Minds and hearts: Cardio-neural crosstalk in cardiovascular and neurodegenerative diseases

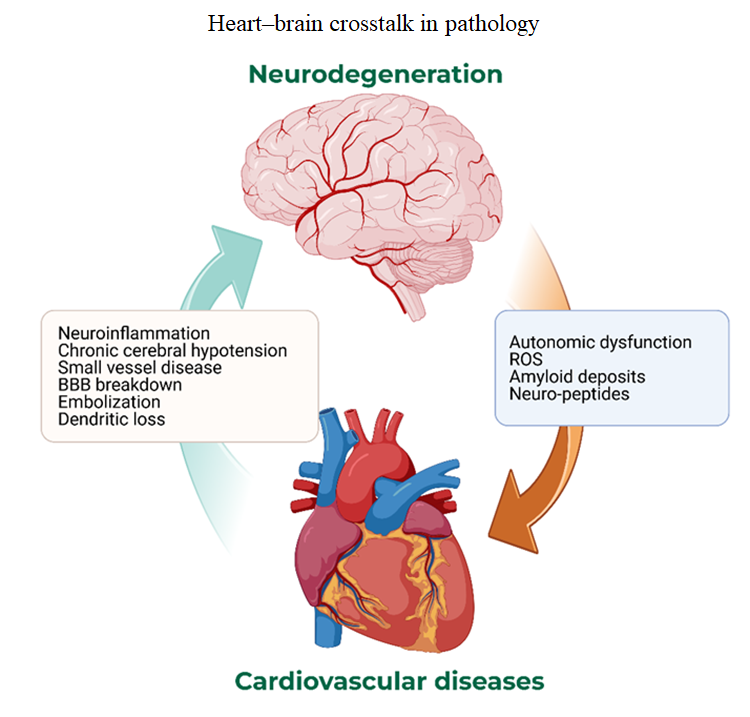
With increasing life expectancy, the prevalence of age-related cardiovascular diseases—including hypertension, atherosclerosis, atrial fibrillation, and heart failure—and neurodegenerative disorders such as Alzheimer’s disease and Parkinson’s disease (PD) continues to rise. Emerging evidence indicates that the heart and brain are linked through complex, bidirectional pathways in which cardiovascular pathology can accelerate neurodegeneration, while neurodegenerative processes can precipitate or exacerbate cardiovascular dysfunction. This review critically appraises current knowledge on cardio-neural crosstalk, distinguishing well-established associations (e.g., hypertension and dementia) from more speculative mechanisms (e.g., receptor changes in ischemic PD). We integrate epidemiological findings with mechanistic insights, highlighting inflammation, hypoperfusion, blood–brain barrier disruption, autonomic dysfunction, and amyloid biology as convergent pathways. To enhance clarity, we summarize evidence strength in expanded tables and figures, including epidemiological hazard ratios, mechanistic pathways, and confidence grading. The main contribution of this review is to provide a structured synthesis of robust and emerging evidence on the bidirectional heart–brain axis, identifying gaps that limit causal inference. We emphasize future directions such as longitudinal biomarker studies, interventional trials repurposing cardiovascular therapies for neuroprotection, and systems biology approaches to integrate cardiovascular and neurodegenerative omics data. By consolidating current evidence and outlining actionable research priorities, this review aims to inform both clinical practice and translational research in cardio-neural disease.
- Justin BN, Turek M, Hakim AM. Heart disease as a risk factor for dementia. Clin Epidemiol. 2013;5:135-145. doi: 10.2147/CLEP.S30621
- Muqtadar H, Testai FD, Gorelick PB. The dementia of cardiac disease. Curr Cardiol Rep. 2012;14(6):732-740. doi: 10.1007/s11886-012-0304-8
- De Roos A, Van Der Grond J, Mitchell G, Westenberg J. Magnetic resonance imaging of cardiovascular function and the brain: Is dementia a cardiovascular-driven disease? Circulation. 2017;135(22):2178-2195. doi: 10.1161/CIRCULATIONAHA.116.021978
- Launer LJ, Masaki K, Petrovitch H, Foley D, Havlik RJ. The association between midlife blood pressure levels and late-life cognitive function. The Honolulu-Asia aging study. JAMA. 1995;274(23):1846-1851. doi: 10.1001/jama.274.23.1846
- Lénárt N, Brough D, Dénes Á. Inflammasomes link vascular disease with neuroinflammation and brain disorders. J Cereb Blood Flow Metab. 2016;36(10):1668-1685. doi: 10.1177/0271678X16662043
- Weber MD, Frank MG, Tracey KJ, Watkins LR, Maier SF. Stress induces the danger-associated molecular pattern HMGB-1 in the hippocampus of male sprague dawley rats: A priming stimulus of microglia and the NLRP3 inflammasome. J Neurosci. 2015;35(1):316-324. doi: 10.1523/JNEUROSCI.3561-14.2015
- Kaushal V, Dye R, Pakavathkumar P, et al. Neuronal NLRP1 inflammasome activation of Caspase-1 coordinately regulates inflammatory interleukin-1-beta production and axonal degeneration-associated Caspase-6 activation. Cell Death Differ. 2015;22(10):1676-1686. doi: 10.1038/cdd.2015.16
- Minkiewicz J, De Rivero Vaccari JP, Keane RW. Human astrocytes express a novel NLRP2 inflammasome. Glia. 2013;61(7):1113-1121. doi: 10.1002/glia.22499
- Nagyőszi P, Nyúl‐Tóth Á, Fazakas C, et al. Regulation of NOD‐like receptors and inflammasome activation in cerebral endothelial cells. J Neurochem. 2015;135(3):551-564. doi: 10.1111/jnc.13197
- Chen Z, Wen L, Martin M, et al. Oxidative stress activates endothelial innate immunity via sterol regulatory element binding protein 2 (SREBP2) transactivation of microRNA- 92a. Circulation. 2015;131(9):805-814. doi: 10.1161/CIRCULATIONAHA.114.013675
- Kunnas T, Määttä K, Nikkari ST. NLR family pyrin domain containing 3 (NLRP3) inflammasome gene polymorphism rs7512998 (C>T) predicts aging-related increase of blood pressure, the TAMRISK study. Immun Ageing A. 2015;12:19. doi: 10.1186/s12979-015-0047-7
- Schneider S, Koch W, Hoppmann P, et al. Association of Toll-like receptor 4 polymorphism with age-dependent systolic blood pressure increase in patients with coronary artery disease. Immun Ageing. 2015;12(1):4. doi: 10.1186/s12979-015-0031-2
- Martin‐Rodriguez S, Caballo C, Gutierrez G, et al. TLR4 and NALP3 inflammasome in the development of endothelial dysfunction in uraemia. Eur J Clin Invest. 2015;45(2):160-169. doi: 10.1111/eci.12392
- Heneka MT, Carson MJ, Khoury JE, et al. Neuroinflammation in Alzheimer’s disease. Lancet Neurol. 2015;14(4):388-405. doi: 10.1016/S1474-4422(15)70016-5
- Heneka MT, Kummer MP, Stutz A, et al. NLRP3 is activated in Alzheimer’s disease and contributes to pathology in APP/ PS1 mice. Nature. 2013;493(7434):674-678. doi: 10.1038/nature11729
- Lu M, Sun XL, Qiao C, Liu Y, Ding JH, Hu G. Uncoupling protein 2 deficiency aggravates astrocytic endoplasmic reticulum stress and nod-like receptor protein 3 inflammasome activation. Neurobiol Aging. 2014;35(2):421-430. doi: 10.1016/j.neurobiolaging.2013.08.015
- Iturria-Medina Y, Sotero RC, Toussaint PJ, et al. Early role of vascular dysregulation on late-onset Alzheimer’s disease based on multifactorial data-driven analysis. Nat Commun. 2016;7(1):11934. doi: 10.1038/ncomms11934
- Johann S, Heitzer M, Kanagaratnam M, et al. NLRP3 inflammasome is expressed by astrocytes in the SOD1 mouse model of ALS and in human sporadic ALS patients. Glia. 2015;63(12):2260-2273. doi: 10.1002/glia.22891
- Liu L, Chan C. IPAF inflammasome is involved in interleukin-1β production from astrocytes, induced by palmitate; Implications for Alzheimer’s Disease. Neurobiol Aging. 2014;35(2):309-321. doi: 10.1016/j.neurobiolaging.2013.08.016
- Halle A, Hornung V, Petzold GC, et al. The NALP3 inflammasome is involved in the innate immune response to amyloid-β. Nat Immunol. 2008;9(8):857-865. doi: 10.1038/ni.1636
- Huang MY, Tu CE, Wang SC, et al. Corylin inhibits LPS-induced inflammatory response and attenuates the activation of NLRP3 inflammasome in microglia. BMC Complement Altern Med. 2018;18(1):221. doi: 10.1186/s12906-018-2287-5
- Duewell P, Kono H, Rayner KJ, et al. NLRP3 inflammasomes are required for atherogenesis and activated by cholesterol crystals. Nature. 2010;464(7293):1357-1361. doi: 10.1038/nature08938
- Brodacki B, Staszewski J, Toczyłowska B, et al. Serum interleukin (IL-2, IL-10, IL-6, IL-4), TNFalpha, and INFgamma concentrations are elevated in patients with atypical and idiopathic parkinsonism. Neurosci Lett. 2008;441(2):158-162. doi: 10.1016/j.neulet.2008.06.040
- Blum-Degen D, Müller T, Kuhn W, Gerlach M, Przuntek H, Riederer P. Interleukin-1 beta and interleukin-6 are elevated in the cerebrospinal fluid of Alzheimer’s and de novo Parkinson’s disease patients. Neurosci Lett. 1995;202(1-2):17-20. doi: 10.1016/0304-3940(95)12192-7
- Codolo G, Plotegher N, Pozzobon T, et al. Triggering of inflammasome by aggregated α-synuclein, an inflammatory response in synucleinopathies. PloS One. 2013;8(1):e55375. doi: 10.1371/journal.pone.0055375
- Youm YH, Grant RW, McCabe LR, et al. Canonical Nlrp3 inflammasome links systemic low-grade inflammation to functional decline in aging. Cell Metab. 2013;18(4):519-532. doi: 10.1016/j.cmet.2013.09.010
- Maltez VI, Miao EA. Reassessing the evolutionary importance of inflammasomes. J Immunol. 2016;196(3):956-962. doi: 10.4049/jimmunol.1502060
- Duncombe J, Kitamura A, Hase Y, Ihara M, Kalaria RN, Horsburgh K. Chronic cerebral hypoperfusion: A key mechanism leading to vascular cognitive impairment and dementia. Closing the translational gap between rodent models and human vascular cognitive impairment and dementia. Clin Sci (Lond). 2017;131(19):2451-2468. doi: 10.1042/CS20160727
- Pantoni L. Cerebral small vessel disease: From pathogenesis and clinical characteristics to therapeutic challenges. Lancet Neurol. 2010;9(7):689-701. doi: 10.1016/S1474-4422(10)70104-6
- Farkas E, Luiten PGM, Bari F. Permanent, bilateral common carotid artery occlusion in the rat: A model for chronic cerebral hypoperfusion-related neurodegenerative diseases. Brain Res Rev. 2007;54(1):162-180. doi: 10.1016/j.brainresrev.2007.01.003
- Shibata M, Ohtani R, Ihara M, Tomimoto H. White matter lesions and glial activation in a novel mouse model of chronic cerebral hypoperfusion. Stroke. 2004;35(11):2598-2603. doi: 10.1161/01.STR.0000143725.19053.60
- Kitamura A, Fujita Y, Oishi N, et al. Selective white matter abnormalities in a novel rat model of vascular dementia. Neurobiol Aging. 2012;33(5):1012.e25-1012.e35. doi: 10.1016/j.neurobiolaging.2011.10.033
- Poggesi A, Pasi M, Pescini F, Pantoni L, Inzitari D. Circulating biologic markers of endothelial dysfunction in cerebral small vessel disease: A review. J Cereb Blood Flow Metab. 2016;36(1):72-94. doi: 10.1038/jcbfm.2015.116
- Di Marco LY, Venneri A, Farkas E, Evans PC, Marzo A, Frangi AF. Vascular dysfunction in the pathogenesis of Alzheimer’s disease--a review of endothelium-mediated mechanisms and ensuing vicious circles. Neurobiol Dis. 2015;82:593-606. doi: 10.1016/j.nbd.2015.08.014
- Skoog I, Kalaria RN, Breteler MMB. Vascular factors and Alzheimer disease. Alzheimer Dis Assoc Disord. 1999;13(Supplement 3):S106-S114. doi: 10.1097/00002093-199912003-00016
- Reimer MM, McQueen J, Searcy L, et al. Rapid disruption of axon-glial integrity in response to mild cerebral hypoperfusion. J Neurosci. 2011;31(49):18185-18194. doi: 10.1523/JNEUROSCI.4936-11.2011
- Simpson JE, Ince PG, Higham CE, et al. Microglial activation in white matter lesions and nonlesional white matter of ageing brains. Neuropathol Appl Neurobiol. 2007;33(6):670-683. doi: 10.1111/j.1365-2990.2007.00890.x
- Manso Y, Holland PR, Kitamura A, et al. Minocycline reduces microgliosis and improves subcortical white matter function in a model of cerebral vascular disease. Glia. 2018;66(1):34-46. doi: 10.1002/glia.23190
- Sloane JA, Hollander W, Moss MB, Rosene DL, Abraham CR. Increased microglial activation and protein nitration in white matter of the aging monkey. Neurobiol Aging. 1999;20(4):395-405. doi: 10.1016/S0197-4580(99)00066-4
- Freeman LR, Keller JN. Oxidative stress and cerebral endothelial cells: Regulation of the blood-brain-barrier and antioxidant based interventions. Biochim Biophys Acta. 2012;1822(5):822-829. doi: 10.1016/j.bbadis.2011.12.009
- Chrissobolis S, Miller AA, Drummond GR, Kemp- Harper BK, Sobey CG. Oxidative stress and endothelial dysfunction in cerebrovascular disease. Front Biosci. 2011;16(1):1733-1745. doi: 10.2741/3816
- Chandler S, Coates R, Gearing A, Lury J, Wells G, Bone E. Matrix metalloproteinases degrade myelin basic protein. Neurosci Lett. 1995;201(3):223-226. doi: 10.1016/0304-3940(95)12173-0
- Seo JH, Miyamoto N, Hayakawa K, et al. Oligodendrocyte precursors induce early blood-brain barrier opening after white matter injury. J Clin Invest. 2013;123:782-786. doi: 10.1172/JCI65863
- Huang Y, Zhang W, Lin L, et al. Is endothelial dysfunction of cerebral small vessel responsible for white matter lesions after chronic cerebral hypoperfusion in rats? J Neurol Sci. 2010;299(1-2):72-80. doi: 10.1016/j.jns.2010.08.035
- Yata K, Nishimura Y, Unekawa M, et al. In vivo imaging of the mouse neurovascular unit under chronic cerebral hypoperfusion. Stroke. 2014;45(12):3698-3703. doi: 10.1161/STROKEAHA.114.005891
- Salvadores N, Searcy JL, Holland PR, Horsburgh K. Chronic cerebral hypoperfusion alters amyloid-β peptide pools leading to cerebral amyloid angiopathy, microinfarcts and haemorrhages in Tg-SwDI mice. Clin Sci (Lond). 2017;131(16):2109-2123. doi: 10.1042/CS20170962
- Niwa K, Porter VA, Kazama K, Cornfield D, Carlson GA, Iadecola C. A beta-peptides enhance vasoconstriction in cerebral circulation. Am J Physiol Heart Circ Physiol. 2001;281(6):H2417-H2424. doi: 10.1152/ajpheart.2001.281.6.H2417
- Bradl M, Lassmann H. Oligodendrocytes: Biology and pathology. Acta Neuropathol (Berl). 2010;119(1):37-53. doi: 10.1007/s00401-009-0601-5
- Coban H, Tung S, Yoo B, Vinters HV, Hinman JD. Molecular disorganization of axons adjacent to human cortical microinfarcts. Front Neurol. 2017;8:405. doi: 10.3389/fneur.2017.00405
- Fernando MS, Simpson JE, Matthews F, et al. White matter lesions in an unselected cohort of the elderly: Molecular pathology suggests origin from chronic hypoperfusion injury. Stroke. 2006;37(6):1391-1398. doi: 10.1161/01.STR.0000221308.94473.14
- Chen J, Zhang C, Wu Y, Zhang D. Association between hypertension and the risk of Parkinson’s disease: A meta-analysis of analytical studies. Neuroepidemiology. 2019;52(3-4):181-192. doi: 10.1159/000496977
- Qiu C, Hu G, Kivipelto M, et al. Association of blood pressure and hypertension with the risk of Parkinson Disease: The national FINRISK study. Hypertension. 2011;57(6):1094-1100. doi: 10.1161/HYPERTENSIONAHA.111.171249
- Bohnen NI, Albin RL. White matter lesions in Parkinson disease. Nat Rev Neurol. 2011;7(4):229-236. doi: 10.1038/nrneurol.2011.21
- Tohgi H, Utsugisawa K, Yoshimura M, Nagane Y, Mihara M. Alterations with aging and ischemia in nicotinic acetylcholine receptor subunits alpha4 and beta2 messenger RNA expression in postmortem human putamen. Implications for susceptibility to parkinsonism. Brain Res. 1998;791(1-2):186-190. doi: 10.1016/S0006-8993(98)00093-6
- Papapetropoulos S, Ellul J, Argyriou AA, Talelli P, Chroni E, Papapetropoulos T. The effect of vascular disease on late onset Parkinson’s disease. Eur J Neurol. 2004;11(4):231-235. doi: 10.1046/j.1468-1331.2003.00748.x
- Kalantarian S, Stern TA, Mansour M, Ruskin JN. Cognitive impairment associated with atrial fibrillation: A meta-analysis. Ann Intern Med. 2013;158(5_Part_1):338-346. doi: 10.7326/0003-4819-158-5-201303050-00007
- Santangeli P, Di Biase L, Bai R, et al. Atrial fibrillation and the risk of incident dementia: A meta-analysis. Heart Rhythm. 2012;9(11):1761-1768.e2. doi: 10.1016/j.hrthm.2012.07.026
- Deckers K, Schievink SHJ, Rodriquez MMF, et al. Coronary heart disease and risk for cognitive impairment or dementia: Systematic review and meta-analysis. PLoS One. 2017;12(9):e0184244. doi: 10.1371/journal.pone.0184244
- Hong CT, Chan L, Wu D, Chen WT, Chien LN. Association between Parkinson’s disease and atrial fibrillation: A population-based study. Front Neurol. 2019;10:22. doi: 10.3389/fneur.2019.00022
- Rivard L, Friberg L, Conen D, et al. Atrial fibrillation and dementia: A report from the AF-SCREEN international collaboration. Circulation. 2022;145(5):392-409. doi: 10.1161/CIRCULATIONAHA.121.055018
- Wolf PA, Abbott RD, Kannel WB. Atrial fibrillation: A major contributor to stroke in the elderly. The framingham study. Arch Intern Med. 1987;147(9):1561. doi: 10.1001/archinte.1987.00370090041008
- Kalantarian S, Ay H, Gollub RL, et al. Association between atrial fibrillation and silent cerebral infarctions: A systematic review and meta-analysis. Ann Intern Med. 2014;161(9):650-658. doi: 10.7326/M14-0538
- Gupta A, Giambrone AE, Gialdini G, et al. Silent brain infarction and risk of future stroke: A systematic review and meta-analysis. Stroke. 2016;47(3):719-725. doi: 10.1161/STROKEAHA.115.011889
- Leys D, Hénon H, Mackowiak-Cordoliani MA, Pasquier F. Poststroke dementia. Lancet Neurol. 2005;4(11):752-759. doi: 10.1016/S1474-4422(05)70221-0
- Sigurdsson S, Aspelund T, Kjartansson O, et al. Incidence of brain infarcts, cognitive change, and risk of dementia in the general population: The AGES-reykjavik study (age gene/environment susceptibility-reykjavik study). Stroke. 2017;48(9):2353-2360. doi: 10.1161/STROKEAHA.117.017357
- Gaita F, Corsinovi L, Anselmino M, et al. Prevalence of silent cerebral ischemia in paroxysmal and persistent atrial fibrillation and correlation with cognitive function. J Am Coll Cardiol. 2013;62(21):1990-1997. doi: 10.1016/j.jacc.2013.05.074
- Iadecola C, Duering M, Hachinski V, et al. Vascular cognitive impairment and dementia: JACC scientific expert panel. J Am Coll Cardiol. 2019;73(25):3326-3344. doi: 10.1016/j.jacc.2019.04.034
- Polidori MC, Mariani E, Mecocci P, Nelles G. Congestive heart failure and Alzheimer’s disease. Neurol Res. 2006;28(6):588-594. doi: 10.1179/016164106X130489
- Saito T, Kawamura Y, Sato N, et al. Non-Vitamin K antagonist oral anticoagulants do not increase cerebral microbleeds. J Stroke Cerebrovasc Dis. 2015;24(6):1373-1377. doi: 10.1016/j.jstrokecerebrovasdis.2015.02.018
- Kummer BR, Diaz I, Wu X, et al. Associations between cerebrovascular risk factors and parkinson disease. Ann Neurol. 2019;86(4):572-581. doi: 10.1002/ana.25564
- Lee H, Kim HK, Kim B, et al. Augmented risk of dementia in hypertrophic cardiomyopathy: A propensity score matching analysis using the nationwide cohort. PLoS One. 2022;17(6):e0269911. doi: 10.1371/journal.pone.0269911
- Jefferson AL, Beiser AS, Himali JJ, et al. Low cardiac index is associated with incident dementia and Alzheimer disease: The Framingham heart study. Circulation. 2015;131(15):1333-1339. doi: 10.1161/CIRCULATIONAHA.114.012438
- Kemp CD, Conte JV. The pathophysiology of heart failure. Cardiovasc Pathol. 2012;21(5):365-371. doi: 10.1016/j.carpath.2011.11.007
- Tanai E, Frantz S. Pathophysiology of heart failure. In: Prakash YS, editor. Comprehensive Physiology. 1st ed. United States: Wiley; 2015. p. 187-214. doi: 10.1002/cphy.c140055
- Qiu C, Winblad B, Marengoni A, Klarin I, Fastbom J, Fratiglioni L. Heart failure and risk of dementia and Alzheimer disease: A population-based cohort study. Arch Intern Med. 2006;166(9):1003. doi: 10.1001/archinte.166.9.1003
- Jinawong K, Apaijai N, Chattipakorn N, Chattipakorn SC. Cognitive impairment in myocardial infarction and heart failure. Acta Physiol. 2021;232(1):e13642. doi: 10.1111/apha.13642
- Borchert T, Hess A, Lukačević M, Ross TL, Bengel FM, Thackeray JT. Angiotensin-converting enzyme inhibitor treatment early after myocardial infarction attenuates acute cardiac and neuroinflammation without effect on chronic neuroinflammation. Eur J Nucl Med Mol Imaging. 2020;47(7):1757-1768. doi: 10.1007/s00259-020-04736-8
- Althammer F, Ferreira-Neto HC, Rubaharan M, et al. Three-dimensional morphometric analysis reveals time-dependent structural changes in microglia and astrocytes in the central amygdala and hypothalamic paraventricular nucleus of heart failure rats. J Neuroinflammation. 2020;17(1):221. doi: 10.1186/s12974-020-01892-4
- Lee CH, Hwang IK, Choi JH, et al. Calcium binding proteins immunoreactivity in the rat basolateral amygdala following myocardial infarction. Cell Mol Neurobiol. 2010;30(3):333-338. doi: 10.1007/s10571-009-9465-4
- Thomas T, Thomas G, McLendon C, Sutton T, Mullan M. beta-Amyloid-mediated vasoactivity and vascular endothelial damage. Nature. 1996;380(6570):168-171. doi: 10.1038/380168a0
- Malick M, Gilbert K, Brouillette J, Godbout R, Rousseau G. Cognitive deficits following a post-myocardial infarct in the rat are blocked by the serotonin-norepinephrine reuptake inhibitor desvenlafaxine. Int J Mol Sci. 2018;19(12):3748. doi: 10.3390/ijms19123748
- Gipson CD, Olive MF. Structural and functional plasticity of dendritic spines - root or result of behavior? Genes Brain Behav. 2017;16(1):101-117. doi: 10.1111/gbb.12324
- Meissner A, Visanji NP, Momen MA, et al. Tumor necrosis factor‐α underlies loss of cortical dendritic spine density in a mouse model of congestive heart failure. J Am Heart Assoc. 2015;4(5):e001920. doi: 10.1161/JAHA.115.001920
- Zesiewicz TA, Strom JA, Borenstein AR, et al. Heart failure in Parkinson’s disease: Analysis of the United States medicare current beneficiary survey. Parkinsonism Relat Disord. 2004;10(7):417-420. doi: 10.1016/j.parkreldis.2004.04.001
- Kennelly SP, Lawlor BA, Kenny RA. Blood pressure and the risk for dementia: A double edged sword. Ageing Res Rev. 2009;8(2):61-70. doi: 10.1016/j.arr.2008.11.001
- Chhatre S, Weiner MG, Jayadevappa R, Johnson JC. Incremental burden of congestive heart failure among elderly with Alzheimer’s. Aging Ment Health. 2009;13(4):628-634. doi: 10.1080/13607860902774469
- Tönnies E, Trushina E. Oxidative stress, synaptic dysfunction, and Alzheimer’s disease. J Alzheimer’s Dis. 2017;57(4):1105-1121. doi: 10.3233/JAD-161088
- Ganguly U, Kaur U, Chakrabarti SS, et al. Oxidative stress, neuroinflammation, and NADPH oxidase: Implications in the pathogenesis and treatment of Alzheimer’s disease. Oxid Med Cell Longev. 2021;2021(1):7086512. doi: 10.1155/2021/7086512
- Xie J, Van Hoecke L, Vandenbroucke RE. The impact of systemic inflammation on Alzheimer’s disease pathology. Front Immunol. 2022;12:796867. doi: 10.3389/fimmu.2021.796867
- Montagne A, Zhao Z, Zlokovic BV. Alzheimer’s disease: A matter of blood-brain barrier dysfunction? J Exp Med. 2017;214(11):3151-3169. doi: 10.1084/jem.20171406
- Serra JA, Domínguez RO, Marschoff ER, Guareschi EM, Famulari AL, Boveris A. Systemic oxidative stress associated with the neurological diseases of aging. Neurochem Res. 2009;34(12):2122-2132. doi: 10.1007/s11064-009-9997-5
- Cai H, Harrison DG. Endothelial dysfunction in cardiovascular diseases: The role of oxidant stress. Circ Res. 2000;87(10):840-844. doi: 10.1161/01.RES.87.10.840
- Vanempel V, Bertrand A, Hofstra L, Crijns H, Doevendans P, Dewindt L. Myocyte apoptosis in heart failure. Cardiovasc Res. 2005;67(1):21-29. doi: 10.1016/j.cardiores.2005.04.012
- Jabir NR, Firoz CK, Baeesa SS, et al. Synopsis on the linkage of Alzheimer’s and Parkinson’s disease with chronic diseases. CNS Neurosci Ther. 2015;21(1):1-7. doi: 10.1111/cns.12344
- Ottaviani G, Lavezzi AM, Rossi L, Matturri L. Proliferating cell nuclear antigen (PCNA) and apoptosis in hyperacute and acute myocardial infarction. Eur J Histochem. 1999;43(1):7-14.
- Troncone L, Luciani M, Coggins M, et al. Aβ amyloid pathology affects the hearts of patients with Alzheimer’s disease. J Am Coll Cardiol. 2016;68(22):2395-2407. doi: 10.1016/j.jacc.2016.08.073
- Gianni D, Li A, Tesco G, et al. Protein aggregates and novel presenilin gene variants in idiopathic dilated cardiomyopathy. Circulation. 2010;121(10):1216-1226. doi: 10.1161/CIRCULATIONAHA.109.879510
- Chen Z, Li G, Liu J. Autonomic dysfunction in Parkinson’s disease: Implications for pathophysiology, diagnosis, and treatment. Neurobiol Dis. 2020;134:104700. doi: 10.1016/j.nbd.2019.104700
- Chen Y, Dorn GW 2nd. PINK1-phosphorylated mitofusin 2 is a parkin receptor for culling damaged mitochondria. Science. 2013;340(6131):471-475. doi: 10.1126/science.1231031
- Grosu L, Grosu A, Crisan D, Zlibut A, PerjuDumbrava L. Parkinson’s disease and cardiovascular involvement: Edifying insights (review). Biomed Rep. 2023;18(3):25. doi: 10.3892/br.2023.1607
- Kwaśniak-Butowska M, Dulski J, Pierzchlińska A, Białecka M, Wieczorek D, Sławek J. Cardiovascular dysautonomia and cognition in Parkinson’s disease - a possible relationship. Neurol Neurochir Pol. 2021;55(6):525-535. doi: 10.5603/PJNNS.a2021.0040
- Cereja F, Alves M, Ferreira JJ, Caldeira D. Atrial fibrillation risk on Parkinson’s disease - a systematic review and meta-analysis. J Thromb Thrombolysis. 2023;55(4):747-750. doi: 10.1007/s11239-023-02792-z
- Tan CMJ, Green P, Tapoulal N, Lewandowski AJ, Leeson P, Herring N. The role of neuropeptide Y in cardiovascular health and disease. Front Physiol. 2018;9:1281. doi: 10.3389/fphys.2018.01281
- Widiapradja A, Chunduri P, Levick SP. The role of neuropeptides in adverse myocardial remodeling and heart failure. Cell Mol Life Sci. 2017;74(11):2019-2038. doi: 10.1007/s00018-017-2452-x
- Qin YY, Huang XR, Zhang J, et al. Neuropeptide Y attenuates cardiac remodeling and deterioration of function following myocardial infarction. Mol Ther. 2022;30(2):881-897. doi: 10.1016/j.ymthe.2021.10.005
- Yeo XY, Cunliffe G, Ho RC, Lee SS, Jung S. Potentials of neuropeptides as therapeutic agents for neurological diseases. Biomedicines. 2022;10(2):343. doi: 10.3390/biomedicines10020343

