Antibacterial potential, phytochemical constituents, and toxicity assessment of Azadirachta indica leaf extracts in combination with antibiotics

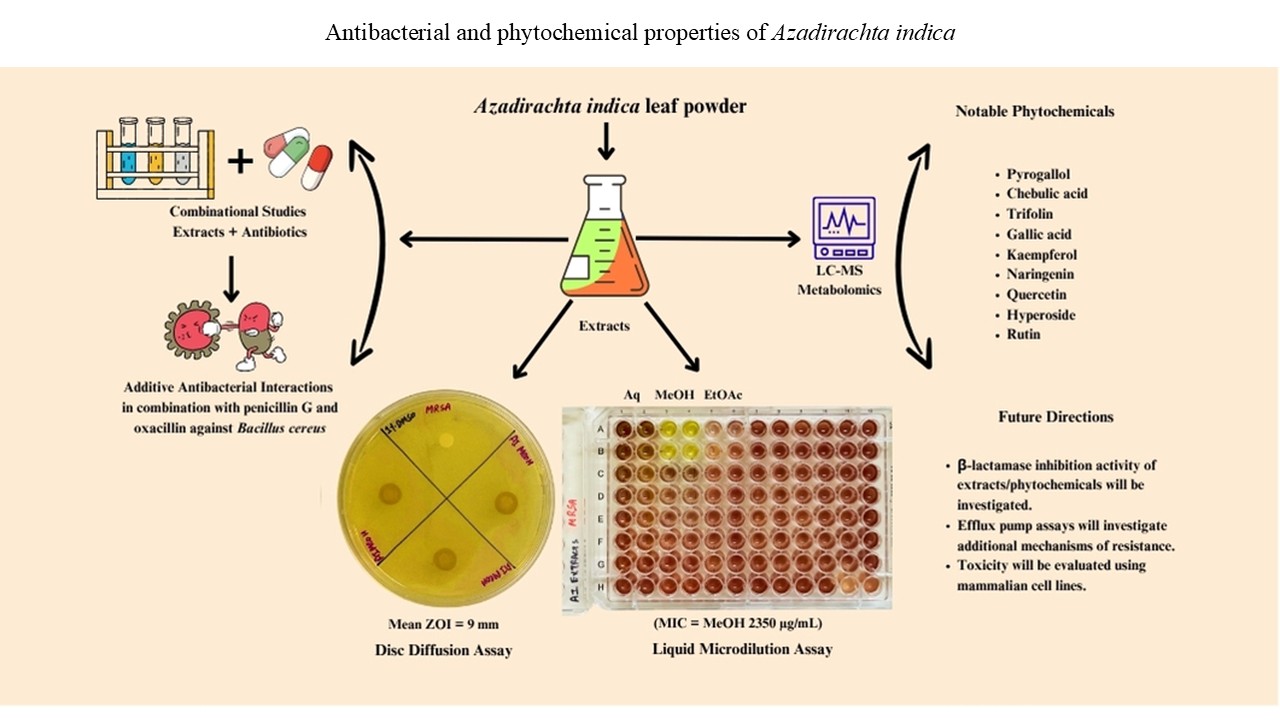
The rise of antimicrobial resistance (AMR) has highlighted the need for exploration of alternative therapeutic agents, including medicinal plants. This study investigates the antibacterial potential, phytochemical composition, and toxicity of Azadirachta indica leaf extracts, along with their interactions with conventional antibiotics. Extracts prepared in water (AQ), methanol (MeOH), and ethyl acetate (EtOAc) were evaluated for growth-inhibitory effects against a range of bacterial species, including drug-resistant isolates, through both disc diffusion and microdilution methods. Zone of inhibition (ZOI) measurements ranged from 7 to 10 mm, while minimum inhibitory concentration values varied between 2938 and 5875 μg/mL for AQ extracts, and between 1175 and 4700 μg/mL for MeOH extracts. Notably, EtOAc extracts did not inhibit bacterial growth in either assay. Fractional inhibitory concentration (FIC) analysis revealed additive interactions of extracts with β-lactam antibiotics (penicillin G, oxacillin, amoxicillin) and protein synthesis inhibitors (tetracycline, chloramphenicol), particularly against Bacillus cereus, Staphylococcus aureus, and Shigella spp., suggesting a potential role as antibiotic adjuvants. However, antagonistic interactions were also observed in combinations containing gentamicin and polymyxin B. Liquid chromatography–mass spectrometry (LC-MS) analysis confirmed the presence of phytochemicals, including flavonoids, tannins, and phenolics, which are known for their antimicrobial properties. Evaluation of extract safety using brine shrimp (Artemia franciscana) assays revealed no toxic responses. To ensure their safety for therapeutic applications, future toxicity evaluations should be conducted on mammalian cell lines to confirm these findings. Future research should focus on isolating bioactive compounds from the extracts, elucidating their mechanisms of action, and optimizing extract-antibiotic formulations to combat resistant bacterial infections effectively.
- Arip M, Selvaraja MR, Tan LF, et al. Review on plant-based management in combating antimicrobial resistance - mechanistic perspective. Front Pharmacol. 2022;13:879495. doi: 10.3389/fphar.2022.879495
- World Bank Group. Antimicrobial Resistance (AMR). World Bank Group; 2025, Available from: https://www.worldbank. org/en/topic/health/brief/antimicrobial-resistance-amr [Last accessed on 2025 Jul 27].
- Centers for Disease Control and Prevention (CDC). CDC Partners Estimate Healthcare Cost of Antimicrobial-resistant Infection. Centers for Disease Control and Prevention; 2025. Available from: https://www.cdc.gov/antimicrobial-resistance/stories/partner-estimates.html [Last accessed on 2025 Aug 14].
- Khameneh B, Eskin NAM, Iranshahy M, Bazzaz BSF. Phytochemicals: A promising weapon in the arsenal against antibiotic-resistant bacteria. Antibiotics (Basel). 2021;10(9):1044. doi: 10.3390/antibiotics10091044
- Vaou N, Stavropoulou E, Voidarou C, Tsigalou C, Bezirtzoglou E. Towards advances in medicinal plant antimicrobial activity: A review study on challenges and future perspectives. Microorganisms. 2021;9(10):2041. doi: 10.3390/microorganisms9102041
- Cha JD, Moon SE, Kim JY, Jung EK, Lee YS. Antibacterial activity of sophoraflavanone G isolated from the roots of Sophora flavescens against methicillin-resistant Staphylococcus aureus. Phytother Res. 2009;23:1326-1331. doi: 10.1002/ptr.2540
- Tiwana G, Cock IE, Cheesman MJ. Combinations of Terminalia bellirica (Gaertn.) Roxb. and Terminalia chebula Retz. extracts with selected antibiotics against antibiotic-resistant bacteria: Bioactivity and phytochemistry. Antibiotics (Basel). 2024;13(10):994. doi: 10.3390/antibiotics13100994
- Tiwana G, Cock IE, Cheesman MJ. A review of Ayurvedic principles and the use of Ayurvedic plants to control diarrhoea and gastrointestinal infections. Pharmacogn Commun. 2023;13(4):152-162. doi: 10.5530/pc.2023.4.25
- Cheesman M, Ilanko A, Blonk B, Cock IE. Developing new antimicrobial therapies: Are synergistic combinations of plant extracts/compounds with conventional antibiotics the solution? Pharmacogn Rev. 2017;11(22):57-72. doi: 10.4103/phrev.phrev_21_17
- Francine U, Jeannette U, Pierre R. Assessment of antibacterial activity of neem plant (Azadirachta indica) on Staphylococcus aureus and Escherichia coli. J Med Plant Stud. 2015;3(4):85-91.
- Koona S, Budida S. Antibacterial potential of the extracts of the leaves of Azadirachta indica Linn. Not Sci Biol. 2011;3(1):65-69. doi: 10.15835/nsb315470
- Altayb HN, Yassin NF, Hosawi S, Kazmi I. In-vitro and in-silico antibacterial activity of Azadirachta indica (neem), methanolic extract, and identification of Beta.d-mannofuranoside as a promising antibacterial agent. BMC Plant Biol. 2022;22:262. doi: 10.1186/s12870-022-03650-5
- Balouiri M, Sadiki M, Ibnsouda SK. Methods for in vitro evaluating antimicrobial activity: A review. J Pharm Anal. 2016;6(2):71-79. doi: 10.1016/j.jpha.2015.11.005
- Tiwana G, Cock IE, Cheesman MJ. Phytochemical analysis and antimicrobial activity of Terminalia bellirica (Gaertn.) Roxb. and Terminalia chebula Retz. fruit extracts against gastrointestinal pathogens: Enhancing antibiotic efficacy. Microorganisms. 2024;12:2664. doi: 10.3390/microorganisms12122664
- Eloff JN. A proposal towards a rational classification of the antimicrobial activity of acetone tree leaf extracts in a search for new antimicrobials. Planta Med. 2021;87:836-840. doi: 10.1055/a-1482-1410
- Doern CD. When does 2 plus 2 equal 5? A review of antimicrobial synergy testing. J Clin Microbiol. 2014;52(12):4124-4128. doi: 10.1128/jcm.01121-14
- Cajka T, Hricko J, Rudl Kulhava L, Paucova M, Novakova M, Kuda O. Optimization of mobile phase modifiers for fast LC-MS-based untargeted metabolomics and lipidomics. Int J Mol Sci. 2023;24(3):1987. doi: 10.3390/ijms24031987
- Angelini P. Plant-derived antimicrobials and their crucial role in combating antimicrobial resistance. Antibiotics (Basel). 2024;13:746. doi: 10.3390/antibiotics13080746
- Tiwana G, Cock IE, White A, Cheesman MJ. Use of specific combinations of the Triphala plant component extracts to potentiate the inhibition of gastrointestinal bacterial growth. J Ethnopharmacol. 2020;260:112937. doi: 10.1016/j.jep.2020.112937
- Altemimi A, Lakhssassi N, Baharlouei A, Watson DG, Lightfoot DA. Phytochemicals: Extraction, isolation, and identification of bioactive compounds from plant extracts. Plants (Basel). 2017;6(4):42. doi: 10.3390/plants6040042
- Bonev B, Hooper J, Parisot J. Principles of assessing bacterial susceptibility to antibiotics using the agar diffusion method. J Antimicrob Chemother. 2008;61(6):1295-1301. doi: 10.1093/jac/dkn090
- Paterson DL, Bonomo RA. Extended-spectrum beta-lactamases: A clinical update. Clin Microbiol Rev. 2005;18(4):657-686. doi: 10.1128/cmr.18.4.657-686.2005
- Auda IG, Ali Salman IM, Odah JG. Efflux pumps of gram-negative bacteria in brief. Gene Rep. 2020;20:100666. doi: 10.1016/j.genrep.2020.100666
- Puzari M, Sharma M, Chetia P. Emergence of antibiotic resistant Shigella species: A matter of concern. J Infect Public Health. 2018;11(4):451-454. doi: 10.1016/j.jiph.2017.09.025
- Alenazy R. Antibiotic resistance in Salmonella: Targeting multidrug resistance by understanding efflux pumps, regulators and the inhibitors. J King Saud Univ Sci. 2020;34:102275. doi: 10.1016/j.jksus.2022.102275
- Arsene MM, Viktorovna PI, Davares AK, et al. Antimicrobial and antibiotic-resistance reversal activity of some medicinal plants from Cameroon against selected resistant and non-resistant uropathogenic bacteria. Front Biosci (Elite Ed). 2022;14(4):4-25. doi: 10.31083/j.fbe1404025
- Singh AA, Naaz ZT, Rakaseta E, et al. Antimicrobial activity of selected plant extracts against common food borne pathogenic bacteria. Food Humanity. 2023;1:64-70. doi: 10.1016/j.foohum.2023.04.002
- Ousman BM, Mennane Z, Boussaoudi I, et al. First investigation of phytochemical screening, HPLC-MS characterization, and antibacterial activity of Azadirachta indica A. Juss (Mim or Neem) leaf extracts, grown in the Republic of Chad. Nat Prod Commun. 2025;20(2):1-16. doi: 10.1177/1934578X251322360
- Gibbons S. Phytochemicals for bacterial resistance-- strengths, weaknesses and opportunities. Planta Med. 2008;74(6):594-602. doi: 10.1055/s-2008-1074518
- Zavascki AP, Goldani LZ, Li J, Nation RL. Polymyxin B for the treatment of multidrug-resistant pathogens: A critical review. J Antimicrob Chemother. 2007;60(6):1206-1215. doi: 10.1093/jac/dkm357
- Cai X, Javor S, Gan BH, Köhler T, Reymond JL. The antibacterial activity of peptide dendrimers and polymyxin B increases sharply above pH 7.4. Chem Commun. 2021;57(46):5654-5657. doi: 10.1039/d1cc01838h
- Siddiqui BS, Rasheed M. Three new triterpenoids from Azadirachta indica. Helv Chim Acta. 2001;84(7):1962-1928. doi: 10.1002/1522-2675(20010711)84:7<1962:AID-HLCA1962>3.0.CO;2-V
- Pandey G, Verma K, Singh M. Evaluation of phytochemical, antibacterial and free radical scavenging properties of Azadirachta indica (neem) leaves. Int J Pharm Pharm Sci. 2014;6(2):444-447.
- Gupta R, Nand P, Drabu S. Insignificant anti-acne activity of Azadirachta indica leaves and bark. J Pharm Negat Results. 2012;3(1):29-33. doi: 10.4103/0976-9234.99650
- Saleem S, Muhammad G, Hussain MA, Bukhari SNA. A comprehensive review of phytochemical profile, bioactives for pharmaceuticals, and pharmacological attributes of Azadirachta indica. Phytother Res. 2018;32(7):1241-1272. doi: 10.1002/ptr.6076
- Pal A, Tripathi A. Demonstration of bactericidal and synergistic activity of quercetin with meropenem among pathogenic carbapenem resistant Escherichia coli and Klebsiella pneumoniae. Microb Pathog. 2020;143:104120. doi: 10.1016/j.micpath.2020.104120
- Yi L, Cao M, Chen X, et al. In vitro antimicrobial synergistic activity and the mechanism of the combination of naringenin and amikacin against antibiotic-resistant Escherichia coli. Microorganisms. 2024;12(9):1871. doi: 10.3390/microorganisms12091871
- Yi L, Bai Y, Chen X, et al. Synergistic effects and mechanisms of action of rutin with conventional antibiotics against Escherichia coli. Int J Mol Sci. 2024;25:13684. doi: 10.3390/ijms252413684
- Jiamboonsri P, Eurtivong C, Wanwong S. Assessing the potential of gallic acid and methyl gallate to enhance the efficacy of β-lactam antibiotics against methicillin-resistant Staphylococcus aureus by targeting β-lactamase: In silico and in vitro studies. Antibiotics (Basel). 2023;12(11):1622. doi: 10.3390/antibiotics12111622
- Lima VN, Oliveira-Tintino CDM, Santos ES, et al. Antimicrobial and enhancement of the antibiotic activity by phenolic compounds: Gallic acid, caffeic acid and pyrogallol. Microb Pathog. 2016;99:56-61. doi: 10.1016/j.micpath.2016.08.004
- Kang J, Liu L, Liu M, Wu X, Li J. Antibacterial activity of gallic acid against Shigella flexneri and its effect on biofilm formation by repressing mdoH gene expression. Food Control. 2018;94:147-154. doi: 10.1016/j.foodcont.2018.07.011
- Chew YL, Arasi C, Goh JK. Pyrogallol induces antimicrobial effect and cell membrane disruption on methicillin-resistant Staphylococcus aureus (MRSA). Curr Bioact Compd. 2021;18:38-46. doi: 10.2174/1573407217666210526121512

