Ellagic acid from Terminalia leiocarpa downregulates estrogen-dependent signaling and epithelial-to-mesenchymal transition canonical pathways in MCF-7 cells

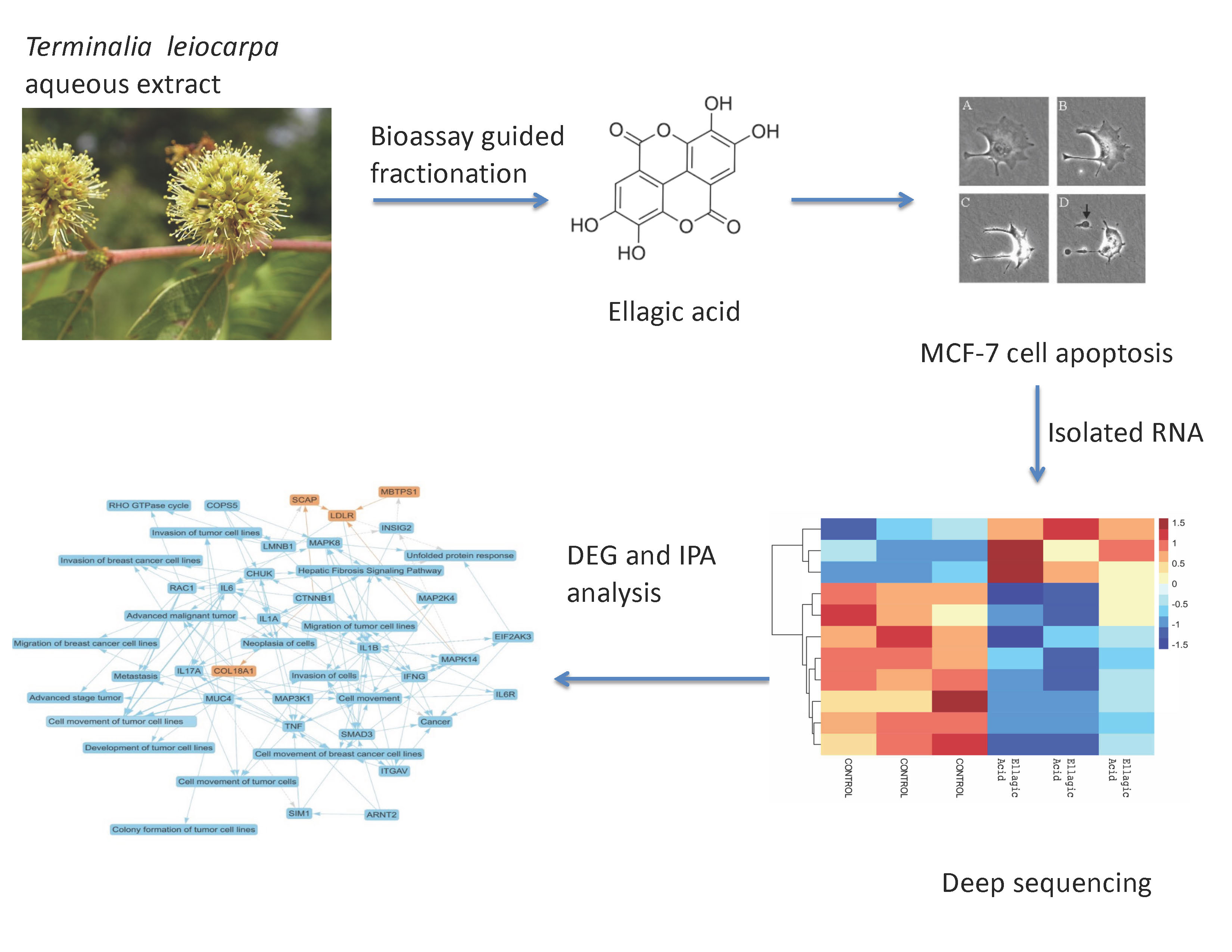
Breast cancer remains the most diagnosed cancer among women worldwide, and its prevalence continues to increase annually. Previously, we reported that ellagic acid (EA), a compound isolated from Terminalia leiocarpa (DC.) Baill., a plant used in Nigerian traditional medicine, inhibited the proliferation of cultured human MCF-7 breast cancer cells. EA is a pharmacologically active polyphenolic compound with antioxidant, anti-inflammatory, and anti-estrogenic activities. In this work, we investigated the effects of EA on the transcriptome of MCF-7 cells using next-generation sequencing. After EA treatment, MCF-7 cells were harvested, and total RNA was isolated and used to prepare RNA sequencing libraries. Sequencing was performed using NovaSeq 6000. The results showed that EA significantly altered gene expression patterns in MCF-7 cells. Bioinformatic analysis of the sequencing data revealed that 4,848 transcripts were differentially expressed, with 2,180 upregulated genes and 2,668 downregulated genes (q ≤ 0.01). Ingenuity Pathway Analysis indicated that EA-induced expression changes mapped to 98 canonical pathways, including upregulation of apoptosis signaling pathways and downregulation of estrogen signaling and estrogen-dependent breast cancer signaling pathways (q ≤ 0.05). Furthermore, EA downregulated gene expression in the epithelial-to-mesenchymal transition (EMT) canonical pathway, such as PRRX1/2, TGF, TWIST1, and VIM. These data indicate that EA has multiple mechanisms of action in MCF-7 cells, including the induction of apoptosis and the reduction of both estrogen and EMT signaling. Thus, with further research, EA may serve as an inexpensive, safe, and effective treatment for breast cancer and metastatic diseases.
- World Health Organisation: Breast cancer factsheet. Updated 2025. Available from: https://www.who.int/newsroom/fact-sheets/detail/breast-cancer [Last accessed on 2025 May 24].
- Azubuike SO, Muirhead C, Hayes L, McNally R. Rising global burden of breast cancer: the case of sub-Saharan Africa (with emphasis on Nigeria) and implications for regional development: a review. World J Surg Onc. 2018;16:63. doi: 10.1186/s12957-018-1345-2
- Adeloye D, Sowunmi OY, Jacobs W, David RA, Adeosun AA, Amuta AO, Misra S, Gadanya M, Auta A, Harhay MO, Chan KY. Estimating the incidence of breast cancer in Africa: a systematic review and meta-analysis. J Glob Health. 2018;8(1):010419. doi: 10.7189/jogh.08.010419.
- Onyia AF, Nana TA, Adewale EA, et al. Breast cancer phenotypes in Africa: A scoping review and meta-analysis. JCO Glob Oncol. 2018;9:e2300135. doi: 10.1200/GO.23.00135.
- Ntekim A, Oluwasanu M, Odukoya O. Breast cancer in adolescents and young adults less than 40 years of age in Nigeria: A retrospective analysis. Int J Breast Cancer. 2022;2022:9943247. doi: 10.1155/2022/9943247.
- Asuzu CC, Akin-Odanye EO, Asuzu MC, Holland J. A socio-cultural study of traditional healers’ role in African health care. Infect Agent Cancer. 2019;14:15. doi: 10.1186/s13027-019-0232-y.
- Lawal TO, Patel S, Raut N, Mahady GB. Extracts of Anogeissus leiocarpus and Dillenia indica inhibit the growth of MCF-7 breast cancer and COV434 granulosa tumour cells by inducing apoptosis and autophagy. Current Bioactive Compounds. 2021;17:e190721191390. doi: 10.2174/1573407217666210215092955
- Odumosu BT, Kolude B, Adeniyi BA. Antimicrobial screening and kinetic study of two Nigerian medicinal plants against oral pathogens. CIBTec J Pharm Sci. 2014;3(2):7-14.
- Ezeome ER. Delays in presentation and treatment of breast cancer in Enugu, Nigeria. Niger J Clin Pract. 2010;13(3):311–316.
- WFO Plant List: Snapshots of the Taxonomy. Available from: https://www.WFOplantlist.org [Last accessed on 2025 May 06].
- Adekunle YA, Samuel BB, Nahar L, Fatokun AA, Sarker SD. Anogeissus leiocarpus (DC.) Guill. & Perr. (Combretaceae): A review of the traditional uses, phytochemistry and pharmacology of African birch. Fitoterapia. 2024;176:105979. doi: 10.1016/j.fitote.2024.105979.
- Micheli L, Ferrara V, Akande T, et al. Ellagitannins and triterpenoids extracts of Anogeissus leiocarpus stem bark extracts: Protective effects against osteoarthritis. Phytother Res. 2023;37(6):2381-2394. doi: 10.1002/ptr.7760.
- Singh D, Baghel US, Gautam A, et al. The genus Anogeissus: A review on ethnopharmacology, phytochemistry and pharmacology. J Ethnopharmacol. 2016;194:30-56. doi: 10.1016/j.jep.2016.08.025.
- Arbab AH. Review on Anogeissus leiocarpus a potent African traditional drug. Int J Res Pharm Chem. 2014;4:496–500.
- Salau AK, Yakubu MT, Oladiji, AT. Cytotoxic activity of aqueous extracts of Anogeissus leiocarpus and Terminalia avicennioides root barks against Ehrlich ascites carcinoma cells. Indian J Pharmacol. 2013;45(4):381-385. doi: 10.4103/0253-7613.115023
- Olugbami JO, Damoiseaux R, France B, et al. A comparative assessment of antiproliferative properties of resveratrol and ethanol leaf extract of Anogeissus leiocarpus (DC) Guill and Perr against HepG2 hepatocarcinoma cells. BMC Complement Altern Med. 2017;17(1). doi: 10.1186/s12906-017-1873-2
- Hassan LEA, Al-Suede FS, Fadul SM, Abdul Majid AMS. Evaluation of antioxidant, antiangiogenic and antitumor properties of Anogeissus leiocarpus against colon cancer. Angiotherapy. 2018;1(2):56–66. doi: 10.25163/angiotherapy.1200021526100818
- Lawal TO, Patel S, Mahady, GB. Food and medicinal plants from Nigeria with anti-Helicobacter pylori activities induce apoptosis in colon and gastric cancer cell lines. Functional Foods in Health and Disease. 2023;13(7):372-387. doi: 10.31989/ffhd.v13i7.1105
- Seigler, DS. Plant Secondary Metabolism. Kluwer Academic Publishers, Springer Science & Business Media. 1998:208.
- Golmei P, Kasna S, Roy KP, Kumar S. A review on pharmacological advancement of ellagic acid. J Pharmacol Pharmacother. 2024;15(2):93-104. doi: 10.1177/0976500X241240634
- Kanabar PN, Los NS, Lawal TO, Patel S, Raut NA, Maienschein-Cline M, et al. Combinations of vitamin A and D induced are synergistic in breast cancer cells and alter gene expression in the endoplasmic reticulum stress, unfolded protein and oestrogen signalling canonical. Funct Foods Health Dis. 2023;13(3):135-155. doi: 10.31989/ffhd.v13i3.1069
- Ostos Mendoza KC, Garay Buenrostro KD, Kanabar PN, et al. Peonidin-3-O-glucoside and resveratrol increase the viability of cultured human hFOB osteoblasts and alter the expression of genes associated with apoptosis, osteoblast differentiation and osteoclastogenesis. Nutrients. 2023;15(14):3233. doi: 10.3390/nu15143233
- Dobin A, Davis CA, Schlesinger F, et al. STAR: ultrafast universal RNA-seq aligner. Bioinformatics. 2013;29(1):15–21. doi: 10.1093/bioinformatics/bts635
- Liao Y, Smyth GK, Shi W. FeatureCounts: An efficient general-purpose program for assigning sequence reads to genomic features. Bioinformatics. 2014;30(7):923–930. doi: 10.1093/bioinformatics/btt656
- McCarthy DJ, Chen Y, Smyth GK. Differential expression analysis of multifactor RNA-Seq experiments with respect to biological variation. Nucleic Acids Res. 2012;40(10):4288–4297. doi: 10.1093/nar/gks042
- Benjamini Y, Hochberg Y. Controlling the false discovery rate: A practical and powerful approach to multiple testing. J R Stat Soc Ser B (Methodol.). 1995;57(1):289-300. doi: 10.1111/j.2517-6161.1995.tb02031.x
- Cai Y, Dai F, Ye Y, Qian J. The global burden of breast cancer among women of reproductive age: a comprehensive analysis. Sci Rep. 2025;15(1):9347. doi: 10.1038/s41598-025-93883-9
- Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71(3):209-249. doi: 10.3322/caac.21660
- Rotimi SO, Rotimi OA, Salhia B. A review of cancer genetics and genomics studies in Africa. Front Oncol. 2021;10:606400. doi: 10.3389/fonc.2020.606400
- Orlando G, Ferrante C, Zengin G, et al. Qualitative chemical characterization and multidirectional biological investigation of leaves and bark extracts of Anogeissus leiocarpus (DC.) Guill. & Perr. (Combretaceae). Antioxidants. 2019;8(9):343. doi: 10.3390/antiox8090343
- Assuncao PID, da Conceicao ED, Borges LL, Marciano de Paula JA. Development and validation of a HPLC-UV method for the evaluation of ellagic acid in liquid extracts of Eugenia uniflora L. (Myrtaceae) leaves and its ultrasoundassisted extraction optimization. Evid Based Complement Alt Med. 2017;2017(1):1501038. doi: 10.1155/2017/1501038
- Cheng H, Lu C, Tang R, Pan Y, Bao S, Qiu Y, Xie M. Ellagic acid inhibits the proliferation of human pancreatic carcinoma PANC-1 cells in vitro and in vivo. Oncotarget. 2017;8(7):12301-12310. doi: 10.18632/oncotarget.14811
- Wang F, Chen J, Xiang D, Lian X, Wu C, Quan J. Ellagic acid inhibits cell proliferation, migration, and invasion in melanoma via EGFR pathway. Am J Transl Res. 2020;12(5):2295-2304.
- Naiki-Ito A, Chewonarin T, Tang M, et al. Ellagic acid, a component of pomegranate fruit juice, suppresses androgen-dependent prostate carcinogenesis via induction of apoptosis. Prostate. 2015;75(2):151-160. doi: 10.1002/pros.22900
- Li TM, Chen GW, Su CC, et al. Ellagic acid induced p53/p21 expression, G1 arrest and apoptosis in human bladder cancer T24 cells. Anticancer Res. 2005;25(2A):971-979.
- Kim H, Lee RA, Moon BI, Choe KJ. Ellagic acid shows different anti-proliferative effects between the MDA-MB-231 and MCF-7 human breast cancer cell lines. J Breast Cancer. 2009;12(2):85-91. doi: 10.4048/jbc.2009.12.2.85
- Saleem A, Husheem M, Harkonen P, Pihlaja K. Inhibition of cancer cell growth by crude extract and the phenolics of Terminalia chebula retz. fruit. J Ethnopharmacol. 2002;81(3):327-336. doi: 10.1016/s0378-8741(02)00099-5
- Golmohammadi M, Zamanian MY, Jalal SM, et al. A comprehensive review on ellagic acid in breast cancer treatment: From cellular effects to molecular mechanisms of action. Food Sci Nutr, 2023;11(12):7458-7468. doi: 10.1002/fsn3.3699
- Chen HS, Bai MH, Zhang T, Li GD, Liu M. Ellagic acid induces cell cycle arrest and apoptosis through TGF-β/Smad3 signaling pathway in human breast cancer MCF-7 cells. Int J Oncol. 2015;46(4):1730-1738. doi: 10.3892/ijo.2015.2870
- Yousuf M, Shamsi A, Khan P, et al. Ellagic acid controls cell proliferation and induces apoptosis in breast cancer cells via inhibition of cyclin-dependent kinase 6. Int J Mol Sci. 2020;21(10):3526. doi: 10.3390/ijms21103526
- Yousuf M, Shamsi A, Mohammad T, et al. Inhibiting cyclindependent kinase 6 by taurine: Implications in anticancer therapeutics. ACS Omega, 2022;7(29):25844-25852. doi: 10.1021/acsomega.2c03479
- Wang N, Wang ZY, Mo SL, et al. Ellagic acid, a phenolic compound, exerts anti-angiogenesis effects via VEGFR-2 signaling pathway in breast cancer. Breast Cancer Res Treat. 2012;134(3):943-955. doi: 10.1007/s10549-012-1977-9
- Papoutsi Z, Kassi E, Tsiapara A, Fokialakis N, Chrousos GP, Moutsatsou P. Evaluation of estrogenic/antiestrogenic activity of ellagic acid via the estrogen receptor subtypes ERα and ERβ. J Agric Food Chem. 2005;53(20):7715-7720. doi: 10.1021/jf0510539
- Poirier D, Roy J, Maltais R. A targeted-covalent inhibitor of 17β-HSD1 blocks two estrogen-biosynthesis pathways: In vitro (metabolism) and in vivo (xenograft) studies in T-47D breast cancer models. Cancers. 2021;13(8):1841. doi: 10.3390/cancers13081841
- Wang XQ, Aka JA, Li T, Xu D, Doillon CJ, Lin SX. Inhibition of 17beta-hydroxysteroid dehydrogenase type 7 modulates breast cancer protein profile and enhances apoptosis by down-regulating GRP78. J Steroid Biochem Mol Biol. 2017;172:188-197. doi: 10.1016/j.jsbmb.2017.06.009
- He W, Gauri M, Li T, Wang R, Lin SX. Current knowledge of the multifunctional 17β-hydroxysteroid dehydrogenase type 1 (HSD17B1). Gene. 2016;588(1):54-61. doi: 10.1016/j.gene.2016.04.031
- Zhang CY, Wang WQ, Chen J, Lin SX. Reductive 17betahydroxysteroid dehydrogenases which synthesize estradiol and inactivate dihydrotestosterone constitute major and concerted players in ER+ breast cancer cells. J Steroid Biochem Mol Biol. 2015;150:24-34. doi: 10.1016/j.jsbmb.2014.09.017
- Kang Y, Massague J. Epithelial-mesenchymal transitions: twist in development and metastasis. Cell. 2004;118(3):277-279. doi: 10.1016/j.cell.2004.07.011
- Wang Y, Liu J, Ying X, Lin PC, Zhou BP. Twist-mediated epithelial-mesenchymal transition promotes breast tumor cell invasion via inhibition of Hippo pathway. Sci Rep. 2016;6:24606. doi: 10.1038/srep24606
- Wu S, Du Y, Beckford J, Alachkar H. Upregulation of the EMT marker vimentin is associated with poor clinical outcome in acute myeloid leukemia. J Transl Med. 2018;16(1):170. doi: 10.1186/s12967-018-1539-y
- Berr AL, Wiese K, Dos Santos G, et al. Vimentin is required for tumor progression and metastasis in a mouse model of non-small cell lung cancer. Oncogene. 2023;42(25):2074-2087. doi: 10.1038/s41388-023-02703-9
- Usman S, Waseem NH, Nguyen, TKN, et al. Vimentin is at the heart of epithelial mesenchymal transition (EMT) mediated metastasis. Cancers. 2021;13:4985. doi: 10.3390/cancers13194985.
- Du W, Liu X, Yang M, Wang W, Sun J. The regulatory role of PRRX1 in cancer epithelial-mesenchymal transition. Onco Targets Ther. 2021;14:4223-4229. doi: 10.2147/OTT.S316102.
- Pandey P, Khan F, Upadhyay TK, Seungjoon M, Park MN, Kim B. New insights about the PDGF/PDGFR signalling pathway as a promising target to develop cancer therapeutic strategies. Biomed Pharmacother. 2023;161:114491. doi: 10.1016/j.biopha.2023.114491.
- Lim SC, Hwang H, Han SI. Ellagic acid inhibits extracellular acidity-induced invasiveness and expression of COX, Snail, Twist 1, and c-myc in gastric carcinoma cells. Nutrients. 2019;11(12):3023-3034. doi: 10.3390/nu11123023.

