The glymphatic system in migraine: A narrative review of emerging mechanisms, glial biomarkers, and diagnostic imaging approaches

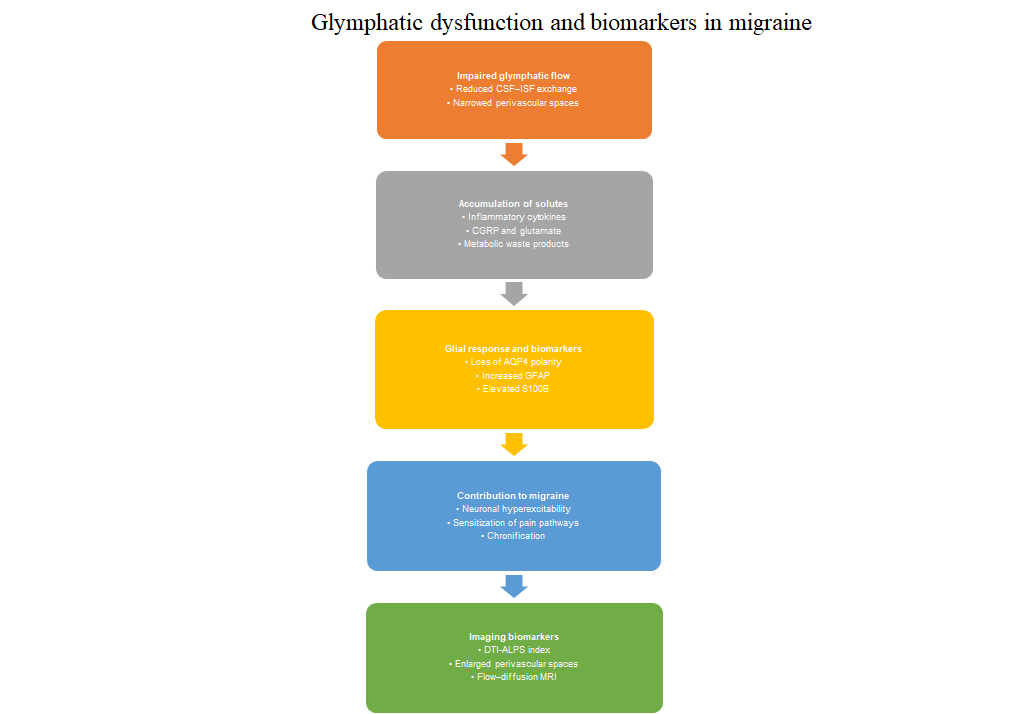
Emerging evidence suggests that dysfunction of the glymphatic system, a brain-wide clearance pathway responsible for regulating cerebrospinal and interstitial fluid exchange, plays a meaningful role in migraine pathophysiology. Impaired glymphatic flow may allow inflammatory mediators, metabolic waste products, and vasoactive peptides to accumulate within perivascular and interstitial spaces, promoting trigeminovascular activation, increasing neuronal excitability, and contributing to the progression from episodic to chronic migraine. Glia-derived biomarkers such as aquaporin-4, glial fibrillary acidic protein, and S100B provide important insight into astrocytic polarity, structural integrity, and neuroimmune activation, and may serve as clinically relevant indicators of glymphatic disturbance. Advances in neuroimaging, including diffusion-based analyses along perivascular pathways, quantitative assessment of enlarged perivascular spaces, and emerging fluid-sensitive magnetic resonance imaging techniques, offer noninvasive tools capable of detecting alterations in perivascular transport and microstructural clearance associated with migraine. Although methodological differences across studies require careful interpretation, converging findings indicate that migraine may be characterized not by a single uniform defect, but by heterogeneous patterns of impaired fluid handling and glial dysfunction. Incorporating molecular and imaging biomarkers into future research may improve diagnostic precision and inform therapeutic strategies aimed at restoring glymphatic and perivascular homeostasis.
- GBD 2016 Headache Collaborators. Global, regional, and national burden of migraine and tension-type headache, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2018;17(11):954-976. doi: 10.1016/S1474-4422(18)30322-3.
- Puledda F, Martins Silva E, Suwanlaong K, Goadsby PJ. Migraine: from pathophysiology to treatment. J Neurol. 2023;270(7):3654-3666. doi: 10.1007/s00415-023-11706-1.
- Dodick DW. A phase-by-phase review of migraine pathophysiology. Headache. 2018;58(Suppl 1):4-16. doi: 10.1111/head.13300
- Tanaka M, Tuka B, Vecsei L. Navigating the neurobiology of migraine: from pathways to potential therapies. Cells. 2024;13(13):1098. doi: 10.3390/cells13131098
- Olesen J. Personal view: modelling pain mechanisms of migraine without aura. Cephalalgia. 2022;42(13):1425-1435. doi: 10.1177/03331024221111529
- Borkum JM. Brain energy deficit as a source of oxidative stress in migraine: a molecular basis for migraine susceptibility. Neurochem Res. 2021;46(8):1913-1932. doi: 10.1007/s11064-021-03335-9
- Battaglia S, Avenanti A, Vecsei L, Tanaka M. Neurodegeneration in cognitive impairment and mood disorders for experimental, clinical and translational neuropsychiatry. Biomedicines. 2024;12(3):574. doi: 10.3390/biomedicines12030574
- Ashina M, Katsarava Z, Do TP, et al. Migraine: epidemiology and systems of care. Lancet. 2021;397(10283):1485–1495. doi: 10.1016/S0140-6736(20)32160-7
- Szlufik S, Kopec K, Szleszkowski S, Koziorowski D. Glymphatic system pathology and neuroinflammation as two risk factors of neurodegeneration. Cells. 2024;13(3):286. doi: 10.3390/cells13030286
- Shirolapov IV, Zakharov AV, Bulgakova SV, et al. Glymphatic dysfunction in the pathogenesis of neurodegenerative diseases and pathological aging. Genes Cells. 2023;18(4):309-322. doi: 10.23868/gc546022
- Kwon HS, Koh SH. Neuroinflammation in neurodegenerative disorders: the roles of microglia and astrocytes. Transl Neurodegener. 2020;9(1):42. doi: 10.1186/s40035-020-00221-2
- Benveniste H, Elkin R, Heerdt PM, et al. The glymphatic system and its role in cerebral homeostasis. J Appl Physiol (1985). 2020;129(6):1330-1340. doi: 10.1152/japplphysiol.00852.2019
- Natale G, Limanaqi F, Busceti CL, et al. Glymphatic system as a gateway to connect neurodegeneration from periphery to central nervous system. Front Neurosci. 2021;15:639140. doi: 10.3389/fnins.2021.639140
- Bohr T, Fan Z, Rasmussen PM, et al. The glymphatic system: current understanding and modeling. iScience. 2022;25(9):104987. doi: 10.1016/j.isci.2022.104987
- Lohela TJ, Lilius TO, Nedergaard M. The glymphatic system: implications for drugs for central nervous system diseases. Nat Rev Drug Discov. 2022;21(10):763-779. doi: 10.1038/s41573-022-00500-9
- Chen S, Li X, Wang Y, et al. The glymphatic system: a self-purification circulation in the brain. Front Cell Neurosci. 2025;19:1528995. doi: 10.3389/fncel.2025.1528995
- Cha MJ, Kang KW, Shin JW, Kim HS, Kim J. Understanding the connection between the glymphatic system and migraine: a systematic review. Headache Pain Res. 2024;25(2):86-95. doi: 10.62087/hpr.2024.0014
- Zou K, Deng Q, Zhang H, Huang C. Glymphatic system: a gateway for neuroinflammation. Neural Regen Res. 2024;19(12):2661-2672. doi: 10.4103/1673-5374.391312
- Chen H, Tang X, Li J, et al. IL-17 crosses the blood-brain barrier to trigger neuroinflammation: a novel mechanism in nitroglycerin-induced chronic migraine. J Headache Pain. 2022;23:1. doi: 10.1186/s10194-021-01374-9
- Corbali O, Levey AI. Glymphatic system in neurological disorders and implications for brain health. Front Neurol. 2025;16:1543725. doi: 10.3389/fneur.2025.1543725
- Eide PK, Lashkarivand A, Pripp AH, et al. Plasma neurodegeneration biomarker concentrations associate with glymphatic and meningeal lymphatic measures in neurological disorders. Nat Commun. 2023;14:2084. doi: 10.1038/s41467-023-37685-5
- van Dongen RM, Zielman R, Noga M, et al. Migraine biomarkers in cerebrospinal fluid: a systematic review and meta-analysis. Cephalalgia. 2017;37(1):49-63. doi: 10.1177/0333102416646768
- Demartini C, Francavilla M, Zanaboni AM, et al. Biomarkers of migraine: an integrated evaluation of preclinical and clinical findings. Int J Mol Sci. 2023;24(6):5334. doi: 10.3390/ijms24065334
- Chu C, Zhong R, Cai M, Li N, Lin W. Elevated blood S100B levels in patients with migraine: a systematic review and meta-analysis. Front Neurol. 2022;13:914051. doi: 10.3389/fneur.2022.914051
- Celikbilek A, Sabah S, Tanik N, Ak H, Atalay T, Yilmaz N. Is serum S100B protein a useful biomarker in migraine? Neurol Sci. 2014;35(8):1197-1201. doi: 10.1007/s10072-014-1679-7
- Teepker M, Munk K, Mylius V, et al. Serum concentrations of S100B and neuron-specific enolase in migraine. Headache. 2009;49(2):245–252. doi: 10.1111/j.1526-4610.2008.01228.x
- Gonen M, Ozdogan S, Balgetir F, Demir CF, Aytac E, Mungen B. S100B and neuron-specific enolase levels in episodic and chronic migraine. Acta Neurol Scand. 2021;143(3):298-302. doi: 10.1111/ane.13365
- Yilmaz S. Serum NO, S100B, NSE concentrations in migraine and their relationship. J Clin Neurosci. 2020;82:32–35. doi: 10.1016/j.jocn.2020.10.046
- Peng S, Liu J, Liang C, Yang L, Wang G. Aquaporin-4 in glymphatic system, and its implication for central nervous system disorders. Neurobiol Dis. 2023;179:106035. doi: 10.1016/j.nbd.2023.106035
- Gozalov E, Karlsson WK, Christensen RH, et al. Serum GFAP and NfL levels in migraine: a registry for migraine (REFORM) study. Cephalalgia. 2025;45(5):03331024251338178. doi: 10.1177/03331024251338178
- Colombo E, Doretti A, Rao R, et al. Plasma levels of glial fibrillary acidic protein and neurofilament light chain in patients with chronic migraine: a multicenter case-control study. Neurol Sci. 2025;46(5):2209-2216. doi: 10.1007/s10072-025-08011-2
- Janigro D, Mondello S, Posti JP, Unden J. GFAP and S100B: what you always wanted to know and never dared to ask. Front Neurol. 2022;13:835597. doi: 10.3389/fneur.2022.835597
- Zhu B, Zhou W, Chen C, et al. Aquaporin-4 is an emerging regulator of pathological pain: a narrative review. Cell Mol Neurobiol. 2023;43(8):3997-4005. doi: 10.1007/s10571-023-01422-9
- Carta S, Chiodega V, Tiberi R, et al. Glial fibrillary acidic protein as a marker of astrocytic damage correlated with medication overuse in migraine. Immunol Res. 2025;73:119. doi: 10.1007/s12026-025-09674-x
- Holper S, Loveland P, Churilov L, Italiano D, Watson R, Yassi N. Blood astrocyte biomarkers in Alzheimer disease: a systematic review and meta-analysis. Neurology. 2024;103(3):e209537. doi: 10.1212/WNL.0000000000209537
- Kamada J, Hamanaka T, Oshimo A, et al. Glial fibrillary acidic protein’s usefulness as an astrocyte biomarker using the fully automated LUMIPULSE system. Diagnostics 2024;14(22):2520. doi: 10.3390/diagnostics14222520
- Gayger-Dias V, Vizuete AF, Rodrigues L, et al. How S100B crosses brain barriers and why it is considered a peripheralmarker of brain injury. Exp Biol Med. 2023;248(22):2109-2119. doi: 10.1177/15353702231214260
- Ghaderi S, Mohammadi S, Fatehi F. Glymphatic system impairment along the perivascular space in adults with moyamoya disease: a systematic review and meta-analysis. Heliyon. 2025;11(11):e43451. doi: 10.1016/j.heliyon.2025.e43451
- Yu L, Hu X, Li H, Zhao Y. Perivascular spaces, glymphatic system and MR. Front Neurol. 2022;13:844938. doi: 10.3389/fneur.2022.844938
- Ringstad G. Glymphatic imaging: a critical look at the DTIALPS index. Neuroradiology. 2024;66(2):157-160. doi: 10.1007/s00234-023-03270-2
- Tomizawa Y, Hagiwara A, Hoshino Y, et al. The glymphatic system as a potential biomarker and therapeutic target in secondary progressive multiple sclerosis. Mult Scler Relat Disord. 2024;83:105437. doi: 10.1016/j.msard.2024.105437
- Marin I, Torres F, Riveros R, et al. Evaluation of the DTIALPS index as a biomarker of the glymphatic system at 1.5T. Open Neuroimaging J. 2025;18:e18744400389049. doi: 10.2174/0118744400389049250723091323
- Zhang X, Wang W, Bai X, et al. Increased glymphatic system activity in migraine chronification by diffusion tensor image analysis along the perivascular space. J Headache Pain. 2023;24:147. doi: 10.1186/s10194-023-01673-3
- Sun L, Cui K, Dong L, et al. Diffusion tensor image analysis along the perivascular space index and visual analogue scales in female patients with patent foramen ovale and migraine perform better after percutaneous closure. Quant Imaging Med Surg. 2025;15(8):6969-6980. doi: 10.21037/qims-2025-265
- Iliff JJ, Wang M, Liao Y, et al. A paravascular pathway facilitates cerebrospinal fluid flow through the brain parenchyma and the clearance of interstitial solutes, including amyloid-β. Sci Transl Med. 2012;4(147):147ra111. doi: 10.1126/scitranslmed.3003748

