Superparamagnetic iron oxide nanoparticle-based nanosystems for cancer theranostics

The advancement of nanotechnology in the biomedical field has garnered growing interest due to the diverse physical properties of nanoparticles (NPs) and their potential applications in cancer theranostics. Although various nanodrug delivery systems, such as polymeric, lipid-based, and gold NPs, are available, superparamagnetic iron oxide NPs (SPIONs) have attracted significant attention due to their multifunctionality, biocompatibility, ease of surface functionalization, and responsiveness to external magnetic fields, enabling both cancer diagnostics and drug delivery applications. This review provides an overview of SPIONs as a powerful theranostic tool in cancer treatment. It also focuses on the synthesis and stabilization of SPIONs, discussing various protection and stabilization strategies using both organic coatings (synthetic and natural polymers) and inorganic coatings (such as silica and gold), as well as liposomes. In addition, the review highlights targeted biomedical applications of SPIONs, including magnetic resonance imaging, hyperthermia, and drug delivery. Finally, it also addresses the challenges and limitations of SPIONs, as well as their future perspective in clinical translation in cancer theranostics.
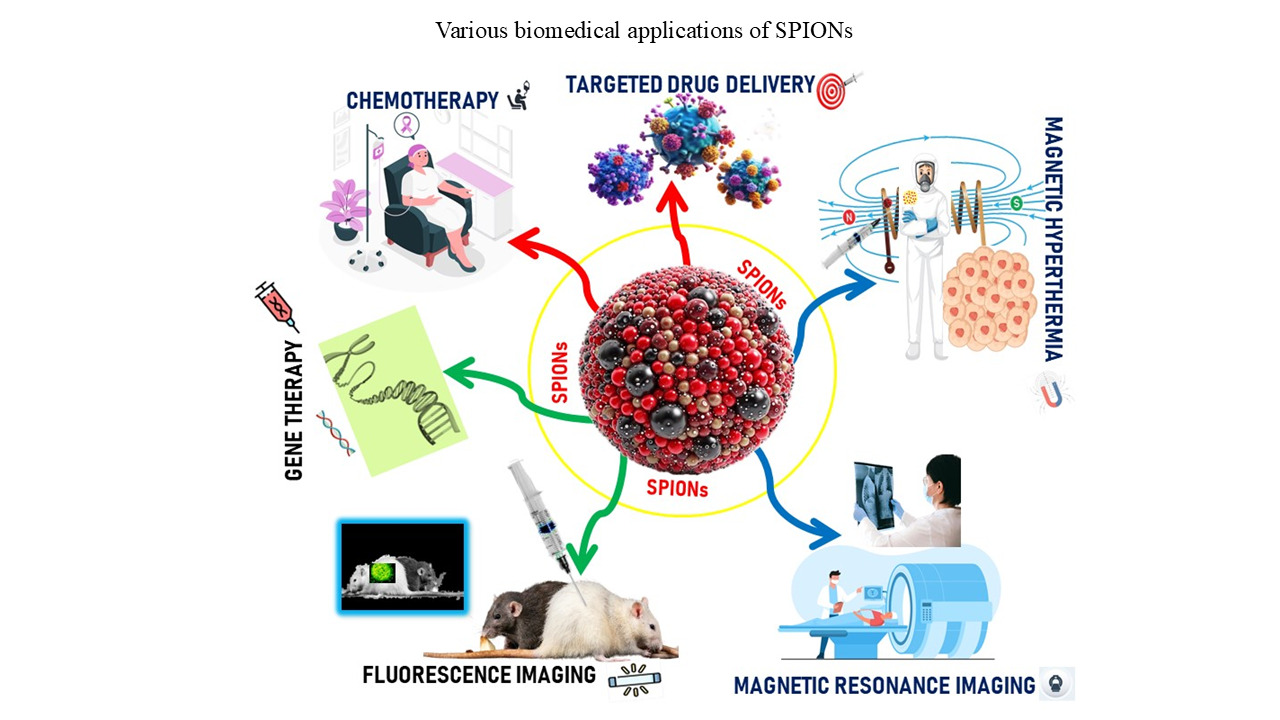
- Lee N, Yoo D, Ling D, Cho MH, Hyeon T, Cheon J. Iron oxide based nanoparticles for multimodal imaging and magneto responsive therapy. Chem Rev. 2015;115:10637-10638. doi: 10.1021/acs.chemrev.5b00112.
- Gao J, Gu H, Xu B. Multifunctional magnetic nanoparticles: Design, synthesis, and biomedical applications. Acc Chem Res. 2009;42(8):1097-1107. doi: 10.1021/ar9000026
- Panda J, Ghouri K, Sarkar R, Tudu B. Fabrication and characterization of self-assembled zinc ferrite nanospheres for biomedical applications. Appl Phys A. 2022;128(4):1-9. doi: 10.1007/s00339-022-05456-6
- Hao R, Xing R, Xu Z, Hou Y, Gao S, Sun S. Synthesis, functionalization, and biomedical applications of multifunctional magnetic nanoparticles. Adv Mater. 2010;22:2729-2742. doi: 10.1002/adma.201000260
- Poller JM, Zaloga J, Schreiber E, et al. Selection of potential iron oxide nanoparticles for breast cancer treatment based on in vitro cytotoxicity and cellular uptake. Int J Nanomedicine. 2017;12:3207-3220. doi: 10.2147/IJN.S132369
- Panda J, Satapathy BS, Sarkar R, Tudu B. A zinc ferrite nanodrug carrier for delivery of docetaxel: Synthesis, characterization, and in vitro tests on C6 glioma cells. J Microencapsul. 2022;39(2):136-144. doi: 10.1080/02652048.2022.2053757
- Hu Y, Mignani S, Majoral JP, Shen M, Shi X. Construction of iron oxide nanoparticle-based hybrid platforms for tumor imaging and therapy. Chem Soc Rev. 2018;47:1874-1900. doi: 10.1039/c7cs00657h
- Bray F, Laversanne M, Sung H, et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74(3):229-263. doi: 10.3322/caac.21834
- Mitchell MJ, Billingsley MM, Haley RM, Wechsler ME, Peppas NA, Langer R. Engineering precision nanoparticles for drug delivery. Nat Rev Drug Discov. 2021;20:101-124. doi: 10.1038/s41573-020-0090-8
- Caster JM, Patel AN, Zhang T, Wang A. Investigational nanomedicines in 2016: A review of nanotherapeutics currently undergoing clinical trials. Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2017;9:1416-1425. doi: 10.1002/wnan.1416
- Panda J, Satapathay BS, Mishra A, Biswal B, Sahoo PK. Potential of Ferrite-Based Nanoparticles for Improved Cancer Therapy: Recent Progress and Challenges Ahead. Applications of Ferrites. London: Intech Open; 2023. p. 1-18. doi: 10.5772/intechopen.1002346
- Cui Y, Zhang M, Zeng F, Jin H, Xu Q, Huang Y. Dual-targeting magnetic PLGA nanoparticles for codelivery of paclitaxel and curcumin for brain tumor therapy. ACS Appl Mater Interfaces. 2016;8:32159-32169. doi: 10.1021/acsami.6b10175
- Shabatina TI, Vernya OI, Shabatin VP, Melnikov MY. Magnetic nanoparticles for biomedical purposes: Modern trends and prospects. Magnetochemistry. 2020;6:30. doi: 10.3390/magnetochemistry6030030
- Panda J, Satapathy BS, Majumder S, Sarkar R, Mukherjee B, Tudu B. Engineered polymeric iron oxide nanoparticles as potential drug carrier for targeted delivery of docetaxel to breast cancer cells. J Magn Magn Mater. 2019;485:165-173. doi: 10.1016/j.jmmm.2019.04.058
- Fatima H, Charinpanitkul T, Kim KS. Fundamentals to apply magnetic nanoparticles for hyperthermia therapy. Nanomaterials (Basel). 2021;11:1203. doi: 10.3390/nano11051203
- Demirer GS, Okur AC, Kizilel S. Synthesis and design of biologically inspired biocompatible iron oxide nanoparticles for biomedical application. J Mater Chem B. 2015;3:7831-7849. doi: 10.1039/c5tb00931f
- Lu AH, Salabas EL, Schuth F. Magnetic nanoparticles: Synthesis, protection, functionalization, and application. Angew Chem Int Ed. 2007;46:1222-1244. doi: 10.1002/anie.200602866
- Massart R. Preparation of aqueous magnetic liquids in alkaline and acidic media. IEEE Trans Magn. 1981;17:1247-1248. doi: 10.1109/TMAG.1981.1061188
- Wang X, Zhuang J, Peng Q, Li Y. A general strategy for nanocrystal synthesis. Nature. 2005;437:121-124. doi: 10.1038/nature03968
- Yang H, Ogawa T, Hasegawa D, Takahashi M. Synthesis and magnetic properties of monodisperse magnetite nanocubes. J Appl Phys. 2008;103:07D526. doi: 10.1063/1.2833820
- Dolores R, Raquel S, Adianex GL. Sonochemical synthesis of iron oxide nanoparticles loaded with folate and cisplatin: Effect of ultrasonic frequency. Ultrason Sonochem. 2014;23:39-398. doi: 10.1016/j.ultsonch.2014.08.005
- Gambhir RP, Rohiwal SS, Tiwari AP. Multifunctional surface functionalized magnetic iron oxide nanoparticles for biomedical applications: A review. Appl Surf Sci Adv. 2022;11:100303. doi: 10.1016/j.apsadv.2022.100303
- Laurent S, Forge D, Port M, et al. Magnetic iron oxide nanoparticles: Synthesis, stabilization, vectorization, physicochemical characterizations, and biological applications. Chem Rev. 2008;108:2064-2110. doi: 10.1021/cr068445e
- Sairam M, Naidu BV, Nataraj SK, Sreedhar B, Aminabhavi TM. Poly(vinyl alcohol)-iron oxide nanocomposite membranes for pervaporation dehydration of isopropanol, 1,4-dioxane and tetrahydrofuran. J Membr Sci. 2006;283:65-73. doi: 10.1016/j.memsci.2006.06.013
- McCarthy JR, Weissleder R. Multifunctional magnetic nanoparticles for targeted imaging and therapy. Adv Drug Deliv Rev. 2008;60(11):1241-1251. doi: 10.1016/j.addr.2008.03.014
- Belessi V, Zboril R, Tucek J, Mashlan M, Tzitzios VK, Petridis D. Ferrofluids from magnetic-chitosan hybrids. Chem Mater. 2008;20:3298-3305. doi: 10.1021/cm702990t
- Wu W, He Q, Jiang C. Magnetic iron oxide nanoparticles: Synthesis and surface functionalization strategies. Nanoscale Res Lett. 2008;3:397-415. doi: 10.1007/s11671-008-9174-9
- Bruce IJ, Sen T. Surface modification of magnetic nanoparticles with alkoxysilanes and their application in magnetic bio separation. Langmuir. 2005;21:7029-7035. doi: 10.1021/la050553t
- Ding HL, Zhang YX, Wang S, Xu JM, Xu SC, Li GH. Fe3 O4 @SiO2 Core/Shell nanoparticles: The silica coating regulations with a single core for different core sizes and shell thicknesses. Chem Mater. 2012;24:4572-4580. doi: 10.1021/cm302828d
- Lin J, Zhou WL, Kumbhar A, et al. Gold-coated iron (Fe@Au) nanoparticles: Synthesis, characterization, and magnetic field-induced self-assembly. J Solid State Chem. 2001;159:26-31. doi: 10.1006/jssc.2001.9117
- Yu H, Chen M, Rice PM, Wang SX, White RL, Sun S. Dumbbell-like bi-functional Au-Fe3O4 nanoparticles. Nano Lett. 2005;5:379-382. doi: 10.1021/nl047955q
- Wang L, Luo J, Maye MM, et al. Iron oxide-gold core-shell nanoparticles and thin film assembly. J Mater Chem. 2005;15:1821-1832. doi: 10.1039/B501375E
- Lapusan R, Borlan R, Focsan M. Advancing MRI with magnetic nanoparticles: A comprehensive review of translational research and clinical trials. Nanoscale Adv. 2024;6:2234-2259. doi: 10.1039/d3na01064c
- Liao Z, Wang H, Lv R, et al. Polymeric liposomes-coated superparamagnetic iron oxide nanoparticles as contrast agent for targeted magnetic resonance imaging of cancer cells. Langmuir. 2011;27:3100-3105. doi: 10.1021/la1050157
- Li J, Hu Y, Yang J, et al. Hyaluronic acid-modified Fe3O4@ Au core/shell nanostars for multimodal imaging and photothermal therapy of tumors. Biomaterials. 2015;38:10-21. doi: 10.1016/j.biomaterials.2014.10.065
- Jun YW, Huh YM, Choi YS, et al. Nanoscale size effect of magnetic nanocrystals and their utilization for cancer diagnosis via magnetic resonance imaging. J Am Chem Soc. 2005;127:12387-12391. doi: 10.1021/ja0422155
- Pellico J, Cabello JR, Herranz F. Radiolabeled iron oxide nanomaterials for multimodal nuclear imaging and positive contrast magnetic resonance imaging (MRI): A review. ACS Appl Nano Mater. 2023;6:20523-20538. doi: 10.1021/acsanm.3c04269
- Hundt W, Petsch R, Helmberger T, Reiser M. Signal changes in liver and spleen after endorem administration in patients with and without liver cirrhosis. Eur Radiol. 2000;10:409-416. doi: 10.1007/s003300050067
- Reimer P, Balzer T. Ferucarbotran (resovist): A new clinically approved RES-specific contrast agent for contrast enhanced MRI of the liver: Properties, clinical development, and applications. Eur Radiol. 2003;13:1266-1276. doi: 10.1007/s00330-002-1721-7
- Fortuin AS, Brüggemann R, van der Linden J, et al. Ultra-small superparamagnetic iron oxides for metastatic lymph node detection: Back on the block. Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2018;10:e147. doi: 10.1002/wnan.1471
- Nguyen KL, Yoshida T, Kathuria-Prakash N, et al. Multicenter safety and practice for off-label diagnostic use of ferumoxytol in MRI. Radiology. 2019;293:554-564. doi: 10.1148/radiol.2019190477
- Ding L, Wang R, Hu Y, et al. Folic acid-modified Laponite®- stabilized Fe3O4 nanoparticles for targeted T2-weighted MR imaging of tumour. Appl Clay Sci. 2020;186:105447. doi: 10.1016/j.clay.2020.105447
- Zhao Z, Zhou Z, Bao J, et al. Octapod iron oxide nanoparticles as high-performance T contrast agents for magnetic resonance imaging. Nat Commun. 2013;4:2266. doi: 10.1038/ncomms3266
- Zhu J, Peng C, Sun W, et al. Formation of iron oxide nanoparticle-loaded γ-polyglutamic acid nanogels for MR imaging of tumors. J Mater Chem B. 2015;3:8684-8693. doi: 10.1039/C5TB01854D
- Li J, Zheng L, Cai H, et al. Polyethyleneimine-mediated synthesis of folic acid-targeted iron oxide nanoparticles for in vivo tumor MR imaging. Biomaterials. 2013;4:8382-8392. doi: 10.1016/j.biomaterials.2013.07.070
- Coey JMD. Noncollinear spin arrangement in ultrafine ferrimagnetic crystallites. Phys Rev Lett. 1971;27:1140-1142. doi: 10.1103/PhysRevLett.27.1140
- Luo Y, Yang J, Yan Y, et al. RGD-functionalized ultrasmall iron oxide nanoparticles for targeted T1-weighted MR imaging of gliomas. Nanoscale. 2015;7:14538-14546. doi: 10.1039/C5NR04003E
- Bhavesh R, Lechuga-Vieco AV, Ruiz-Cabello J, Herranz F. T1-MRI fluorescent iron oxide nanoparticles by microwave assisted synthesis. Nanomaterials. 2015;5:1888-1890. doi: 10.3390/nano5041880
- Wei H, Bruns OT, Kaul MG, et al. Exceedingly small iron oxide nanoparticles as positive MRI contrast agents. Proc Natl Acad Sci U S A. 2017;114:2325-2330. doi: 10.1073/pnas.1620145114
- Zhou Z, Zhao Z, Zhang H, Wang Z, Chen X, Wang R, et al. Interplay between longitudinal and transverse contrasts in Fe3O4 nanoplates with (111) exposed surfaces. ACS Nano. 2013;7:3287-3296. doi: 10.1021/nn5038652
- Taboada E, Rodríguez R, Roig A, Oró J, Roch A, Muller RN. Relaxometric and magnetic characterization of ultrasmall iron oxide nanoparticles with high magnetization. Evaluation as potential T1 magnetic resonance imaging contrast agents for molecular imaging. Langmuir. 2007;23:4583-4588. doi: 10.1021/la063415s
- Sun Y, Zheng Y, Ran H, et al. Superparamagnetic PLGA-iron oxide microcapsules for dual-modality US/MR imaging and high intensity focused US breast cancer ablation. Biomaterials. 2012;33:5854-5864. doi: 10.1016/j.biomaterials.2012.04.062
- Bae KH, Kim YB, Lee Y, et al. Bioinspired synthesis and characterization of gadolinium-labeled magnetite nanoparticles for dual contrast t1- and T2-weighted magnetic resonance imaging. Bioconjug Chem. 2010;21:505-512. doi: 10.1021/bc900424u
- Johannsen M, Gneveckow U, Eckelt L, et al. Clinical hyperthermia of prostate cancer using magnetic nanoparticles: Presentation of a new interstitial technique. Eur Urol. 2007;52:1653-1662. doi: 10.1080/02656730500158360
- Guardia P, Di Corato R, Lartigue L, et al. Water-soluble iron oxide nanocubes with high values of specific absorption rate for cancer cell hyperthermia treatment. ACS Nano. 2012;6:3080-3091. doi: 10.1021/nn2048137
- Chastellain M, Petri A, Gupta A, Rao KV, Hofmann H. Superparamagnetic silica-iron oxide nanocomposites for application in hyperthermia. Adv Eng Mater. 2004;6:235-241. doi: 10.1002/adem.200300574
- Johannsen M, Thiesen B, Jordan A, Wust P. Magnetic nanoparticle hyperthermia for prostate cancer. Int J Hyperthermia. 2010;26:790-795. doi: 10.3109/02656731003745740
- Gordon AC, Lewandowski RJ, Salem R, Day DE, Omary RA, Larson AC. Localized hyperthermia with iron oxide-doped yttrium microparticles: Steps towards image-guided thermoradiotherapy in liver cancer. J Vasc Interv Radiol. 2014;25:397-404. doi: 10.1016/j.jvir.2013.10.022
- Ghosh R, Pradhan L, Devi YP, et al. Induction heating studies of Fe3O4 magnetic nanoparticles capped with oleic acid and polyethylene glycol for hyperthermia. J Mater Chem. 2011;21:13388-13398. doi: 10.1039/C1JM10092K
- Hayashi K, Ono K, Suzuki H, et al. One-pot biofunctionalization of magnetic nanoparticles via thiolene click reaction for magnetic hyperthermia and magnetic resonance imaging. Chem Mater. 2010;22:3768-3772. doi: 10.1021/cm100810g
- Espinosa A, Di Corato R, Kolosnjaj-Tabi J, Flaud P, Pellegrino T, Wilhelm C. The duality of iron oxide nanoparticles in cancer therapy: Amplification of heating efficiency by magnetic hyperthermia and photothermal bimodal treatment. ACS Nano. 2016;10:2436-2446. doi: 10.1021/acsnano.5b07249
- Corato D, Béalle G, Kolosnjaj-Tabi J, et al. Combining magnetic hyperthermia and photodynamic therapy for tumor ablation with photoresponsive magnetic liposomes. ACS Nano. 2015;9:2904-2916. doi: 10.1021/nn506949t
- Fortin JP, Wilhelm C, Servais J, Ménager C, Bacri JC, Gazeau F. Size-sorted anionic iron oxide nanomagnets as colloidal mediators for magnetic hyperthermia. J Am Chem Soc. 2007;129:2628-2635. doi: 10.1021/ja067457e
- Landeghem V, Maier-Hauff FK, Jordan K, et al. Post-mortem studies in glioblastoma patients treated with thermotherapy using magnetic nanoparticles. J Neurooncology. 2007;81:53-60. doi: 10.1016/j.biomaterials.2008.09.044
- Kandasamy G, Maity D. Recent advances in superparamagnetic iron oxide nanoparticles (SPIONs) for in vitro and in vivo cancer nanotheranostics. Int J Pharm. 2015;496:191-218. doi: 10.1016/j.ijpharm.2015.10.058
- Widder K, Senyei AE, Scarpelli DG. Magnetic microspheres: A model system of site specific drug delivery in vivo. Proc Soc Exp Biol Med. 1978;158:141-146. doi: 10.3181/00379727-158-40158
- Senyei AE, Widder K, Czerlinski G. Magnetic guidance of drug‐carrying microspheres. J Appl Phys. 1978;49:3578-3583. doi: 10.1063/1.325219
- Price PM, Mahmoud WE, Al-Ghamdi AA, Bronstein LM. Magnetic drug delivery: Where the field is going. Front Chem. 2018;6:619-626. doi: 10.3389/fchem.2018.00619
- Bhuyan T, Singh AK, Dutta D, Unal A, Ghosh SS, Bandyopadhyay D. Magnetic field guided chemotaxis of Mushbots for targeted anticancer therapeutics. ACS Biomater Sci Eng. 2017;3:1627-1640. doi: 10.1021/acsbiomaterials.7b00086
- Mai BT, Fernandes S, Balakrishnan PB, Pellegrino T. Nanosystems based on magnetic nanoparticles and thermoorph responsive polymers: An update and future perspectives. Acc Chem Res. 2018;51:999-1013. doi: 10.1021/acs.accounts.7b00549
- Chen Z, Kankala RK, Long L, Xie S, Chen A, Zou L. Current understanding of passive and active targeting nanomedicines to enhance tumor accumulation. Coord Chem Rev. 2023;481:215051. doi: 10.1016/j.ccr.2023.215051
- Wahajuddin, Arora S. Superparamagnetic iron oxide nanoparticles: Magnetic nanoplatforms as drug carriers. Int J Nanomed. 2012;7:3445-3471. doi: 10.2147/IJN.S30320
- Alexiou C, Arnold W, Klein RJ, et al. Locoregional cancer treatment with magnetic drug targeting. Cancer Res. 2000;60:6641-6648.
- Kim J, Lee JE, Lee JH, et al. Designed fabrication of a multifunctional polymer nanomedical platform for simultaneous cancer- targeted imaging and magnetically guided drug delivery. Adv Mater. 2008;20:478-483. doi: 10.1002/adma.200701726
- Basuki JS, Duong HTT, Macmillan A, et al. Using fluorescence lifetime imaging microscopy to monitor theranostic nanoparticle uptake and intracellular doxorubicin release. ACS Nano. 2013;7:10175-10189. doi: 10.1021/nn404407g
- Ling Y, Wei K, Luo Y, Gao X, Zhong S. Dual docetaxel/ superparamagnetic iron oxide loaded nanoparticles for both targeting magnetic resonance imaging and cancer therapy. Biomaterials. 2011;32:7139-7150. doi: 10.1016/j.biomaterials.2011.05.089
- Reczyńska K, Marszałek M, Zarzycki A, et al. Superparamagnetic iron oxide nanoparticles modified with silica layers as potential agents for lung cancer treatment. Nanomaterials. 2020;10:1076. doi: 10.3390/nano10061076
- Horvat NK, Chocarro S, Marques O, et al. Superparamagnetic iron oxide nanoparticles reprogram the tumor microenvironment and reduce lung cancer regrowth after crizotinib treatment. ACS Nano. 2024;18:11025-11041. doi: 10.1021/acsnano.3c08335
- Wang ZL, Qiao RR, Tang NG, et al. Active targeting theranostic iron oxide nanoparticles for MRI and magnetic resonance-guided focused ultrasound ablation of lung cancer. Biomaterials. 2017;127:25-35. doi: 10.1016/j.biomaterials.2017.02.037
- Patra JK, Das G, Fraceto LF, et al. Nano based drug delivery systems: Recent developments and future prospects. J Nanobiotechnol. 2018;16(71):1-33. doi: 10.1186/s12951-018-0392-8
- Available from: https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-datopotamab-deruxtecan-dlnk-unresectable-or-metastatic-hr-positive-her2-negative-breast cancer [Last accessed on 2025 Jan 17].

