Three-dimensional printing-based solutions for osteochondral regeneration: Tailoring strategies to region-specific requirements

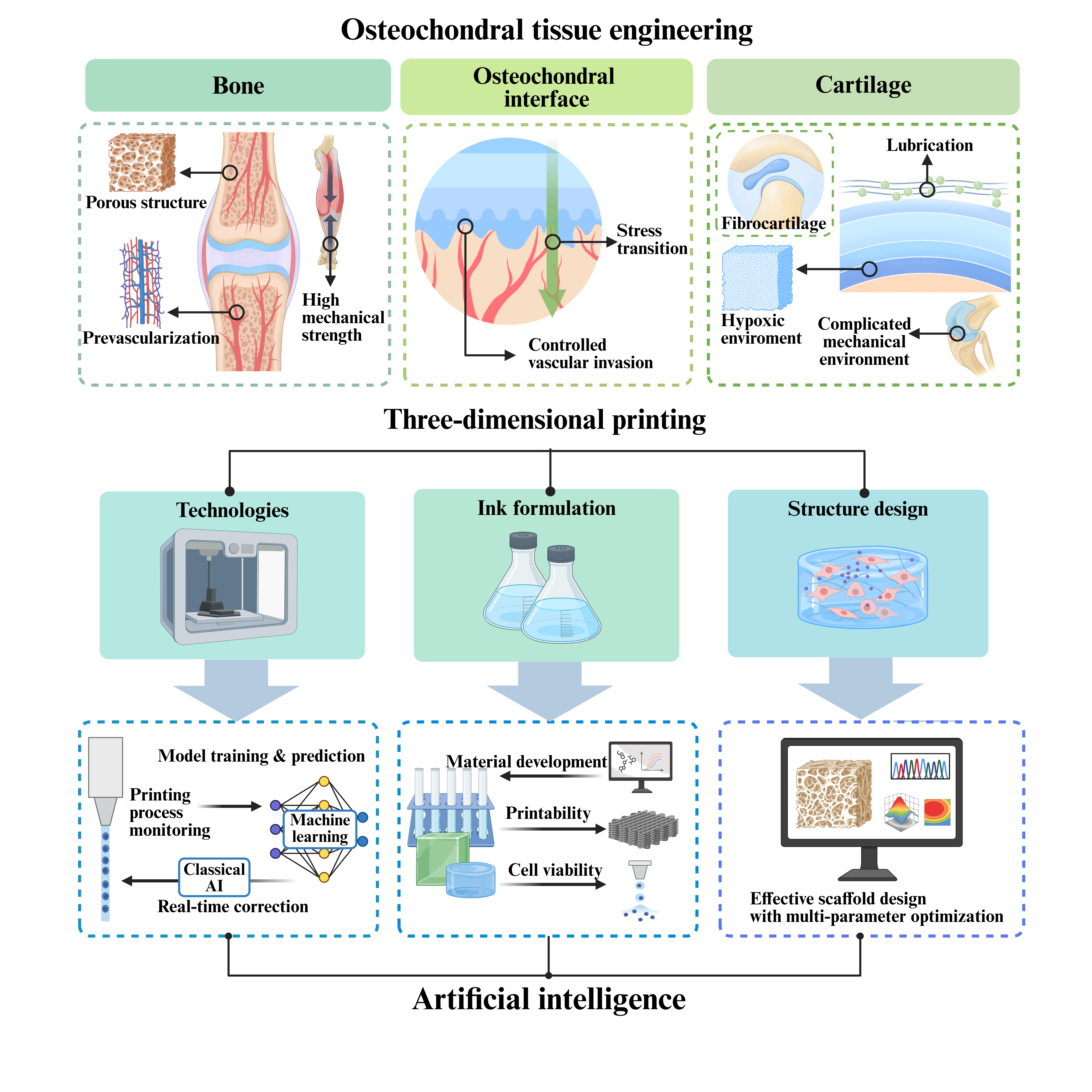
Tissue engineering (TE) holds significant potential for repairing osteochondral (OC) defects caused by trauma and degenerative diseases. However, the structural and functional heterogeneity between cartilage and bone imposes distinct requirements for regenerative outcomes, while stable integration of the OC interface remains a critical clinical hurdle. Three-dimensional (3D) printing technology, leveraging the advantages of personalized manufacturing and precise structural control, systematically optimizes the synergistic application of core TE elements (cells, growth factors, and scaffolds) during fabrication, offering advanced solutions for OC TE. By mimicking the biomechanics and the physiological regulatory mechanisms of native joints, 3D printing facilitates appropriate microenvironments across material, structural, and mechanical levels. Endowed with outstanding reasoning and predictive advantages, artificial intelligence (AI) has greatly advanced the development of 3D printing. In OC TE, AI exhibits promising applications throughout the 3D printing workflow, including printing process parameter regulation, ink evaluation, and scaffold design optimization. This paper systematically reviews OC TE’s general and region-specific requirements, followed by 3D printing’s innovative solutions and AI-assisted breakthroughs. Finally, we discuss the limitations and prospects of this interdisciplinary integration of 3D printing and AI in OC TE.
- Seims KB, Hunt NK, Chow LW. Strategies to Control or Mimic Growth Factor Activity for Bone, Cartilage, and Osteochondral Tissue Engineering. Bioconjug Chem. 2021;32(5):861-878. doi: 10.1021/acs.bioconjchem.1c00090
- Golebiowska AA, Nukavarapu SP. Bio-inspired zonal-structured matrices for bone-cartilage interface engineering. Biofabrication. 2022;14(2). doi: 10.1088/1758-5090/ac5413
- Boyde A. The Bone Cartilage Interface and Osteoarthritis. Calcif Tissue Int. 2021;109(3):303-328. doi: 10.1007/s00223-021-00866-9
- Yang X, Wang L, Chen X, Ling B, Xiao J. Digital light processing 3D bioprinting of collagen-based gradient osteochondral scaffold for cartilage-bone regeneration. Int J Biol Macromol. 2025. doi: 10.1016/j.ijbiomac.2025.148403
- Sun Y, You Y, Jiang W, Wang B, Wu Q, Dai K. 3D bioprinting dual-factor releasing and gradient-structured constructs ready to implant for anisotropic cartilage regeneration. Sci Adv. 2020;6(37):eaay1422. doi: 10.1126/sciadv.aay1422
- Xu Q, Huo L, Wei X, et al. Clinical application of customized total temporomandibular joint prosthesis by 3D printing: a five-year follow-up study. Clin Oral Investig. 2025;29(4):210. doi: 10.1007/s00784-025-06260-1
- Wu X, Wang H, Li C, et al. Antioxidant functionalized double-net/TA dynamic hydrogel promotes cartilage regeneration through stabilization of chondrocyte phenotype. Mater Today Bio. 2025;34:102203. doi: 10.1016/j.mtbio.2025.102203
- Doyle SE, Snow F, Duchi S, et al. 3D Printed Multiphasic Scaffolds for Osteochondral Repair: Challenges and Opportunities. Int J Mol Sci. 2021;22(22):12420. doi: 10.3390/ijms222212420
- Yang Q, Chen A, Zhang X, Wu Z, Zhang C. Functional poly(ether-ketone-ketone) composite scaffold with enhanced cell-material interaction, anti-inflammatory and osteogenesis for facilitating osteointegration and bone regeneration. Mater Today Bio. 2025;31:101533. doi: 10.1016/j.mtbio.2025.101533
- Xulin H, Hu L, Liang Q, et al. 369Fabrication of 3D gel-printed β-tricalcium phosphate/titanium dioxide porous scaffolds for cancellous bone tissue engineering. Int J Bioprint. 2023;9(2):673. doi: 10.18063/ijb.v9i2.673
- Gu X, Zha Y, Li Y, et al. Integrated polycaprolactone microsphere-based scaffolds with biomimetic hierarchy and tunable vascularization for osteochondral repair. Acta Biomater. 2022;141:190-197. doi: 10.1016/j.actbio.2022.01.021
- Wubneh A, Tsekoura EK, Ayranci C, Uludağ H. Current state of fabrication technologies and materials for bone tissue engineering. Acta Biomater. 2018;80:1-30. doi: 10.1016/j.actbio.2018.09.031
- Kouhi M, Khodaei M, Behrouznejad B, Savabi O, Bodaghi M. Zein/ZnO-Modified 3D-Printed PCL/Sphene Scaffolds with Improved Bacterial Inhibition and Osteoblast Activity for Bone Regeneration Applications. ACS Biomater Sci Eng. 2025;11(5):2898-2909. doi: 10.1021/acsbiomaterials.4c02193
- Yang CJ, Huang WK, Lin KP. Three-Dimensional Printing Quality Inspection Based on Transfer Learning with Convolutional Neural Networks. Sensors. 2023;23(1):491. doi: 10.3390/s23010491
- Ren X, Wei J, Luo X, et al. HydrogelFinder: A Foundation Model for Efficient Self-Assembling Peptide Discovery Guided by Non-Peptidal Small Molecules. Adv Sci. 2024;11(26):e2400829. doi: 10.1002/advs.202400829
- Fielder M, Nair AK. Predicting ultrasound wave stimulated bone growth in bioinspired scaffolds using machine learning. J Mech Behav Biomed Mater. 2024;159:106684. doi: 10.1016/j.jmbbm.2024.106684
- Mohammadrezaei D, Podina L, Silva J, Kohandel M. Cell viability prediction and optimization in extrusion-based bioprinting via neural network-based Bayesian optimization models. Biofabrication. 2024;16(2). doi: 10.1088/1758-5090/ad17cf
- Bracco F, Zanderigo G, Paynabar K, Colosimo BM. Leveraging transfer learning for efficient bioprinting. Biofabrication. 2025;17(3). doi: 10.1088/1758-5090/ade62f
- Duan Q, Shao H, Luo N, et al. 3D-printed artificial bone scaffolds: the design of materials, the incorporation of bioactive substances, and the integration of vascularized tissue flaps. Front Bioeng Biotechnol. 2025;13:1614727. doi: 10.3389/fbioe.2025.1614727
- Liu W, Zhang Y, Lyu Y, Bosiakov S, Liu Y. Inverse design of anisotropic bone scaffold based on machine learning and regenerative genetic algorithm. Front Bioeng Biotechnol. 2023;11:1241151. doi: 10.3389/fbioe.2023.1241151
- Chung JK, Im JS, Park MS. Development of Photo- Polymerization-Type 3D Printer for High-Viscosity Ceramic Resin Using CNN-Based Surface Defect Detection. Materials. 2023;16(13):4734. doi: 10.3390/ma16134734
- Zhou L, Gjvm VO, Malda J, et al. Innovative Tissue- Engineered Strategies for Osteochondral Defect Repair and Regeneration: Current Progress and Challenges. Adv Healthc Mater. 2020;9(23):e2001008. doi: 10.1002/adhm.202001008
- Semitela A, Marques PAAP, Completo A. Strategies to engineer articular cartilage with biomimetic zonal features: a review. Biomater Sci. 2024;12(23):5961-6005. doi: 10.1039/d4bm00579a
- Ligon SC, Liska R, Stampfl J, Gurr M, Mülhaupt R. Polymers for 3D Printing and Customized Additive Manufacturing. Chem Rev. 2017;117(15):10212-10290. doi: 10.1021/acs.chemrev.7b00074
- Xia J, Yuan Y, Wu H, Huang Y, Weitz DA. Decoupling the effects of nanopore size and surface roughness on the attachment, spreading and differentiation of bone marrow-derived stem cells. Biomaterials. 2020;248:120014. doi: 10.1016/j.biomaterials.2020.120014
- Yang Y, Xu T, Bei HP, et al. Gaussian curvature-driven direction of cell fate toward osteogenesis with triply periodic minimal surface scaffolds. Proc Natl Acad Sci USA. 2022;119(41):e2206684119. doi: 10.1073/pnas.2206684119
- Chen H, Jiang N, Zhang J, et al. Micron/Submicron Scaled Hierarchical Ti Phosphate/Ti Oxide Hybrid Coating on 3D Printed Scaffolds for Improved Osteointegration. ACS Biomater Sci Eng. 2023;9(3):1274-1284. doi: 10.1021/acsbiomaterials.2c01354
- Gehrke SA, da Costa EM, Júnior JA, Eilers Treichel TL, Del Fabbro M, Scarano A. Comparison Between Micro-and Micro-Nano Surface Texturization in the Initial Osseointegration Process: An Experimental In Vitro and In Vivo Preclinical Study. Bioengineering. 2025;12(2):175. doi: 10.3390/bioengineering12020175
- Critchley S, Sheehy EJ, Cunniffe G, et al. 3D printing of fibre-reinforced cartilaginous templates for the regeneration of osteochondral defects. Acta Biomater. 2020;113:130-143. doi: 10.1016/j.actbio.2020.05.040
- Wu X, Liu S, Chen K, et al. 3D printed chitosan-gelatine hydrogel coating on titanium alloy surface as biological fixation interface of artificial joint prosthesis. Int J Biol Macromol. 2021;182:669-679. doi: 10.1016/j.ijbiomac.2021.04.046
- Tan L, Ye Z, Zhuang W, et al. 3D printed PLGA/MgO/ PDA composite scaffold by low-temperature deposition manufacturing for bone tissue engineering applications. Regen Ther. 2023;24:617-629. doi: 10.1016/j.reth.2023.09.015
- Zhang Y, Han Y, Peng Y, Lei J, Chang F. Bionic biphasic composite scaffolds with osteochondrogenic factors for regeneration of full-thickness osteochondral defects. Biomater Sci. 2022;10(7):1713-1723. doi: 10.1039/d2bm00103a
- Freeman FE, Pitacco P, van Dommelen LHA, et al. 3D bioprinting spatiotemporally defined patterns of growth factors to tightly control tissue regeneration. Sci Adv. 2020;6(33):eabb5093. doi: 10.1126/sciadv.abb5093
- Wang P, Berry D, Moran A, et al. Controlled Growth Factor Release in 3D-Printed Hydrogels. Adv Healthc Mater. 2020;9(15):e1900977. doi: 10.1002/adhm.201900977
- Hunziker EB, Quinn TM, Häuselmann HJ. Quantitative structural organization of normal adult human articular cartilage. Osteoarthritis Cartilage. 2002;10(7):564-572. doi: 10.1053/joca.2002.0814
- Antons J, Marascio MGM, Nohava J, et al. Zone-dependent mechanical properties of human articular cartilage obtained by indentation measurements. J Mater Sci Mater Med. 2018;29(5):57. doi: 10.1007/s10856-018-6066-0
- Dennis JE, Whitney GA, Rai J, Fernandes RJ, Kean TJ. Physioxia Stimulates Extracellular Matrix Deposition and Increases Mechanical Properties of Human Chondrocyte- Derived Tissue-Engineered Cartilage. Front Bioeng Biotechnol. 2020;8:590743. doi: 10.3389/fbioe.2020.590743
- Motavalli M, Akkus O, Mansour JM. Depth-dependent shear behavior of bovine articular cartilage: relationship to structure. J Anat. 2014;225(5):519-526. doi: 10.1111/joa.12230
- Yu X, Hu Y, Zou L, et al. A bilayered scaffold with segregated hydrophilicity-hydrophobicity enables reconstruction of goat hierarchical temporomandibular joint condyle cartilage. Acta Biomater. 2021;121:288-302. doi: 10.1016/j.actbio.2020.11.031
- Camarero-Espinosa S, Rothen-Rutishauser B, Foster EJ, Weder C. Articular cartilage: from formation to tissue engineering. Biomater Sci. 2016;4(5):734-767. doi: 10.1039/c6bm00068a
- Lei X, Wang X, Li Y, et al. Comparison of knee joint and temporomandibular joint development in pig embryos. Anim Biotechnol. 2024;35(1):2337760. doi: 10.1080/10495398.2024.2337760
- Kupratis ME, Rahman A, Burris DL, Corbin EA, Price C. Enzymatic digestion does not compromise sliding-mediated cartilage lubrication. Acta Biomater. 2024;178:196-207. doi: 10.1016/j.actbio.2024.02.040
- Lin W, Klein J. Recent Progress in Cartilage Lubrication. Adv Mater. 2021;33(18):e2005513. doi: 10.1002/adma.202005513
- Forster H, Fisher J. The influence of loading time and lubricant on the friction of articular cartilage. Proc Inst Mech Eng H. 1996;210(2):109-119. doi: 10.1243/PIME_PROC_1996_210_399_02
- Pouran B, Raoof A, de Winter DAM, et al. Topographic features of nano-pores within the osteochondral interface and their effects on transport properties -a 3D imaging and modeling study. J Biomech. 2021;123:110504. doi: 10.1016/j.jbiomech.2021.110504
- Ruggiero L, Zimmerman BK, Park M, et al. Roles of the Fibrous Superficial Zone in the Mechanical Behavior of TMJ Condylar Cartilage. Ann Biomed Eng. 2015;43(11):2652- 2662. doi: 10.1007/s10439-015-1320-9
- Buckley MR, Gleghorn JP, Bonassar LJ, Cohen I. Mapping the depth dependence of shear properties in articular cartilage. J Biomech. 2008;41(11):2430-2437. doi: 10.1016/j.jbiomech.2008.05.021
- Barthold JE, McCreery KP, Martinez J, et al. Particulate ECM biomaterial ink is 3D printed and naturally crosslinked to form structurally-layered and lubricated cartilage tissue mimics. Biofabrication. 2022;14(2):025021. doi: 10.1088/1758-5090/ac584c
- Zubillaga V, Alonso-Varona A, Fernandes SCM, Salaberria AM, Palomares T. Adipose-Derived Mesenchymal Stem Cell Chondrospheroids Cultured in Hypoxia and a 3D Porous Chitosan/Chitin Nanocrystal Scaffold as a Platform for Cartilage Tissue Engineering. Int J Mol Sci. 2020;21(3):1004. doi: 10.3390/ijms21031004
- Theodoridis K, Aggelidou E, Manthou ME, Kritis A. Hypoxia Promotes Cartilage Regeneration in Cell-Seeded 3D-Printed Bioscaffolds Cultured with a Bespoke 3D Culture Device. Int J Mol Sci. 2023;24(7):6040. doi: 10.3390/ijms24076040
- Jiang N, Su Z, Sun Y, et al. Spatial Heterogeneity Directs Energy Dissipation in Condylar Fibrocartilage. Small. 2023;19(37):e2301051. doi: 10.1002/smll.202301051
- Stocum DL, Roberts WE. Part I: Development and Physiology of the Temporomandibular Joint. Curr Osteoporos Rep. 2018;16(4):360-368. doi: 10.1007/s11914-018-0447-7
- Daly AC, Critchley SE, Rencsok EM, Kelly DJ. A comparison of different bioinks for 3D bioprinting of fibrocartilage and hyaline cartilage. Biofabrication. 2016;8(4):045002. doi: 10.1088/1758-5090/8/4/045002
- Gologorsky CJ, Middendorf JM, Cohen I, Bonassar LJ. Depth-dependent patterns in shear modulus of temporomandibular joint cartilage correspond to tissue structure and anatomic location. J Biomech. 2021;129:110815. doi: 10.1016/j.jbiomech.2021.110815
- Hu S, Yi Y, Ye C, Liu J, Wang J. Advances in 3D printing techniques for cartilage regeneration of temporomandibular joint disc and mandibular condyle. Int J Bioprint. 2023;9(5):761. doi: 10.18063/ijb.761
- Wang X, Wang J, Zhang Y, He Y, Chen S. Piezo1 regulates fibrocartilage stem cell in cartilage growth and osteoarthritis. Osteoarthritis Cartilage. 2025;33(8):980-991. doi: 10.1016/j.joca.2025.04.013
- Zhao Y, Xie L. An Update on Mesenchymal Stem Cell- Centered Therapies in Temporomandibular Joint Osteoarthritis. Stem Cells Int. 2021;2021:6619527. doi: 10.1155/2021/6619527
- Su FY, Pang S, Ling YTT, et al. Deproteinization of Cortical Bone: Effects of Different Treatments. Calcif Tissue Int. 2018;103(5):554-566. doi: 10.1007/s00223-018-0453-x
- Beverly M, Marks BE, Murray DW. Subchondral pressures and perfusion during weight bearing. J Orthop Surg Res. 2020;15(1):239. doi: 10.1186/s13018-020-01754-y
- Chen H, Liu Y, Wang C, et al. Design and properties of biomimetic irregular scaffolds for bone tissue engineering. Comput Biol Med. 2021;130:104241. doi: 10.1016/j.compbiomed.2021.104241
- Jiao J, Hong Q, Zhang D, et al. Influence of porosity on osteogenesis, bone growth and osteointegration in trabecular tantalum scaffolds fabricated by additive manufacturing. Front Bioeng Biotechnol. 2023;11:1117954. doi: 10.3389/fbioe.2023.1117954
- Cheng A, Humayun A, Cohen DJ, Boyan BD, Schwartz Z. Additively manufactured 3D porous Ti-6Al-4V constructs mimic trabecular bone structure and regulate osteoblast proliferation, differentiation and local factor production in a porosity and surface roughness dependent manner. Biofabrication. 2014;6(4):045007. doi: 10.1088/1758-5082/6/4/045007
- Kumar S, Tan S, Zheng L, Kochmann DM. Inverse-designed spinodoid metamaterials. npj Comput Mater. 2020;6:73. doi: 10.1038/s41524-020-0341-6
- Zhang M, Lin R, Wang X, et al. 3D printing of Haversian bone-mimicking scaffolds for multicellular delivery in bone regeneration. Sci Adv. 2020;6(12):eaaz6725. doi: 10.1126/sciadv.aaz6725
- Pathria MN, Chung CB, Resnick DL. Acute and Stress-related Injuries of Bone and Cartilage: Pertinent Anatomy, Basic Biomechanics, and Imaging Perspective. Radiology. 2016;280(1):21-38. doi: 10.1148/radiol.16142305
- Burke G, Devine DM, Major I. Effect of Stereolithography 3D Printing on the Properties of PEGDMA Hydrogels. Polymers. 2020;12(9):2015. doi: 10.3390/polym12092015
- Sun T, Huang H, Zhao Y, Li Z, Wang H, Zhou G. Low- Temperature Deposited Amorphous Poly(aryl ether ketone) Hierarchically Porous Scaffolds with Strontium-Doped Mineralized Coating for Bone Defect Repair. Adv Healthc Mater. 2024;13(23):e2400927. doi: 10.1002/adhm.202400927
- Ghobadi E, Yahay Z, Nouri N, Karamali F, Masaeli E. 3D printing of an anatomically shaped bone model inspired by vascularized tubular bone structure. Biomater Adv. 2025;176:214348. doi: 10.1016/j.bioadv.2025.214348
- Chen H, Gonnella G, Huang J, Di-Silvio L. Fabrication of 3D Bioprinted Bi-Phasic Scaffold for Bone-Cartilage Interface Regeneration. Biomimetics. 2023;8(1):87. doi: 10.3390/biomimetics8010087
- Wang L, Chen X, Wang X, Chen H, Yang X, Xiao J. Highly biomimetic three-layer mineralized collagen scaffold featuring a wood-reinforced subchondral bone region for gradient chondrogenic-osteogenic differentiation of bone marrow-derived mesenchymal stem cells. Int J Biol Macromol. 2025;320(Pt 1):145754. doi: 10.1016/j.ijbiomac.2025.145754
- Zhao Y, Cai Y, Wang W, et al. Periosteum-bone inspired hierarchical scaffold with endogenous piezoelectricity for neuro-vascularized bone regeneration. Bioact Mater. 2024;44:339-353. doi: 10.1016/j.bioactmat.2024.10.020
- Tang J, Liu D. Study on the Shock-Absorption Performance of Isolation Systems in High-Rise Vertically Irregular Double-Story Structures. Buildings. 2024; 14(12):3792. doi: 10.3390/buildings14123792
- Wu D, Zheng K, Yin W, et al. Enhanced osteochondral regeneration with a 3D-Printed biomimetic scaffold featuring a calcified interfacial layer. Bioact Mater. 2024;36:317-329. doi: 10.1016/j.bioactmat.2024.03.004
- Zhou Y, Lian XJ, Lu Y, et al. Harnessing oriented arrangement of collagen fibers by 3D printing for enhancing mechanical and osteogenic properties of mineralized collagen scaffolds. Biomed Mater. 2024;19(4):045020. doi: 10.1088/1748-605X/ad5244
- Burdis R, Chariyev-Prinz F, Kelly DJ. Bioprinting of biomimetic self-organised cartilage with a supporting joint fixation device. Biofabrication. 2021;14(1):015008. doi: 10.1088/1758-5090/ac36be
- Wang Z, Tuerxun P, Ng T, et al. Enhancing angiogenesis in peri-implant soft tissue with bioactive silk fibroin microgroove coatings on zirconia surfaces. Regen Biomater. 2024;11:rbae068. doi: 10.1093/rb/rbae068
- Bedell ML, Torres AL, Hogan KJ, et al. Human gelatin-based composite hydrogels for osteochondral tissue engineering and their adaptation into bioinks for extrusion, inkjet, and digital light processing bioprinting. Biofabrication. 2022;14(4):045012. doi: 10.1088/1758-5090/ac8768
- Zub K, Hoeppener S, Schubert US. Inkjet Printing and 3D Printing Strategies for Biosensing, Analytical, and Diagnostic Applications. Adv Mater. 2022;34(31):e2105015. doi: 10.1002/adma.202105015
- Xie M, Gao Q, Zhao H, et al. Electro-Assisted Bioprinting of Low-Concentration GelMA Microdroplets. Small. 2019;15(4):e1804216. doi: 10.1002/smll.201804216
- Li K, Zhang F, Wang D, et al. Silkworm-inspired electrohydrodynamic jet 3D printing of composite scaffold with ordered cell scale fibers for bone tissue engineering. Int J Biol Macromol. 2021;172:124-132. doi: 10.1016/j.ijbiomac.2021.01.013
- Michailidou G, Terzopoulou Z, Kehagia A, Michopoulou A, Bikiaris DN. Preliminary Evaluation of 3D Printed Chitosan/Pectin Constructs for Biomedical Applications. Mar Drugs. 2021;19(1):36. doi: 10.3390/md19010036
- Lee J, Oh SJ, An SH, Kim WD, Kim SH. Machine learning-based design strategy for 3D printable bioink: elastic modulus and yield stress determine printability. Biofabrication. 2020;12(3):035018. doi: 10.1088/1758-5090/ab8707
- Putra NE, Leeflang MA, Klimopoulou M, et al. Extrusion-based 3D printing of biodegradable, osteogenic, paramagnetic, and porous FeMn-akermanite bone substitutes. Acta Biomater. 2023;162:182-198. doi: 10.1016/j.actbio.2023.03.033
- Salehi S, Ghomi H, Hassanzadeh-Tabrizi SA, Koupaei N, Khodaei M. Antibacterial and osteogenic properties of chitosan-polyethylene glycol nanofibre-coated 3D printed scaffold with vancomycin and insulin-like growth factor-1 release for bone repair. Int J Biol Macromol. 2025;298:139883. doi: 10.1016/j.ijbiomac.2025.139883
- Kuss MA, Wu S, Wang Y, et al. Prevascularization of 3D printed bone scaffolds by bioactive hydrogels and cell co-culture. J Biomed Mater Res B Appl Biomater. 2018;106(5):1788-1798. doi: 10.1002/jbm.b.33994
- Xiong Z, Yan Y, Wang S, Zhang R, Zhang C. Fabrication of porous scaffolds for bone tissue engineering via low-temperature deposition. Scripta Materialia. 2002;46(11):771- 776. doi: 10.1016/S1359-6462(02)00071-4.
- Li Q, Xu S, Feng Q, et al. 3D printed silk-gelatin hydrogel scaffold with different porous structure and cell seeding strategy for cartilage regeneration. Bioact Mater. 2021;6(10):3396-3410. doi: 10.1016/j.bioactmat.2021.03.013
- Liu W, Wang D, Huang J, et al. Low-temperature deposition manufacturing: A novel and promising rapid prototyping technology for the fabrication of tissue-engineered scaffold. Mater Sci Eng C Mater Biol Appl. 2017;70(Pt 2):976-982. doi: 10.1016/j.msec.2016.04.014
- Luo Y, Pan H, Jiang J, et al. Desktop-Stereolithography 3D Printing of a Polyporous Extracellular Matrix Bioink for Bone Defect Regeneration. Front Bioeng Biotechnol. 2020;8:589094. doi: 10.3389/fbioe.2020.589094
- Wang Z, Huang C, Wang J, et al. Design and characterization of hydroxyapatite scaffolds fabricated by stereolithography for bone tissue engineering application. Procedia CIRP. 2020;89:170-175. doi: 10.1016/j.procir.2020.05.138
- Eckstein KN, Hergert JE, Uzcategui AC, et al. Controlled Mechanical Property Gradients Within a Digital Light Processing Printed Hydrogel-Composite Osteochondral Scaffold. Ann Biomed Eng. 2024;52(8):2162-2177. doi: 10.1007/s10439-024-03516-x
- Dobos A, Van Hoorick J, Steiger W, et al. Thiol-Gelatin- Norbornene Bioink for Laser-Based High-Definition Bioprinting. Adv Healthc Mater. 2020;9(15):e1900752. doi: 10.1002/adhm.201900752
- Chen Z, Li K, Han P, et al. Stereolithography 3D printing gyroid triply periodic minimal surface vitrified bond diamond grinding wheel. Sci Rep. 2024;14(1):30054. doi: 10.1038/s41598-024-81641-2
- Shuai C, Li D, Xie H, Yao X, Peng S, Gao C. Programmable Lamellar Eutectic Zn-2Al-Mg Biodegradable Implants Manufactured by Laser Powder Bed Fusion for Synergistic Strength-Ductility and Osteogenesis. Adv Healthc Mater. 2025. doi: 10.1002/adhm.202501917
- Kérourédan O, Washio A, Handschin C, et al. Bioactive gelatin-sheets as novel biopapers to support prevascularization organized by laser-assisted bioprinting for bone tissue engineering. Biomed Mater. 2024;19(2):025038. doi: 10.1088/1748-605X/ad270a
- Chen Z, Yan X, Yin S, et al. Influence of the pore size and porosity of selective laser melted Ti6Al4V ELI porous scaffold on cell proliferation, osteogenesis and bone ingrowth. Mater Sci Eng C Mater Biol Appl. 2020;106:110289. doi: 10.1016/j.msec.2019.110289
- Yuan X, Zhu W, Yang Z, et al. Recent Advances in 3D Printing of Smart Scaffolds for Bone Tissue Engineering and Regeneration. Adv Mater. 2024;36(34):e2403641. doi: 10.1002/adma.202403641
- Grant-Jacob JA, Zervas MN, Mills B. Laser induced forward transfer imaging using deep learning. Discov Appl Sci. 2025;7(4):254. doi: 10.1007/s42452-025-06679-x
- Zheng Z, Yu D, Wang H, et al. Advancement of 3D biofabrication in repairing and regeneration of cartilage defects. Biofabrication. 2025;17(2):022003. doi: 10.1088/1758-5090/ada8e1
- Hall GN, Fan Y, Viellerobe B, et al. Laser-assisted bioprinting of targeted cartilaginous spheroids for high density bottom-up tissue engineering. Biofabrication. 2024;16(4):045029. doi: 10.1088/1758-5090/ad6e1a
- Qian Y, Gu Y, Tribukait-Riemenschneider F, Martin I, Shastri VP. Incorporation of Cross-Linked Gelatin Microparticles To Enhance Cell Attachment and Chondrogenesis in Carboxylated Agarose Bioinks for Cartilage Engineering. ACS Appl Mater Interfaces. 2025;17(15):22293-22307. doi: 10.1021/acsami.5c00077
- Feng C, Zhang W, Deng C, et al. 3D Printing of Lotus Root- Like Biomimetic Materials for Cell Delivery and Tissue Regeneration. Adv Sci (Weinh). 2017;4(12):1700401. doi: 10.1002/advs.201700401
- Wu Z, Li Z, Wu Y, Hong Y. The Loading of Dipyridamole and Calcium Sulfate into the Gelatin-Coated Porous Bioceramics to Synergistically Regulate Segmental Bone Regeneration. ACS Appl Mater Interfaces. 2025;17(43):59108-59123. doi: 10.1021/acsami.5c14640
- Jiang Y, Zhou C, Yang X, Ke D. 3D printed bioactive coated scaffolds boost osteogenesis and angiogenesis via the regulation of scaffold microstructure. Biofabrication. 2025;17(3):035017. doi: 10.1088/1758-5090/addc9c
- Buckley C, Wang H, O’Dell R, et al. Creation of Porous, Perfusable Microtubular Networks for Improved Cell Viability in Volumetric Hydrogels. ACS Appl Mater Interfaces. 2024;16(15):18522-18533. doi: 10.1021/acsami.4c00716
- Li D, Luo F, Yang Y, et al. Molten stringing 3D printed microfibrous net-integrated mineralized hydrogels with tunable micromechanical and cell-responsive properties. J Mater Chem B. 2025;13(31):9536-9549. doi: 10.1039/d5tb00449g
- Feng X, Xu P, Shen T, Zhang Y, Ye J, Gao C. Influence of pore architectures of silk fibroin/collagen composite scaffolds on the regeneration of osteochondral defects in vivo. J Mater Chem B. 2020;8(3):391-405. doi: 10.1039/c9tb01558b
- Kim MJ, Lee B, Yang K, et al. BMP-2 peptide-functionalized nanopatterned substrates for enhanced osteogenic differentiation of human mesenchymal stem cells. Biomaterials. 2013;34(30):7236-7246. doi: 10.1016/j.biomaterials.2013.06.019
- He L, Zhao M, Cheung JPY, Zhang T, Ren X. Gaussian random field-based characterization and reconstruction of cancellous bone microstructure considering the constraint of correlation structure. J Mech Behav Biomed Mater. 2024;152:106443. doi: 10.1016/j.jmbbm.2024.106443
- Takagishi T, Yoshioka H, Mikami Y, Oki Y. On-demand inkjet-printed microdisk laser with air cladding by liquid flow microetching. Appl Opt. 2020;59(21):6340-6346. doi: 10.1364/AO.396061
- Mushtaq RT, Rehman M, Bao C, et al. Enhanced biomechanical compatibility of 3D-printed polylactic acid lattice structures: Synergizing mechanical, topography, and microstructural properties for trabecular bone mimicry. Int J Biol Macromol. 2025;317(Pt 2):144373. doi: 10.1016/j.ijbiomac.2025.144373
- Enriquez-Ochoa D, Robles-Ovalle P, Mayolo-Deloisa K, Brunck MEG. Immobilization of Growth Factors for Cell Therapy Manufacturing. Front Bioeng Biotechnol. 2020;8:620. doi: 10.3389/fbioe.2020.00620
- Alarcin E, Akguner ZP, Ozturk AB, et al. Biomimetic 3D bioprinted bilayer GelMA scaffolds for the delivery of BMP-2 and VEGF exogenous growth factors to promote vascularized bone regeneration in a calvarial defect model in vivo. Int J Biol Macromol. 2025;306(Pt 2):141440. doi: 10.1016/j.ijbiomac.2025.141440
- He W, Li C, Zhao S, et al. Integrating coaxial electrospinning and 3D printing technologies for the development of biphasic porous scaffolds enabling spatiotemporal control in tumor ablation and osteochondral regeneration. Bioact Mater. 2024;34:338-353. doi: 10.1016/j.bioactmat.2023.12.020
- Loxley GA, Coser C, Ghaemmaghami AM, Yang J. Long-term interleukin-4 release from 3D printable affinity hydrogels promotes M2-like macrophage polarisation in vitro. Biomater Sci. 2025;13(9):2489-2502. doi: 10.1039/d4bm01623h
- Wei P, Ma Y, Qin K, Fan Z. A 3D printed biomimetic scaffold for cartilage regeneration with lubrication, load-bearing, and adhesive fixation properties. Tribol Int. 2024;192:109328. doi: 10.1016/j.triboint.2024.109328
- Ngadimin KD, Stokes A, Gentile P, Ferreira AM. Biomimetic hydrogels designed for cartilage tissue engineering. Biomater Sci. 2021;9(12):4246-4259. doi: 10.1039/d0bm01852j
- Sasikumar SC, Goswami U, Raichur AM. Mucin-Based Dual Cross-Linkable IPN Hydrogel Bioink for 3D Bioprinting and Cartilage Tissue Engineering. ACS Appl Bio Mater. 2025;8(2):1186-1200. doi: 10.1021/acsabm.4c01505
- Wu M, Liu H, Zhu Y, et al. Bioinspired soft-hard combined system with mild photothermal therapeutic activity promotes diabetic bone defect healing via synergetic effects of immune activation and angiogenesis. Theranostics. 2024;14(10):4014-4057. doi: 10.7150/thno.97335
- Yang J, Wang H, Huang W, et al. A natural polymer-based hydrogel with shape controllability and high toughness and its application to efficient osteochondral regeneration. Mater Horiz. 2023;10(9):3797-3806. doi: 10.1039/d3mh00544e
- Gupta S, Sharma A, Vasantha Kumar J, Sharma V, Gupta PK, Verma RS. Meniscal tissue engineering via 3D printed PLA monolith with carbohydrate based self-healing interpenetrating network hydrogel. Int J Biol Macromol. 2020;162:1358-1371. doi: 10.1016/j.ijbiomac.2020.07.238
- Trachsel L, Johnbosco C, Lang T, Benetti EM, Zenobi-Wong M. Double-Network Hydrogels Including Enzymatically Crosslinked Poly-(2-alkyl-2-oxazoline)s for 3D Bioprinting of Cartilage-Engineering Constructs. Biomacromolecules. 2019;20(12):4502-4511. doi: 10.1021/acs.biomac.9b01266
- Chen Q, Liu S, Yuan Z, Yang H, Xie R, Ren L. Construction and Tribological Properties of Biomimetic Cartilage- Lubricating Hydrogels. Gels. 2022;8(7):415. doi: 10.3390/gels8070415
- Ali N, Demott CJ, Dingus OF, Grunlan MA, Dunn AC. Network interactions simultaneously enhance stiffness and lubricity of triple-network hydrogels. Soft Matter. 2024;20(44):8783-8792. doi: 10.1039/d4sm00969j
- Demott CJ, Jones MR, Chesney CD, Grunlan MA. Adhesive Hydrogel Building Blocks to Reconstruct Complex Cartilage Tissues. ACS Biomater Sci Eng. 2023;9(4):1952-1960. doi: 10.1021/acsbiomaterials.2c01438
- Xu F, Zhuang C, Yao L, et al. High-mobility network hydrogel microsphere system to combat chondrocyte senescence for enhanced cartilage repair and regeneration. Mater Today Bio. 2025;34:102138. doi: 10.1016/j.mtbio.2025.102138
- Lin W, Kluzek M, Iuster N, et al. Cartilage-inspired, lipid-based boundary-lubricated hydrogels. Science. 2020;370(6514):335-338. doi: 10.1126/science.aay8276
- Li S, Tang L, Pu J, et al. Continuous Hyaluronic Acid Supply by a UHMWPE/PEEK Interlocking Scaffold for Metatarsophalangeal Joint Prosthesis Lubricating Applications. ACS Appl Mater Interfaces. 2025;17(8):11704- 11717. doi: 10.1021/acsami.4c19390
- Kaneko D, Tada T, Kurokawa T, Gong J. P, Osada Y. Mechanically Strong Hydrogels with Ultra-Low Frictional Coefficients. Adv. Mater. 2005, 17 (5), 535–538. doi: 10.1002/adma.200400739
- Milner PE, Parkes M, Puetzer JL, et al. A low friction, biphasic and boundary lubricating hydrogel for cartilage replacement. Acta Biomater. 2018;65:102-111. doi: 10.1016/j.actbio.2017.11.002
- Zhang S, Wang L, Kang Y, Wu J, Zhang Z. Nanomaterial-based reactive oxygen species scavengers for osteoarthritis therapy. Acta Biomater. 2023;162:1-19. doi: 10.1016/j.actbio.2023.03.030
- Shafiq M, Chen Y, Hashim R, He C, Mo X, Zhou X. Reactive Oxygen Species-Based Biomaterials for Regenerative Medicine and Tissue Engineering Applications. Front Bioeng Biotechnol. 2021;9:821288. doi: 10.3389/fbioe.2021.821288
- Chen Y, Le Y, Yang J, et al. 3D Bioprinted Xanthan Hydrogels with Dual Antioxidant and Chondrogenic Functions for Post-traumatic Cartilage Regeneration. ACS Biomater Sci Eng. 2024;10(3):1661-1675. doi: 10.1021/acsbiomaterials.3c01636
- Sun X, Xu X, Zhao X, et al. Three-Dimensional Bioprinted Scaffolds Loaded with Multifunctional Magnesium-Based Metal-Organic Frameworks Improve the Senescence Microenvironment Prompting Aged Bone Defect Repair. ACS Nano. 2025;19(24):22141-22162. doi: 10.1021/acsnano.5c03023
- Wang X, Wu S, Li R, et al. ROS-Activated Nanohydrogel Scaffolds with Multi-Factors Controlled Release for Targeted Dual-Lineage Repair of Osteochondral Defects. Adv Sci. 2025;12(20):e2412410. doi: 10.1002/advs.202412410
- He F, Wu H, He B, Han Z, Chen J, Huang L. Antioxidant hydrogels for the treatment of osteoarthritis: mechanisms and recent advances. Front Pharmacol. 2024;15:1488036. Published 2024 Oct 25. doi: 10.3389/fphar.2024.1488036
- Kuang Y, Hua B, Ye X, Zhao Y, Yu M, Liu X. Dual-functional ROS-responsive hydrogel alleviates temporomandibular joint osteoarthritis by enhancing cartilage repair and mitigating inflammation. Mater Today Bio. 2025;33:102103. doi: 10.1016/j.mtbio.2025.102103
- Shi W, Fang F, Kong Y, et al. Dynamic hyaluronic acid hydrogel with covalent linked gelatin as an anti-oxidative bioink for cartilage tissue engineering. Biofabrication. 2021;14(1):10.1088/1758-5090/ac42de. doi: 10.1088/1758-5090/ac42d
- Shu C, Qin C, Chen L, et al. Metal-Organic Framework Functionalized Bioceramic Scaffolds with Antioxidative Activity for Enhanced Osteochondral Regeneration. Adv Sci. 2023;10(13):e2206875. doi: 10.1002/advs.202206875
- Zhu S, Zhou Z, Chen X, et al. High mechanical performance and multifunctional degraded fucoidan-derived bioink for 3D bioprinting. Carbohydr Polym. 2025;348(Pt A):122805. doi: 10.1016/j.carbpol.2024.122805
- Helgeland E, Mohamed-Ahmed S, Shanbhag S, et al. 3D printed gelatin-genipin scaffolds for temporomandibular joint cartilage regeneration. Biomed Phys Eng Express. 2021;7(5):055025. doi: 10.1088/2057-1976/ac1e68
- Kalpakci KN, Kim EJ, Athanasiou KA. Assessment of growth factor treatment on fibrochondrocyte and chondrocyte co-cultures for TMJ fibrocartilage engineering. Acta Biomater. 2011;7(4):1710-1718. doi: 10.1016/j.actbio.2010.12.015
- Yu X, Hu Y, Zou L, et al. A bilayered scaffold with segregated hydrophilicity-hydrophobicity enables reconstruction of goat hierarchical temporomandibular joint condyle cartilage. Acta Biomater. 2021;121:288-302. doi: 10.1016/j.actbio.2020.11.031
- Dufour A, Essayan L, Kim B, Salles V, Marquette C. Biofabrication of Spatially Organized Temporo-mandibular Fibrocartilage Assembloids. Adv Healthc Mater. 2025;14(15):e2405000. doi: 10.1002/adhm.202405000
- Jeong HJ, Koch A, Park S, Tarafder S, Lee CH. Bioactive scaffolds integrated with micro-precise spatiotemporal delivery and in vivo degradation tracking for complex tissue regeneration. Eng Regen. 2025;6:34-44. doi: 10.1016/j.engreg.2025.01.001
- Yi P, Liang J, Huang F, et al. Composite system of 3D-printed polymer and acellular matrix hydrogel to repair temporomandibular joint disc. Front Mater. 2021;8:621416. doi: 10.3389/fmats.2021.621416
- Yu Z, Xing F, Li J, et al. 3D printed polycaprolactone/ phosphoester-modified poly(amino acid)-graphene oxide scaffold for meniscal regeneration. J Mater Chem B. 2025;13(35):11055-11074. doi: 10.1039/d5tb00012b
- Trindade D, Alves N, Moura C. From Animal to Human: (Re)using Acellular Extracellular Matrices for Temporomandibular Disc Substitution. J Funct Biomater. 2022;13(2):61. doi: 10.3390/jfb13020061
- Jiang N, Chen H, Zhang J, et al. Decellularized-disc based allograft and xenograft prosthesis for the long-term precise reconstruction of temporomandibular joint disc. Acta Biomater. 2023;159:173-187. doi: 10.1016/j.actbio.2023.01.042
- Zhang J, Xie L, She Y, Luo H, Zhu S, Jiang N. Microstructural and Micromechanical Properties of Decellularized Fibrocartilaginous Scaffold. ACS Biomater Sci Eng. 2025;11(3):1562-1570. doi: 10.1021/acsbiomaterials.4c01195
- Lee CH, Rodeo SA, Fortier LA, Lu C, Erisken C, Mao JJ. Protein-releasing polymeric scaffolds induce fibrochondrocytic differentiation of endogenous cells for knee meniscus regeneration in sheep. Sci Transl Med. 2014;6(266):266ra171. doi: 10.1126/scitranslmed.3009696
- Shen S, Li Y, Chen M, et al. Bionic scaffolds with integrated structural components based on low-temperature deposition manufacturing 3D printing technology for the treatment of meniscus defects. Bioeng Transl Med. 2025;10(5):e70022. doi: 10.1002/btm2.70022
- Savin G, Caillol S, Bethry A, et al. Collagen/polyester-polyurethane porous scaffolds for use in meniscal repair. Biomater Sci. 2024;12(11):2960-2977. doi: 10.1039/d4bm00234b
- Kremer A, Ribitsch I, Reboredo J, et al. Three-Dimensional Coculture of Meniscal Cells and Mesenchymal Stem Cells in Collagen Type I Hydrogel on a Small Intestinal Matrix-A Pilot Study Toward Equine Meniscus Tissue Engineering. Tissue Eng Part A. 2017;23(9-10):390-402. doi: 10.1089/ten.TEA.2016.0317
- Shen S, Chen M, Gao S, et al. 3D da yin zhi bei ju ji nei zhi/I xing jiao yuan zu zhi gong cheng ban yue ban zhi jia ji qi li hua te xing de yan jiu [Study on the preparation of polycaprolactone/type I collagen tissue engineered meniscus scaffold by three-dimensional printing and its physiochemical properties]. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2018;32(9):1205-1210. [In Chinese]. doi: 10.7507/1002-1892.201803074
- Yao K, Guo K, Wang H, Zheng X. Multi-Nozzles 3D Bioprinting Collagen/Thermoplastic Elasto-Mer Scaffold with Interconnect Pores. Micromachines. 2025;16(4):429. doi: 10.3390/mi16040429
- Gupta T, Rath P, Pakharenko V, et al. Bioinspired thermoreversible bioink orchestrates focal adhesion-dependent osteogenesis. Trends Biotechnol. 2026;44(1):239- 265. doi: 10.1016/j.tibtech.2025.09.007
- Liang R, Li R, Mo W, et al. Engineering biomimetic silk fibroin hydrogel scaffolds with “organic-inorganic assembly” strategy for rapid bone regeneration. Bioact Mater. 2024;40:541-556. doi: 10.1016/j.bioactmat.2024.06.024
- Lu Y, Gong T, Yang Z, Zhu H, Liu Y, Wu C. Designing anisotropic porous bone scaffolds using a self-learning convolutional neural network model. Front Bioeng Biotechnol. 2022;10:973275. doi: 10.3389/fbioe.2022.973275
- Liang H, Chao L, Xie D, et al. Trabecular-like Ti–6Al–4V scaffold for bone repair: A diversified mechanical stimulation environment for bone regeneration. Compos. Part B Eng. 2022, 241:110057. doi: 10.1016/j.compositesb.2022.110057
- Luo L, Zheng W, Li J, et al. 3D-Printed Titanium Trabecular Scaffolds with Sustained Release of Hypoxia-Induced Exosomes for Dual-Mimetic Bone Regeneration. Adv Sci. 2025;12(23):e2500599. doi: 10.1002/advs.202500599
- Lee SH, Lee J, Kang NU, et al. PCL scaffold with well-defined hierarchical pores effectively controls cell migration and alignment of human mesenchymal stem cells. Sci Rep. 2025;15(1):11542. doi: 10.1038/s41598-025-96027-1
- Tariq S, Shah SA, Hameed F, et al. Tissue engineered periosteum: Fabrication of a gelatin basedtrilayer composite scaffold with biomimetic properties for enhanced bone healing. Int J Biol Macromol. 2024;263(Pt 2):130371. doi: 10.1016/j.ijbiomac.2024.130371
- Feng H, Lian X, Lv S, et al. Bioinspired bilayer 3D printing periosteum scaffold with hierarchical structure based on silk fibroin and sodium alginate for bone regeneration. Int J Biol Macromol. 2025;310(Pt 2):143175. doi: 10.1016/j.ijbiomac.2025.143175
- Chang CH, Lin FH, Lin CC, Chou CH, Liu HC. Cartilage tissue engineering on the surface of a novel gelatin-calcium-phosphate biphasic scaffold in a double-chamber bioreactor. J Biomed Mater Res B Appl Biomater. 2004;71(2):313-321. doi: 10.1002/jbm.b.30090
- Liu G, Wei X, Zhai Y, et al. 3D printed osteochondral scaffolds: design strategies, present applications and future perspectives. Front Bioeng Biotechnol. 2024;12:1339916. doi: 10.3389/fbioe.2024.1339916
- Bai L, Fang C, Qi Y, Wang C, Wang M. Cryogenic 3D printing of heterogeneous scaffolds with capability to spatially tune cellular morphology of mesenchymal stem cells for integrated osteochondral regeneration. Mater Today Bio. 2025;35:102560. doi: 10.1016/j.mtbio.2025.102560
- Nowicki MA, Castro NJ, Plesniak MW, Zhang LG. 3D printing of novel osteochondral scaffolds with graded microstructure. Nanotechnology. 2016;27(41):414001. doi: 10.1088/0957-4484/27/41/414001
- Kotlarz M, O’Keeffe C, S Kronemberger G, et al. Biofabrication and in vivo evaluation of a hybrid osteochondral implant consisting of structurally organised engineered cartilage on a 3D-printed metal scaffold. Biomaterials. 2026;327:123788. doi: 10.1016/j.biomaterials.2025.123788
- Peng Y, Zhuang Y, Liu Y, et al. Bioinspired gradient scaffolds for osteochondral tissue engineering. Exploration. 2023;3(4):20210043. doi: 10.1002/EXP.20210043
- Wang L, Chen X, Wang X, et al. Bio-inspired mineralized collagen scaffolds with precisely controlled gradients for the treatment of severe osteoarthritis in a male rabbit model. Int J Biol Macromol. 2025;300:139843. doi: 10.1016/j.ijbiomac.2025.139843
- Shin J, Kang R, Hyun K, et al. Machine Learning-Enhanced Optimization for High-Throughput Precision in Cellular Droplet Bioprinting. Adv Sci. 2025;12(20):e2412831. doi: 10.1002/advs.202412831
- Yuan B, Guss GM, Wilson AC, et al. Machine-Learning- Based Monitoring of Laser Powder Bed Fusion. Adv Mater Technol. 2018;3(12):1800136. doi: 10.1002/admt.201800136
- Shin J, Lee Y, Li Z, Hu J, Park SS, Kim K. Optimized 3D Bioprinting Technology Based on Machine Learning: A Review of Recent Trends and Advances. Micromachines (Basel). 2022;13(3):363. doi:10.3390/mi13030363
- Jin Z, Zhang Z, Shao X, Gu GX. Monitoring Anomalies in 3D Bioprinting with Deep Neural Networks. ACS Biomater Sci Eng. 2023;9(7):3945-3952. doi: 10.1021/acsbiomaterials.0c0176
- Shi J, Song J, Song B, Lu WF. Multi-Objective Optimization Design through Machine Learning for Drop-on-Demand Bioprinting. Engineering. 2019;5(3):586-593. doi: 10.1016/j.eng.2018.12.009
- Li J, Zhou Z, Yang J, et al. MedShapeNet - a large-scale dataset of 3D medical shapes for computer vision. Biomed Tech. 2024;70(1):71-90. doi: 10.1515/bmt-2024-0396
- Choi E, An K, Kang KT. Deep-Learning-Based Microfluidic Droplet Classification for Multijet Monitoring. ACS Appl Mater Interfaces. 2022;14(13):15576-15586. doi: 10.1021/acsami.1c22048
- Jeong JJ, Koo G. AdaLo: Adaptive learning rate optimizer with loss for classification. Inf Sci. 2025;690:121607. doi: 10.1016/j.ins.2024.121607.
- Chitnis K, Lu Y, Rhoads B, Chakka LRJ, Choudhury S, Maniruzzaman M. Optimization of print parameters for batch and continuous manufacturing of three-dimensional (3D) printed dosage forms using artificial intelligence and machine learning. Drug Deliv and Transl Res. 2025. doi: 10.1007/s13346-025-02006-4
- Jablonka KM, Patiny L, Smit B. Making the collective knowledge of chemistry open and machine actionable. Nat Chem. 2022;14(4):365-376. doi: 10.1038/s41557-022-00910-7
- Li F, Han J, Cao T, et al. Design of self-assembly dipeptide hydrogels and machine learning via their chemical features. Proc Natl Acad Sci USA. 2019;116(23):11259-11264. doi: 10.1073/pnas.1903376116
- Javaid S, Gorji HT, Soulami KB, Kaabouch N. Identification and ranking biomaterials for bone scaffolds using machine learning and PROMETHEE. Res Biomed Eng. 2023;39:129- 138. doi:0.1007/s42600-022-00257-5
- Nadernezhad A, Groll J. Machine Learning Reveals a General Understanding of Printability in Formulations Based on Rheology Additives. Adv Sci. 2022;9(29):e2202638. doi: 10.1002/advs.202202638
- Chen H, Liu Y, Balabani S, Hirayama R, Huang J. Machine Learning in Predicting Printable Biomaterial Formulations for Direct Ink Writing. Research. 2023;6:0197. doi: 10.34133/research.0197
- Yu J, Yao D, Wang L, Xu M. Machine Learning in Predicting and Optimizing Polymer Printability for 3D Bioprinting. Polymers (Basel). 2025;17(13):1873. doi:10.3390/polym17131873
- Mai M, Luo S, Fasciano S, et al. Morphology-based deep learning approach for predicting adipogenic and osteogenic differentiation of human mesenchymal stem cells (hMSCs). Front Cell Dev Biol. 2023;11:1329840. doi: 10.3389/fcell.2023.1329840
- Xu H, Liu Q, Casillas J, et al. Prediction of cell viability in dynamic optical projection stereolithography-based bioprinting using machine learning. J Intell Manuf. 2022;33:995-1005. doi: 10.1007/s10845-020-01708-5
- Zhang C., Elvitigala K.C.M.L., Mubarok W., Okano Y., Sakai S. Machine learning-based prediction and optimisation framework for as-extruded cell viability in extrusion-based 3D bioprinting. Virtual Phys Prototyp. 2024;19:e2400330. doi: 10.1080/17452759.2024.2400330
- Mairpady A, Mourad AI, Mozumder MS. Accelerated Discovery of the Polymer Blends for Cartilage Repair through Data-Mining Tools and Machine-Learning Algorithm. Polymers. 2022;14(9):1802. doi: 10.3390/polym14091802
- Li Z, Song P, Li G, et al. AI energized hydrogel design, optimization and application in biomedicine. Mater Today Bio. 2024;25:101014. doi: 10.1016/j.mtbio.2024.101014
- Junru Zhang, Yang Liu, Durga Chandra Sekhar.P, et al. Rapid, autonomous high-throughput characterization of hydrogel rheological properties via automated sensing and physics-guided machine learning. Appl. Mater. Today. 2023;30:14. doi: 10.1016/j.apmt.2022.101720.
- Seifermann M, Reiser P, Friederich P, Levkin PA. High- Throughput Synthesis and Machine Learning Assisted Design of Photodegradable Hydrogels. Small Methods. 2023;7(9):e2300553. doi: 10.1002/smtd.202300553
- Zhao Y, Li H, Zhou H. et al. A review of graph neural network applications in mechanics-related domains. Artif Intell Rev. 2024;57;315. doi: 10.1007/s10462-024-10931-y
- Zahedi S, Taherkhani A, Baserinia R, Zahedi S, Ali, H, Abdi, M. Comparative Evaluation of Neural Networks and Transfer Learning for Predicting Mechanical Properties of 3D‐Printed Bone Scaffolds. Macromol Mater Eng. 2025; 310. doi: 10.1002/mame.202500073
- Shetty A, Fathima A, Anika B, et al. Computational optimization of 3D printed bone scaffolds using orthogonal array-driven FEA and neural network modeling. Sci Rep. 2025;15(1):30515. doi: 10.1038/s41598-025-15122-5
- Wang, Z., Dabaja, R., Chen, L., & Banu, M. Machine learning unifies flexibility and efficiency of spinodal structure generation for stochastic biomaterial design. Sci Rep. 2023;13. doi: 10.1038/s41598-023-31677-7.
- Ashkan Sedigh, Jacob E. Tulipan, Michael R. Rivlin, Ryan E. Tomlinson. Utilizing Q-Learning to Generate 3D Vascular Networks for Bioprinting Bone. bioRxiv. Preprint online 2020. doi: 10.1101/2020.10.08.331611
- Cadle R, Rogozea D, Moldovan L, Moldovan NI. Design and Implementation of Anatomically Inspired Mesenteric and Intestinal Vascular Patterns for Personalized 3D Bioprinting. Appl Sci. 2022;12(9):4430. doi: 10.3390/app12094430
- Wu J, Zhang Y, Lyu Y, Cheng L. On the Various Numerical Techniques for the Optimization of Bone Scaffold. Materials. 2023;16(3):974. doi: 10.3390/ma16030974
- Drakoulas G, Gortsas T, Polyzos E, Tsinopoulos S, Pyl L, Polyzos D. An explainable machine learning-based probabilistic framework for the design of scaffolds in bone tissue engineering. Biomech Model Mechanobiol. 2024;23(3):987-1012. doi: 10.1007/s10237-024-01817-7
- Naga Ramesh JV, Sonker A, Indumathi G, Balakrishnan D, Nimma D, Karthik J. Bayesian neural networks for probabilistic modeling of thermal dynamics in multiscale tissue engineering scaffolds. J Therm Biol. 2025;130:104134. doi: 10.1016/j.jtherbio.2025.104134
- G Taylor S, Mueller E, Jones LR, Makela AV, Ashammakhi N. Translational Aspects of 3D and 4D Printing and Bioprinting. Adv Healthc Mater. 2024;13(27):e2400463. doi: 10.1002/adhm.202400463
- Seo E, Lee YN, Shin WY, et al. Structural influence on titanium ion dissolution in 3D-printed Ti6Al4V orthopedic implants. Sci Rep. 2025;15(1):37122. doi: 10.1038/s41598-025-21129-9
- Phruekthayanon J, Kühn-Kauffeldt M, Kühn M, et al. Biofunctionalization of 3D printed PEEK using integrated cathodic arc plasma coating: a one-step solution to antimicrobial and bioactive PEEK Implant. J Mater Sci Mater Med. 2025;36(1):109. doi: 10.1007/s10856-025-06971-7
- Grijalva Garces D, Strauß S, Gretzinger S, et al. On the reproducibility of extrusion-based bioprinting: round robin study on standardization in the field. Biofabrication. 2023;16(1):10.1088/1758-5090/acfe3b. doi: 10.1088/1758-5090/acfe3b
- Eraliev O, Lee K-H, Lee C-H. Self-Loosening of a 3D-Printed Bolt by Using Three Different Materials under Cyclical Temperature Changes. Appl Sci. 2022; 12(6):3001. doi: 10.3390/app12063001
- Mandal A, Clegg JR, Anselmo AC, Mitragotri S. Hydrogels in the clinic. Bioeng Transl Med. 2020;5(2):e10158.doi: 10.1002/btm2.10158
- Becerra AG, Gutiérrez M, Lahoz-Beltra R. Computing within bacteria: Programming of bacterial behavior by means of a plasmid encoding a perceptron neural network. Biosystems. 2022;213:104608. doi: 10.1016/j.biosystems.2022.104608
- Sujeeun LY, Phul IC, Goonoo N, Kotov NA, Bhaw-Luximon A. Predicting inflammatory response of biomimetic nanofibre scaffolds for tissue regeneration using machine learning and graph theory. J Mater Chem B. 2025;13(10):3304-3318. doi: 10.1039/d4tb02494j
- El Arab RA, Al Moosa OA, Sagbakken M. Economic, ethical, and regulatory dimensions of artificial intelligence in healthcare: an integrative review. Front Public Health. 2025;13:1617138. doi: 10.3389/fpubh.2025.1617138
212. Silva Robazzi JV, Derman ID, Gupta D, et al. The Synergy of Artificial Intelligence and 3D Bioprinting: Unlocking New Frontiers in Precision and Tissue Fabrication. Adv Funct Mater. 2025. doi: 10.1002/adfm.202509530

