Development and assessment of dimethyl sulfoxide-free antifreeze gelatin methacryloyl hydrogels for integrated three-dimensional bioprinting and cryopreservation

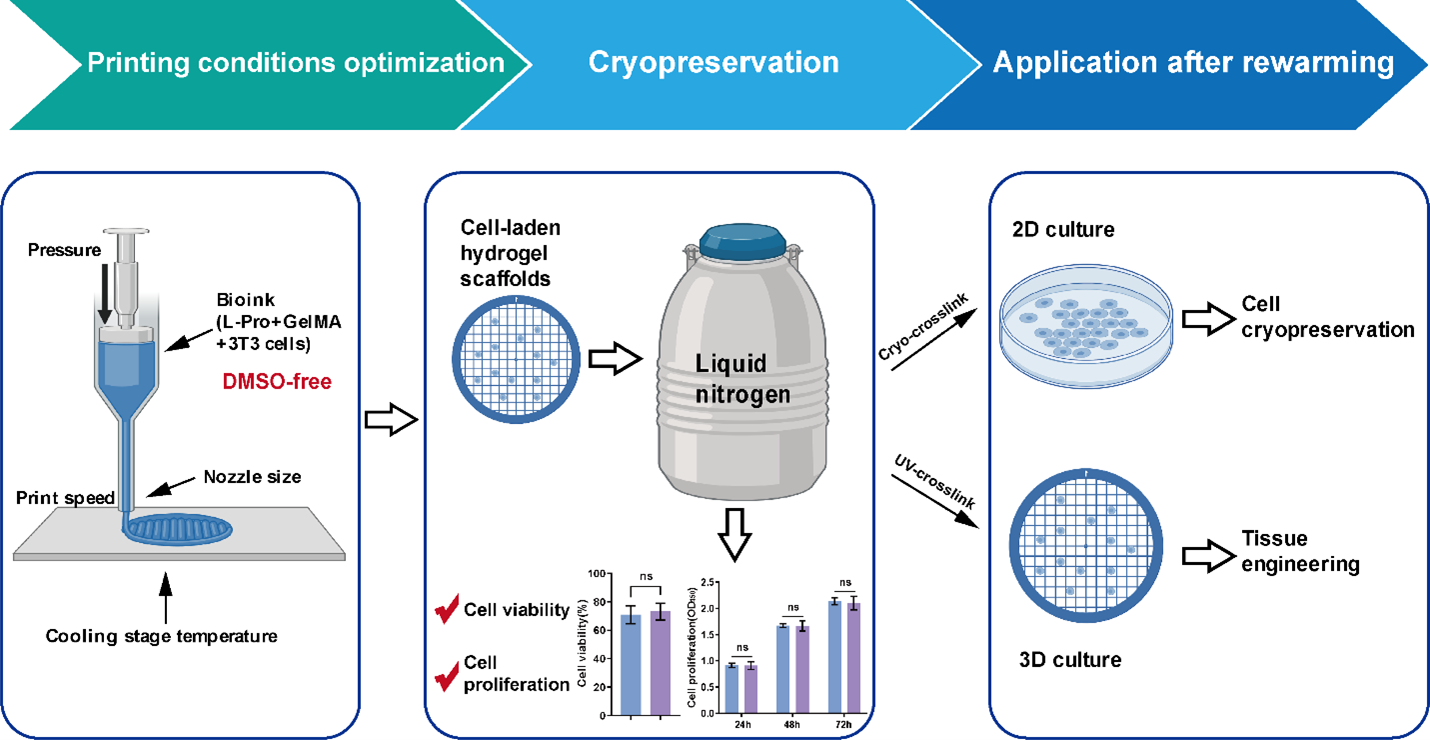
Three-dimensional (3D) bioprinting enables the fabrication of engineered tissues, but cell damage during printing and limitations in long-term preservation hinder practical applications. Traditional cryoprotectants, such as dimethyl sulfoxide (DMSO), introduce cytotoxicity and require complex removal, restricting immediate tissue usability. Here, we present an integrated extrusion-based bioprinting and DMSO-free antifreeze hydrogel strategy to produce cell-laden constructs with high post-thaw viability and proliferative capacity. Systematic optimization of bioink composition (6% L-proline with varying gelatin methacryloyl concentrations), extrusion parameters, and crosslinking conditions enabled high-fidelity scaffold fabrication while preserving cell viability and proliferation. Numerical simulations guided the maximum printable heights for fibers of different diameters, supporting construct scalability. Storing cell-laden 3D-printed scaffolds in cryovials at −80 °C effectively maintained high cell viability compared with alternative cooling protocols. Cells in 3D scaffolds exhibited superior post-thaw proliferation compared with two-dimensional culture, and the platform was validated using C2C12 myoblasts, achieving high survival and robust recovery of proliferative capacity. This study establishes a practical and versatile framework for integrating bioprinting and cryopreservation to support the generation of cell-laden constructs with preserved viability and structural integrity for regenerative medicine applications.
- Liu K, Hu N, Yu Z, et al. 3D printing and bioprinting in urology. Int J Bioprint. 2023;9(6):0969. doi: 10.36922/ijb.0969
- Pantermehl S, Emmert S, Foth A, et al. 3D Printing for Soft Tissue Regeneration and Applications in Medicine. Biomedicines. 2021;9(4):336. doi: 10.3390/biomedicines9040336
- Wang H, Yang Y, Zhou X, et al. Rational design of mechanical bio-metamaterials for biomedical applications. Prog Mater Sci. 2026;156:101545. doi: 10.1016/j.pmatsci.2025.101545
- Habib A, Sathish V, Mallik S, et al. 3D Printability of Alginate-Carboxymethyl Cellulose Hydrogel. Materials. 2018;11(3):454. doi: 10.3390/ma11030454
- Emmermacher J, Spura D, Cziommer J, et al. Engineering considerations on extrusion-based bioprinting: interactions of material behavior, mechanical forces and cells in the printing needle. Biofabrication. 2020;12(2):025022. doi: 10.1088/1758-5090/ab7553
- Ning L, Guillemot A, Zhao J, et al. Influence of Flow Behavior of Alginate-Cell Suspensions on Cell Viability and Proliferation. Tissue Eng Part C Methods. 2016;22(7):652- 662. doi: 10.1089/ten.TEC.2016.0011
- Wu W, Xia R, Qian G, et al. Mechanostructures: Rational mechanical design, fabrication, performance evaluation, and industrial application of advanced structures. Prog Mater Sci. 2023;131:101021. doi: 10.1016/j.pmatsci.2022.101021
- Zhang YS, Haghiashtiani G, Hübscher T, et al. 3D extrusion bioprinting. Nat Rev Methods Primers. 2021;1(1):75. doi: 10.1038/s43586-021-00073-8
- Ng WL, Shkolnikov V. Optimizing cell deposition for inkjet-based bioprinting. Int J Bioprint. 2024;10(2):2135. doi: 10.36922/ijb.2135
- Law ACC, Wang R, Chung J, et al. Process parameter optimization for reproducible fabrication of layer porosity quality of 3D-printed tissue scaffold. J Intell Manuf. 2024;35(4):1825-1844. doi: 10.1007/s10845-023-02141-0
- Das S, Valoor R, Ratnayake P, et al. Low-concentration gelatin methacryloyl hydrogel with tunable 3D extrusion printability and cytocompatibility: exploring quantitative process science and biophysical properties. ACS Appl Bio Mater. 2024;7(5):2809-2835. doi: 10.1021/acsabm.3c01194
- Ji S, Guvendiren M. Recent Advances in Bioink Design for 3D Bioprinting of Tissues and Organs. Front Bioeng Biotechnol. 2017;5:23. doi: 10.3389/fbioe.2017.00023
- Malda J, Visser J, Melchels FP, et al. 25th anniversary article: Engineering hydrogels for biofabrication. Adv Mater. 2013;25(36):5011-5028. doi: 10.1002/adma.201302042
- Chang R, Nam J, Sun W. Effects of dispensing pressure and nozzle diameter on cell survival from solid freeform fabrication-based direct cell writing. Tissue Eng Part A. 2008;14(1):41-48. doi: 10.1089/ten.a.2007.0004
- Correia FP, Monteiro MV, Borralho M, et al. Advanced Toolboxes for Cryobioprinting Human Tissue Analogs. Adv Healthc Mater. 2025;14(10):2405011. doi: 10.1002/adhm.202405011
- Ziani K, Saenz-del-Burgo L, Pedraz J L, et al. Advances in Cryopreservation Strategies for 3D Biofabricated Constructs: From Hydrogels to Bioprinted Tissues. Int J Mol Sci. 2025;26(14):6908. doi: 10.3390/ijms26146908
- Ravanbakhsh H, Luo Z, Zhang X, et al. Freeform Cell-Laden Cryobioprinting for Shelf-Ready Tissue Fabrication and Storage. Matter. 2022;5(2):573-593. doi: 10.1016/j.matt.2021.11.020
- Liu M, Jiang S, Witman N, et al. Intrinsically cryopreservable, bacteriostatic, durable glycerohydrogel inks for 3D bioprinting. Matter. 2023;6(3):983-999. doi: 10.1016/j.matt.2022.12.013
- Liu M, Zhang X, Guo H, et al. Dimethyl Sulfoxide- Free Cryopreservation of Chondrocytes Based on Zwitterionic Molecule and Polymers. Biomacromolecules. 2019;20(10):3980-3988. doi: 10.1021/acs.biomac.9b01024
- Mahdavi SS, Abdekhodaie MJ, Kumar H, et al. Stereolithography 3D Bioprinting Method for Fabrication of Human Corneal Stroma Equivalent. Ann Biomed Eng. 2020;48(7):1955-1970. doi: 10.1007/s10439-020-02537-6
- Cao K, Shen L, Guo X, et al. Hydrogel Microfiber Encapsulation Enhances Cryopreservation of Human Red Blood Cells with Low Concentrations of Glycerol. Biopreserv Biobank. 2020;18(3):228-234. doi: 10.1089/bio.2020.0003
- Munesada D, Sakai D, Nakamura Y, et al. Investigation of the mitigation of DMSO-induced cytotoxicity by hyaluronic acid following cryopreservation of human nucleus pulposus cells. Int J Mol Sci. 2023;24(15):12289. doi: 10.3390/ijms241512289
- Pogozhykh D, Eicke D, Gryshkov O, et al. Towards reduction or substitution of cytotoxic dmso in biobanking of functional bioengineered megakaryocytes. Int J Mol Sci. 2020;21(20):7654. doi: 10.3390/ijms21207654
- Elliott GD, Wang S, Fuller BJ. Cryoprotectants: A review of the actions and applications of cryoprotective solutes that modulate cell recovery from ultra-low temperatures. Cryobiology. 2017;76:74-91. doi: 10.1016/j.cryobiol.2017.04.004
- Troitzsch RZ, Vass H, Hossack WJ, et al. Molecular mechanisms of cryoprotection in aqueous proline: light scattering and molecular dynamics simulations. J Phys Chem B. 2008;112(14):4290-4297. doi: 10.1021/jp076713m
- Szabados LSavouré A. Proline: a multifunctional amino acid. Trends Plant Sci. 2010;15(2):89-97. doi: 10.1016/j.tplants.2009.11.009
- Kaur G, Asthir B. Proline: a key player in plant abiotic stress tolerance. Biologia Plantarum. 2015;59(4):609-619. doi: 10.1007/s10535-015-0549-3
- Li X, Cao Y, Liu C, et al. l-Proline and GelMA hydrogel complex:An efficient antifreeze system for cell cryopreservation. Cryobiology. 2024;116:104942. doi: 10.1016/j.cryobiol.2024.104942
- Lee SC, Gillispie G, Prim P, et al. Physical and chemical factors influencing the printability of hydrogel-based extrusion bioinks. Chem Rev. 2020;120(19):10834-10886. doi: 10.1021/acs.chemrev.0c00015
- Hannah J, Zhou P. Regulation of DNA damage response pathways by the cullin-RING ubiquitin ligases. DNA Repair. 2009;8(4):536-543. doi: 10.1016/j.dnarep.2009.01.011
- Yue K, Trujillo-de Santiago G, Alvarez MM, et al. Synthesis, properties, and biomedical applications of gelatin methacryloyl (GelMA) hydrogels. Biomaterials. 2015;73:254-271. doi: 10.1016/j.biomaterials.2015.08.045
- Paxton N, Smolan W, Böck T, et al. Proposal to assess printability of bioinks for extrusion-based bioprinting and evaluation of rheological properties governing bioprintability. Biofabrication. 2017;9(4):044107. doi: 10.1088/1758-5090/aa8dd8
- Herrada-Manchón H, Fernández M A, Aguilar E. Essential guide to hydrogel rheology in extrusion 3D printing: how to measure it and why it matters? Gels. 2023;9(7):517. doi: 10.3390/gels9070517
- Zhang D, Liu J, Liu X, et al. Biomechanical and mechanobiological design for bioprinting functional microvasculature. Appl Phys Rev. 2025;12(1):011332. doi: 10.1063/5.0227692
- Kielbassa C, Epe B. DNA damage induced by ultraviolet and visible light and its wavelength dependence. Methods Enzymol. 2000;319:436-445. doi: 10.1016/s0076-6879(00)19041-x
- Elango JZamora-Ledezma C. Rheological, structural, and biological trade-offs in bioink design for 3d bioprinting. Gels. 2025;11(8):659. doi: 10.3390/gels11080659
- Nair K, Gandhi M, Khalil S, et al. Characterization of cell viability during bioprinting processes. Biotechnol J. 2009;4(8):1168-1177. doi: 10.1002/biot.200900004
- Blaeser A, Duarte Campos DF, Puster U, et al. Controlling Shear Stress in 3D Bioprinting is a Key Factor to Balance Printing Resolution and Stem Cell Integrity. Adv Healthc Mater. 2016;5(3):326-333. doi: 10.1002/adhm.201500677
- Xie M, Gao Q, Zhao H, et al. Electro-Assisted Bioprinting of Low-Concentration GelMA Microdroplets. Small. 2019;15(4):e1804216. doi: 10.1002/smll.201804216
- Adhikari J, Roy A, Das A, et al. Effects of Processing Parameters of 3D Bioprinting on the Cellular Activity of Bioinks. Macromol Biosci. 2021;21(1):e2000179. doi: 10.1002/mabi.202000179
- Zhang Y, O’Mahony A, He Y, et al. Hydrodynamic shear stress’ impact on mammalian cell properties and its applications in 3D bioprinting. Biofabrication. 2024;16(2):022003. doi: 10.1088/1758-5090/ad22ee
- Chang SF, Chang CA, Lee DY, et al. Tumor cell cycle arrest induced by shear stress: Roles of integrins and Smad. Proc Natl Acad Sci USA. 2008;105(10):3927-3932. doi: 10.1073/pnas.0712353105
- Ahn G, Min KH, Kim C, et al. Precise stacking of decellularized extracellular matrix based 3D cell-laden constructs by a 3D cell printing system equipped with heating modules. Sci Rep. 2017;7(1):8624. doi: 10.1038/s41598-017-09201-5
- Cadet J, Douki T, Ravanat JL. Oxidatively generated damage to cellular DNA by UVB and UVA radiation. Photochem Photobiol. 2015;91(1):140-155. doi: 10.1111/php.12368
- Mazur P. Cryobiology: the freezing of biological systems. Science. 1970;168(3934):939-949. doi: 10.1126/science.168.3934.939
- Yu M, Marquez-Curtis L, Elliott JAW. Cryopreservation-induced delayed injury and cell-type-specific responses during the cryopreservation of endothelial cell monolayers. Cryobiology. 2024;115:104857. doi: 10.1016/j.cryobiol.2024.104857
- Best B P. Cryoprotectant Toxicity: Facts, Issues, and Questions. Rejuvenation Res. 2015;18(5):422-436. doi: 10.1089/rej.2014.1656

