Bioprinting and in vitro characterization of alginate–gelatin constructs incorporating human umbilical vein endothelial cells for potential cardiac tissue engineering

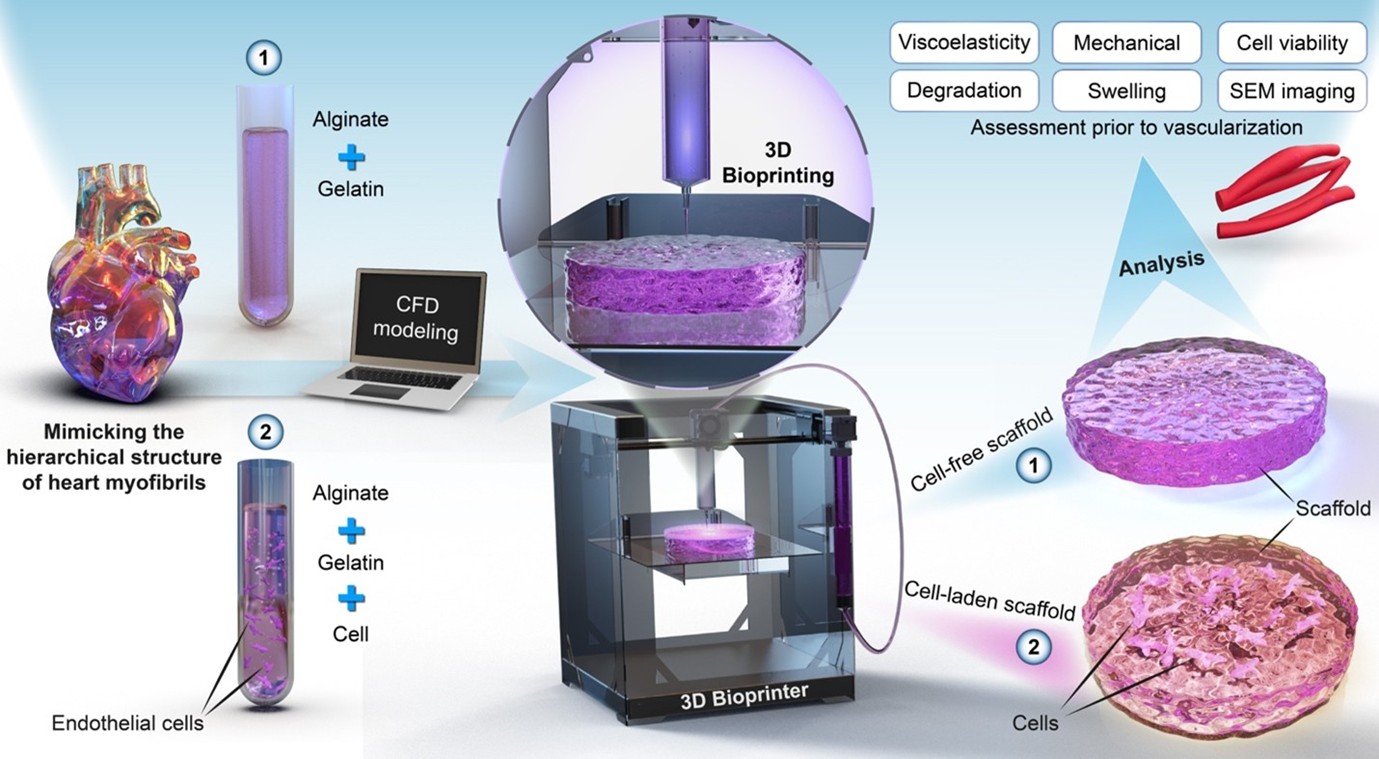
Three-dimensional (3D) bioprinting offers transformative potential for cardiac tissue engineering by enabling the fabrication of cell-laden constructs. However, key challenges remain, including maintaining cell viability within bioprinted constructs and understanding how embedded cells affect their physical and mechanical properties. This study addresses these challenges by incorporating human umbilical vein endothelial cells (HUVECs) into alginate–gelatin hydrogels and evaluating their impact on mechanical, physical, and rheological properties. Bioinks or hydrogels were prepared with or without HUVECs, and their rheological properties were assessed. Computational fluid dynamics (CFD) simulation was employed to determine the appropriate bioprinting pressure while minimizing cell damage. Constructs were designed and 3D-printed with an angular pattern to replicate the orientation of cardiac myofibrils and were characterized over a 21-day period for viscoelasticity, elastic modulus, swelling, mass loss, morphology, and cell viability. The incorporation of cells increased the storage and loss moduli of the bioink, demonstrating shear-thinning behavior as described by the Cross model. CFD simulation combined with preliminary cell viability assays identified 25 kPa as a suitable 3D-printing pressure, effectively preserving cell viability. Both cell-free and cell-laden constructs exhibited viscoelastic properties; however, cell-laden constructs displayed a lower elastic modulus under linear compression, reduced swelling, and greater mass retention. High cell viability was observed immediately post-bioprinting and was maintained for more than 1 week. These findings provide a framework for developing structurally robust, cell-laden constructs with enhanced functional fidelity, supporting their application in cardiac tissue engineering.
- Cho S, Discher DE, Leong KW, Vunjak-Novakovic G, Wu JC. Challenges and opportunities for the next generation of cardiovascular tissue engineering. Nat Methods. 2022;19(9):1064-1071. doi: 10.1038/s41592-022-01591-3
- Garbern JC, Lee RT. Heart regeneration: 20 years of progress and renewed optimism. Dev Cell. 2022;57(4):424-439. doi: 10.1016/j.devcel.2022.01.012
- Izadifar M, Chapman D, Babyn P, Chen X, Kelly ME. UV-assisted 3D-bioprinting of nanoreinforced hybrid cardiac patch for myocardial tissue engineering. Tissue Eng Part C Methods. 2018;24(2):74-88. doi: 10.1089/ten.tec.2017.0346
- Sobanski PZ, Alt-Epping B, Currow DC, et al. Palliative care for people living with heart failure: European Association for Palliative Care Task Force expert position statement. Cardiovasc Res. 2020;116(1):12-27. doi: 10.1093/cvr/cvz200
- Truby LK, Rogers JG. Advanced heart failure: epidemiology, diagnosis, and therapeutic approaches. Heart Fail. 2020;8(7):523-536. doi: 10.1016/j.jchf.2020.01.014
- Tenreiro MF, Louro AF, Alves PM, Serra M. Next generation of heart regenerative therapies: progress and promise of cardiac tissue engineering. NPJ Regen Med. 2021;6(1):30. doi: 10.1038/s41536-021-00140-4
- Wang Z, Wang L, Li T, et al. 3D-bioprinting in cardiac tissue engineering. Theranostics. 2021;11(16):7948. doi: 10.7150/thno.61621
- Liu N, Ye X, Yao B, et al. Advances in 3D-bioprinting technology for cardiac tissue engineering and regeneration. Bioact Mater. 2021;6(5):1388-1401. doi: 10.1016/j.bioactmat.2020.10.021
- Agarwal T, Fortunato GM, Hann SY, et al. Recent advances in bioprinting technologies for engineering cardiac tissue. Mater Sci Eng C. 2021;124:112057. doi: 10.1016/j.msec.2021.112057
- Chang YC, Mirhaidari G, Kelly J, Breuer C. Current challenges and solutions to tissue engineering of large-scale cardiac constructs. Curr Cardiol Rep. 2021;5(47):1-6. doi: 10.1007/s11886-021-01474-7
- Ning L, Betancourt N, Schreyer DJ, Chen X. Characterization of cell damage and proliferative ability during and after bioprinting. ACS Biomater Sci Eng. 2018;4(11):3906-3918. doi: 10.1021/acsbiomaterials.8b00714
- Noroozi R, Arif ZU, Taghvaei H, et al. 3D and 4D bioprinting technologies: a game changer for the biomedical sector? Ann Biomed Eng. 2023;51(8):1683-1712. doi: 10.1007/s10439-023-03243-9
- Tiruvannamalai-Annamalai R, Armant DR, Matthew HWT. A glycosaminoglycan based, modular tissue scaffold system for rapid assembly of perfusable, high cell density, engineered tissues. PLoS One. 2014;9(1):e84287. doi: 10.1371/journal.pone.0084287
- Ketabat F, Alcorn J, Kelly ME, Badea I, Chen X. Cardiac tissue engineering: a journey from scaffold fabrication to in vitro characterization. Small Sci. 2024;4(9):2400079. doi: 10.1002/smsc.202400079
- Chen XB, Anvari-Yazdi AF, Duan X, et al. Biomaterials/ bioinks and extrusion bioprinting. Bioact Mater. 2023;28:511-536. doi: 10.1016/j.bioactmat.2023.06.006
- Ning L, Chen X. A brief review of extrusion‐based tissue scaffold bio‐printing. Biotechnol J. 2017;12(8):1600671. doi: 10.1002/biot.201600671
- Lemarié L, Anandan A, Petiot E, Marquette C, Courtial EJ. Rheology, simulation and data analysis toward bioprinting cell viability awareness. Bioprinting. 2021;21:e00119. doi: 10.1016/j.bprint.2020.e00119
- Krishnamoorthy S, Noorani B, Xu C. Effects of encapsulated cells on the physical–mechanical properties and microstructure of gelatin methacrylate hydrogels. Int J Mol Sci. 2019;20(20):5061. doi: 10.3390/ijms20205061
- Otto IA, Levato R, Webb WR, et al. Progenitor cells in auricular cartilage demonstrate cartilage-forming capacity in 3D hydrogel culture. Eur Cell Mater. 2018;35:132. doi: 10.22203/eCM.v035a10
- Ketabat F, Maris T, Duan X, et al. Optimization of 3D-printing and in vitro characterization of alginate/gelatin lattice and angular scaffolds for potential cardiac tissue engineering. Front Bioeng Biotechnol. 2023;11:1161804. doi: 10.3389/fbioe.2023.1161804
- Maiullari F, Costantini M, Milan M, et al. A multi-cellular 3D-bioprinting approach for vascularized heart tissue engineering based on HUVECs and iPSC-derived cardiomyocytes. Sci Rep. 2018;8(1):13532. doi: 10.1038/s41598-018-31848-x
- Sánchez-Sánchez R, Rodríguez-Rego JM, Macías-García A, Mendoza-Cerezo L, Díaz-Parralejo A. Relationship between shear-thinning rheological properties of bioinks and bioprinting parameters. Int J Bioprint. 2023;9(2):687. doi: 10.18063/ijb.687
- Frank MW. Viscous Fluid Flow. International Edition, 2nd ed. New York, NY: McGraw-Hill; 1991.
- Vey E, Rodger C, Booth J, Claybourn M, Miller AF, Saiani A. Degradation kinetics of poly (lactic-co-glycolic) acid block copolymer cast films in phosphate buffer solution as revealed by infrared and Raman spectroscopies. Polym Degrad Stab. 2011;96(10):1882-1889. doi: 10.1016/j.polymdegradstab.2011.07.011
- Cross MM. Rheology of non-Newtonian fluids: a new flow equation for pseudoplastic systems. J Colloid Sci. 1965;20(5):417-437. doi: 10.1016/0095-8522(65)90022-X
- Chen DXB. Extrusion Bioprinting of Scaffolds for Tissue Engineering. 2nd ed. Cham, Switzerland: Springer International Publishing AG; 2025. doi: 10.1007/978-3-031-72471-8
- Bercea M. Rheology as a tool for fine-tuning the properties of printable bioinspired gels. Molecules. 2023;28(6):2766. doi: 10.3390/molecules28062766
- Bom S, Ribeiro R, Ribeiro HM, Santos C, Marto J. On the progress of hydrogel-based 3D-printing: Correlating rheological properties with printing behaviour. Int J Pharm. 2022;615:121506. doi: 10.1016/j.ijpharm.2022.121506
- Habib, M. A., Khoda, B. Rheological analysis of bio-ink for 3D-bio-printing processes. J Manuf Process. 2022;76:708–718. doi: 10.1016/j.jmapro.2022.02.048
- Chopin-Doroteo M, Mandujano-Tinoco EA, Krötzsch E. Tailoring of the rheological properties of bioinks to improve bioprinting and bioassembly for tissue replacement. Biochim Biophys Acta Gen Subj. 2021;1865(2):129782. doi: 10.1016/j.bbagen.2020.129782
- Ning L, Gil CJ, Hwang B, et al. Biomechanical factors in three-dimensional tissue bioprinting. Appl Phys Rev. 2020;7(4):041319. doi: 10.1063/5.0023206
- Boularaoui S, Al Hussein G, Khan KA, Christoforou N, Stefanini C. An overview of extrusion-based bioprinting with a focus on induced shear stress and its effect on cell viability. Bioprinting. 2020;20:e00093. doi: 10.1016/j.bprint.2020.e00093
- Persaud A, Maus A, Strait L, Zhu D. 3D-bioprinting with live cells. Eng Regen. 2022;3(3):292-309. doi: 10.1016/j.engreg.2022.07.002
- Diamantides N, Dugopolski C, Blahut E, Kennedy S, Bonassar LJ. High density cell seeding affects the rheology and printability of collagen bioinks. Biofabrication. 2019;11(4):045016. doi: 10.1088/1758-5090/ab3524
- Majumder N, Mishra A, Ghosh S. Effect of varying cell densities on the rheological properties of the bioink. Bioprinting. 2022;28:e00241. doi: 10.1016/j.bprint.2022.e00241
- Ouyang L, Yao R, Zhao Y, Sun W. Effect of bioink properties on printability and cell viability for 3D bioplotting of embryonic stem cells. Biofabrication. 2016; 8(3):035020. doi: 10.1088/1758-5090/8/3/035020
- Chen DXB. Preparation of Scaffold solutions and characterization of their flow behavior. In: Extrusion Bioprinting of Scaffolds for Tissue Engineering Applications. Cham, Switzerland: Springer; 2019:91-115. doi: 10.1007/978-3-030-03460-3_5
- Kimbell G, Azad MA. Chapter fifteen — 3D-printing: bioinspired materials for drug delivery. In: Nurunnabi M, ed. Bioinspired and Biomimetic Materials for Drug Delivery. Woodhead Publishing Series in Biomaterials. Cambridge, UK: Woodhead Publishing; 2021:295-318. doi: 10.1016/B978-0-12-821352-0.00011-3
- Gao T, Gillispie GJ, Copus JS, et al. Optimization of gelatin– alginate composite bioink printability using rheological parameters: a systematic approach. Biofabrication. 2018;10(3):034106. doi: 10.1088/1758-5090/aacdc7
- Patra S, Ajayan PM, Narayanan TN. Dynamic mechanical analysis in materials science: the Novice’s Tale. Oxf Open Mater Sci. 2021;1(1):itaa001. doi: 10.1093/oxfmat/itaa001
- Caporizzo MA, Prosser BL. Need for speed: the importance of physiological strain rates in determining myocardial stiffness. Front Physiol. 2021;12:696694. doi: 10.3389/fphys.2021.696694
- Tikenoğulları OZ, Costabal FS, Yao J, Marsden A, Kuhl E. How viscous is the beating heart? Insights from a computational study. Comput Mech. 2022;70(3):565-579. doi: 10.1007/s00466-022-02180-z
- Wang Z, St-Onge MP, Lecumberri B, et al. Body cell mass: model development and validation at the cellular level of body composition. Am J Physiol Endocrinol Metab. 2004;286(1):E123-E128. doi: 10.1152/ajpendo.00227.2003
- Hasan A, Ragaert K, Swieszkowski W, et al. Biomechanical properties of native and tissue engineered heart valve constructs. J Biomech. 2014;47(9):1949-1963. doi: 10.1016/j.jbiomech.2013.09.023
- Levett PA, Hutmacher DW, Malda J, Klein TJ. Hyaluronic acid enhances the mechanical properties of tissue-engineered cartilage constructs. PLoS One. 2014; 9(12):e113216. doi: 10.1371/journal.pone.0113216
- Ghazanfari S, Alberti KA, Xu Q, Khademhosseini A. Evaluation of an elastic decellularized tendon‐derived scaffold for the vascular tissue engineering application. J Biomed Mater Res A. 2019;107(6):1225-1234. doi: 10.1002/jbm.a.36622
- Janmey, P. A., Miller, R. T. Mechanisms of mechanical signaling in development and disease. J Cell Sci. 2011;124(1):9-18. doi: 10.1242/jcs.071001
- Engler AJ, Carag-Krieger C, Johnson CP, et al. Embryonic cardiomyocytes beat best on a matrix with heart-like elasticity: scar-like rigidity inhibits beating. J Cell Sci. 2008;121(22):3794-3802. doi: 10.1242/jcs.029678
- Moreno-Castellanos N, Velásquez-Rincón MC, Rodríguez- Sanabria AV, Cuartas-Gómez E, Vargas-Ceballos O. Encapsulation of beta-pancreatic cells in a hydrogel based on alginate and graphene oxide with high potential application in the diabetes treatment. J Mater Res. 2023;38(10):2823-2837. doi: 10.1557/s43578-023-01009-6
- Mohammadrezaei D, Moghimi N, Vandvajdi S, Powathil G, Hamis S, Kohandel M. Predicting and elucidating the post-printing behavior of 3D-printed cancer cells in hydrogel structures by integrating in-vitro and in-silico experiments. Sci Rep. 2023;13(1):1211. doi: 10.1038/s41598-023-28286-9

