- Mulari MTK, Qu Q, Harkonen PL, Vaananen HK. Osteoblast-like cells complete osteoclastic bone resorption and form new mineralized bone matrix in vitro. Calcif Tissue Int. 2004;75(3):253-261. doi: 10.1007/s00223-004-0172-3
- Zhou Q, Li M, Wang X, et al. Immune-related microRNAs are abundant in breast milk exosomes. Int J Biol Sci. 2012;8(1):118-123. doi: 10.7150/ijbs.8.118
- Huda MN, Nafiujjaman M, Deaguero IG, et al. Potential use of exosomes as diagnostic biomarkers and in targeted drug delivery: progress in clinical and preclinical applications. ACS Biomater Sci Eng. 2021;7(6):2106-2149. doi: 10.1021/acsbiomaterials.1c00217
- Busso-Lopes AF, Carnielli CM, Winck FV, et al. A reductionist approach using primary and metastatic cell-derived extracellular vesicles reveals hub proteins associated with oral cancer prognosis. Mol Cell Proteomics. 2021;20:100118. doi: 10.1016/j.mcpro.2021.100118
- Deliormanlı AM, Atmaca H. Effect of pore architecture on the mesenchymal stem cell responses to graphene/polycaprolactone scaffolds prepared by solvent casting and robocasting. J Porous Mater. 2020;27(1):49-61. doi: 10.1007/s10934-019-00791-1
- Lee H, Han G, Na Y, et al. 3D-Printed Tissue-Specific Nanospike-Based Adhesive Materials for Time-Regulated Synergistic Tumor Therapy and Tissue Regeneration In Vivo. Adv Funct Mater. 2024;34(48):2406237. doi: 10.1002/adfm.202406237
- Bružauskaitė I, Bironaitė D, Bagdonas E, Bernotienė E. Scaffolds and cells for tissue regeneration: different scaffold pore sizes—different cell effects. Cytotechnology. 2016;68(3):355-369. doi: 10.1007/s10616-015-9895-4
- Peng L, Pan Y, Liu Q, et al. Hierarchically functionalized PCL/CS with synergistic PDA-mediated antioxidant therapy and NGF-activated neurogenesis for spinal cord injury regeneration. J Biomater Sci Polym Ed. Published online August 3, 2025. doi: 10.1080/09205063.2025.2542479
- Shariati A, Ebrahimi T, Babadinia P, Shariati FS, Ahangari Cohan R. Synthesis and characterization of Gd3+-loaded hyaluronic acid-polydopamine nanoparticles as a dual contrast agent for CT and MRI scans. Sci Rep. 2023;13(1):4520. doi: 10.1038/s41598-023-31252-0
- Baddela VS, Nayan V, Rani P, Onteru SK, Singh D. Physicochemical biomolecular insights into buffalo milk-derived nanovesicles. Appl Biochem Biotechnol. 2016;178(3):544-557. doi: 10.1007/s12010-015-1893-7
- Cho SJ, Jung SM, Kang M, Shin HS, Youk JH. Preparation of hydrophilic PCL nanofiber scaffolds via electrospinning of PCL/PVP-b-PCL block copolymers for enhanced cell biocompatibility. Polymer. 2015;69:95-102. doi: 10.1016/j.polymer.2015.05.037
- Rim NG, Kim SJ, Shin YM, et al. Mussel-inspired surface modification of poly(l-lactide) electrospun fibers for modulation of osteogenic differentiation of human mesenchymal stem cells. Colloids Surf B Biointerfaces. 2012;91:189-197. doi: 10.1016/j.colsurfb.2011.10.057
- Wu Y, Zhang Y, Zhang R, Chen S. Preparation and properties of antibacterial polydopamine and nano-hydroxyapatite modified polyethylene terephthalate artificial ligament. Front Bioeng Biotechnol. 2021;9:630745. doi: 10.3389/fbioe.2021.630745
- Kyung Kim D, Lee S, Kim M, Jeong Y, Lee S. Exosome coated silk fibroin 3D-scaffold for inducing osteogenic differentiation of bone marrow derived mesenchymal stem cells. Chem Eng J. 2021;406:127080. doi: 10.1016/j.cej.2020.127080
- Serrano MC, Pagani R, Vallet-Regi M, et al. In vitro biocompatibility assessment of poly(ε-caprolactone) films using L929 mouse fibroblasts. Biomaterials. 2004;25(25):5603-5611. doi: 10.1016/j.biomaterials.2004.01.037
- Cai S, Wu C, Yang W, Liang W, Yu H, Liu L. Recent advance in surface modification for regulating cell adhesion and behaviors. Nanotechnol Rev. 2020;9(1):971-989. doi: 10.1515/ntrev-2020-0076
- Liang W, Han B, Hai Y, Sun D, Yin P. Mechanism of action of mesenchymal stem cell-derived exosomes in the intervertebral disc degeneration treatment and bone repair and regeneration. Front Cell Dev Biol. 2022;9:833840. doi: 10.3389/fcell.2021.833840
- Cannalire G, Pilloni S, Esposito S, Biasucci G, Di Franco A, Street ME. Alkaline phosphatase in clinical practice in childhood: Focus on rickets. Front Endocrinol. 2023;14:1111445. doi: 10.3389/fendo.2023.1111445
- Meesuk L, Suwanprateeb J, Thammarakcharoen F, et al. Osteogenic differentiation and proliferation potentials of human bone marrow and umbilical cord-derived mesenchymal stem cells on the 3D-printed hydroxyapatite scaffolds. Sci Rep. 2022;12(1):19509. doi: 10.1038/s41598-022-24160-2
- Zhang M, Li Y, Feng T, et al. Bone Engineering Scaffolds with Exosomes: A Promising Strategy for Bone Defects Repair. Front Bioeng Biotechnol. 2022;10:920378. doi: 10.3389/fbioe.2022.920378
- Hany E, Yahia S, Elsherbeny MF, et al. Evaluation of the osteogenic potential of rat adipose-derived stem cells with different polycaprolactone/alginate-based nanofibrous scaffolds: an in vitro study. Stem Cell Investig. 2020;7:14. doi: 10.21037/sci-2020-019

