Advancements in hydrogel-based design and applications of hair follicle organoids

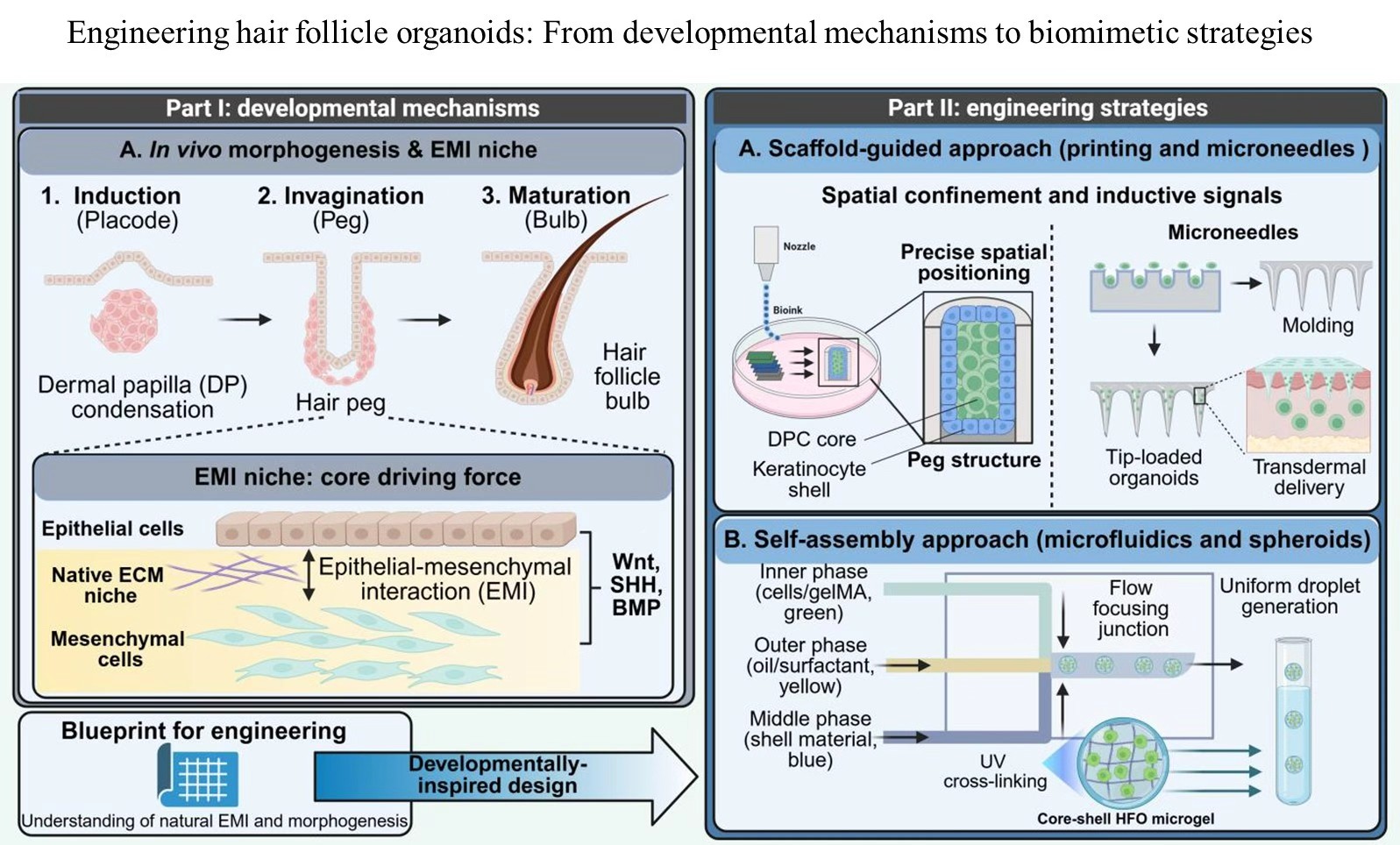
Alopecia is globally recognized as a formidable therapeutic challenge, impacting both physiological health and psychological well-being. The crux of effective treatment lies in achieving de novo hair follicle neogenesis, a process that transcends merely stimulating existing follicles. Research into hair follicle organoids (HFOs) is advancing rapidly, transitioning from rudimentary self-assembly models toward high-fidelity, clinical application-driven paradigms. To provide a rigorous synthesis of the current landscape, this review conducted a systematic literature search across the Web of Science, PubMed, and Google Scholar databases. The search strategy utilized combinations of key terms, including “hair follicle organoid,” “alopecia,” “hydrogel,” and “3D bioprinting,” spanning the last 15 years. We comprehensively summarized the design principles and recent breakthroughs in HFO technology. First, the biological foundations of hair follicle development and the specific requirements of its inductive microenvironment were elucidated. Subsequently, we highlighted design strategies for functionalized hydrogels to simulate the hair follicle niche. This included a detailed discussion on modulating physicochemical properties and integrating advanced manufacturing technologies, such as three-dimensional bioprinting. Finally, the potential of HFOs in high-throughput drug screening and complex wound repair was assessed. By serving as a robust, human-relevant in vitro model, HFOs can significantly reduce reliance on animal testing and accelerate the discovery of hair-growth-promoting compounds. By providing both theoretical frameworks and technical insights, this review aims to support the development of high-performance hair follicle regeneration platforms and accelerate their transition from laboratory research to clinical translation.
- Kang MS, Park TE, Jo HJ, et al. Recent Trends in Macromolecule-Based Approaches for Hair Loss Treatment. Macromol Biosci. 2023;23(10):e2300148. doi: 10.1002/mabi.202300148
- Adil A, Godwin M. The effectiveness of treatments for androgenetic alopecia: A systematic review and meta-analysis. J Am Acad Dermatol. 2017;77(1):136-141.e5. doi: 10.1016/j.jaad.2017.02.054
- Hunt N, McHale S. The psychological impact of alopecia. BMJ. 2005;331(7522):951-953. doi: 10.1136/bmj.331.7522.951
- Villani A, Fabbrocini G, Ocampo-Candiani J, Ruggiero A, Ocampo-Garza SS. Review of oral minoxidil as treatment of hair disorders: in search of the perfect dose. J Eur Acad Dermatol Venereol. 2021;35(7):1485-1492. doi: 10.1111/jdv.17216
- Zhou H, Zhang YX, Lei QK, Li YM, Zheng YW. Causes and therapeutic limitations of clinical alopecia and the advent of human pluripotent stem cell follicular transplantation. Stem Cell Res Ther. 2025;16(1):338. doi: 10.1186/s13287-025-04447-7
- Zheng W, Xu CH. Innovative Approaches and Advances for Hair Follicle Regeneration. ACS Biomater Sci Eng. 2023;9(5):2251-2276. doi: 10.1021/acsbiomaterials.3c00028
- Andl T, Zhou L, Zhang Y. The dermal papilla dilemma and potential breakthroughs in bioengineering hair follicles. Cell Tissue Res. 2023;391(2):221-233. doi: 10.1007/s00441-022-03730-w
- Wang M, Wang M, Jiang J, et al. THSD4 promotes hair growth by facilitating dermal papilla and hair matrix interactions. Theranostics. 2025;15(8):3571-3588. doi: 10.7150/thno.103221
- Cao X, Lu M, Li N, Cai L, Wang Y, Zhao Y. Emerging biomedical engineering strategies for hair follicle regeneration. Bioact Mater. 2025;53:84-113. doi: 10.1016/j.bioactmat.2025.06.051
- Motter Catarino C, Cigaran Schuck D, Dechiario L, Karande P. Incorporation of hair follicles in 3D bioprinted models of human skin. Sci Adv. 2023;9(41):eadg0297. doi: 10.1126/sciadv.adg0297
- Kageyama T, Shimizu A, Anakama R, et al. Reprogramming of three-dimensional microenvironments for in vitro hair follicle induction. Sci Adv. 2022;8(42):eadd4603. doi: 10.1126/sciadv.add4603
- Zhu M, Zhang H, Zhou Q, et al. Dynamic GelMA/DNA Dual-Network Hydrogels Promote Woven Bone Organoid Formation and Enhance Bone Regeneration. Adv Mater. 2025;37(24):e2501254. doi: 10.1002/adma.202501254
- Li X, Li X, Yang J, et al. Living and Injectable Porous Hydrogel Microsphere with Paracrine Activity for Cartilage Regeneration. Small. 2023;19(17):e2207211. doi: 10.1002/smll.202207211
- Yuan H, Xiao P, Huang W, Cui W. Supramolecular Lubricating Hydrogel Microspheres Reshape Damaged Matrix Regeneration. Adv Sci (Weinh). 2025;12(37):e04319. doi: 10.1002/advs.202504319
- Saiding Q, Cai Z, Deng L, Cui W. Inflammation Self-Limiting Electrospun Fibrous Tape via Regional Immunity for Deep Soft Tissue Repair. Small. 2022;18(39):e2203265. doi: 10.1002/smll.202203265
- Chen Y, He W, Ling S, et al. Boosting ribosomal translation via ionizable lipid-hydrogel microplexes for localized mRNA therapy. Bioact Mater. 2026;59:678-696. doi: 10.1016/j.bioactmat.2026.01.007
- Schneider MR, Schmidt-Ullrich R, Paus R. The Hair Follicle as a Dynamic Miniorgan. Curr Biol. 2009;19(3):R132-R142. doi: 10.1016/j.cub.2008.12.005
- Krause K, Foitzik K. Biology of the hair follicle: the basics. Semin Cutan Med Surg. 2006;25(1):2-10. doi: 10.1016/j.sder.2006.01.002
- Qian S, Mao J, Liu Z, et al. Stem cells for organoids. Smart Med. 2022;1(1):e20220007. doi: 10.1002/smmd.20220007
- Mao MQ, Jing J, Miao YJ, Lv ZF. Epithelial-Mesenchymal Interaction in Hair Regeneration and Skin Wound Healing. Front Med (Lausanne). 2022;9:863786. doi: 10.3389/fmed.2022.863786
- Enshell-Seijffers D, Lindon C, Kashiwagi M, Morgan BA. beta-catenin activity in the dermal papilla regulates morphogenesis and regeneration of hair. Dev Cell. 2010;18(4):633-642. doi: 10.1016/j.devcel.2010.01.016
- Liu Y, Guerrero-Juarez CF, Xiao F, et al. Hedgehog signaling reprograms hair follicle niche fibroblasts to a hyper-activated state. Dev Cell. 2022;57(14):1758-1775.e7. doi: 10.1016/j.devcel.2022.06.005
- Fuchs E. The tortoise and the hair: slow-cycling cells in the stem cell race. Cell. 2009;137(5):811-819. doi: 10.1016/j.cell.2009.05.002
- Xu H, Lyu X, Yi M, Zhao W, Song Y, Wu K. Organoid technology and applications in cancer research. J Hematol Oncol. 2018;11(1):116. doi: 10.1186/s13045-018-0662-9
- Lee J, Bӧscke R, Tang PC, Hartman BH, Heller S, Koehler KR. Hair Follicle Development in Mouse Pluripotent Stem Cell-Derived Skin Organoids. Cell Rep. 2018;22(1):242-254. doi: 10.1016/j.celrep.2017.12.007
- Kang MS, Kwon M, Lee SH, et al. 3D Printing of Skin Equivalents with Hair Follicle Structures and Epidermal- Papillary-Dermal Layers Using Gelatin/Hyaluronic Acid Hydrogels. Chem Asian J. 2022;17(18):e202200620. doi: 10.1002/asia.202200620
- Liu RM, Sun RG, Zhang LT, et al. Hyaluronic acid enhances proliferation of human amniotic mesenchymal stem cells through activation of Wnt/β-catenin signaling pathway. Exp Cell Res. 2016;345(2):218-229. doi: 10.1016/j.yexcr.2016.05.019
- Huang YC, Chan CC, Lin WT, et al. Scalable production of controllable dermal papilla spheroids on PVA surfaces and the effects of spheroid size on hair follicle regeneration. Biomaterials. 2013;34(2):442-451. doi: 10.1016/j.biomaterials.2012.09.083
- Ed Demri Y, Goudot G, del Giudice C, et al. Multiple peripheral arterial pseudoaneurysms associated with vascular tuberculosis. J Med Vasc. 2021;46(1):35-41. doi: 10.1016/j.jdmv.2020.10.126
- Sparks HD, Anjum F, Vallmajo-Martin Q, et al. Flowable Polyethylene Glycol Hydrogels Support the in Vitro Survival and Proliferation of Dermal Progenitor Cells in a Mechanically Dependent Manner. ACS Biomater Sci Eng. 2019;5(2):950-958. doi: 10.1021/acsbiomaterials.8b01294
- Chen Y, Wang CC, Ma YS, Chao YY, Tseng CL, Wei Y. Study on Keratin/PEGDA Composite Hydrogel with the Addition of Varied Hair Protein Fractions. ACS Appl Polym Mater. 2022;4(5):3426-3437. doi: 10.1021/acsapm.2c00045
- Dong C, Lv Y. Application of Collagen Scaffold in Tissue Engineering: Recent Advances and New Perspectives. Polymers (Basel). 2016;8(2):42. doi: 10.3390/polym8020042
- Rogers G, Martinet N, Steinert P, et al. Cultivation of Murine Hair Follicles as Organoids in a Collagen Matrix. J Invest Dermatol. 1987;89(4):369-379. doi: 10.1111/1523-1747.ep12471760
- Kageyama T, Anakama R, Hamano S, et al. Hair Follicle Organoids Using Human iPSC-Derived Ectodermal Precursor Cells for Hair Regenerative Medicine. ACS Biomater Sci Eng. 2026;12(3):1704-1714. doi: 10.1021/acsbiomaterials.5c01780
- Rogers GE, Martinet N, Steinert P, et al. A procedure for the culture of hair follicles as functionally intact organoids. Clin Dermatol. 1988;6(4):36-41. doi: 10.1016/0738-081X(88)90064-8
- Kang D, Liu Z, Qian C, et al. 3D bioprinting of a gelatin-alginate hydrogel for tissue-engineered hair follicle regeneration. Acta Biomater. 2023;165:19-30. doi: 10.1016/j.actbio.2022.03.011
- de Souza JR, Rahimnejad M, Soares IPM, et al. 3D Printing β-TCP-laden GelMA/Alginate Interpenetrating- Polymer-Network Biomaterial Inks for Bone Tissue Engineering. Bioprinting. 2025;49:e00413. doi: 10.1016/j.bprint.2025.e00413
- Li J, Moeinzadeh S, Kim C, et al. Development and systematic characterization of GelMA/alginate/PEGDMA/ xanthan gum hydrogel bioink system for extrusion bioprinting. Biomaterials. 2023;293:121969. doi: 10.1016/j.biomaterials.2022.121969
- Srinualchai P, Khorporn T, Sriboonaied P, Thongnuek P. Development of Hair Follicle Dermal Papilla Spheroids Encapsulated in Alginate Hydrogel with Extracellular Matrix Protein. BMEiCON 2024. 2024:1-5. doi: 10.1109/BMEiCON64021.2024.10896310
- Aranaz I, Alcántara AR, Civera MC, et al. Chitosan: An Overview of Its Properties and Applications. Polymers (Basel). 2021;13(19):3256. doi: 10.3390/polym13193256
- Li YCE, Wang JH, Wang YH, Shao HJ, Young LC, Young TH. PCL-Blended Chitosan Substrates for Patterning the Heterotypic Cell Distribution in an Epithelial and Mesenchymal Coculture System. ACS Biomater Sci Eng. 2020;6(7):4225-4235. doi: 10.1021/acsbiomaterials.0c00304
- Yao B, Wang Y, Zhu D, Hu T, Huang S, Zhu P. 3D-printed degradable hair follicular hanging-drop scaffolds integrated with tissue-engineered skin promote hair follicle regeneration in vitro. Int J Bioprint. 2025;11(2):8535. doi: 10.36922/ijb.8535
- Chen Y, Hou Y, Chen J, et al. Construction of Large-Scale Bioengineered Hair Germs and In Vivo Transplantation. Adv Sci (Weinh). 2025;12(16):2416361. doi: 10.1002/advs.202416361
- Huang G, Chen L. Biological activity of biomimetic dermal papilla spheres prepared by culture of dermal papilla cells of mice based on hanging drops of gelatin methacrylate and its hair-inducing function in nude mice. Chin J Burns Wounds. 2021;37(8):770-780. doi: 10.3760/cma.j.cn501120-20210330-00109
- Sachs D, Jakob R, Restivo G, et al. A quadriphasic mechanical model of the human dermis. Biomech Model Mechanobiol. 2024;23(4):1121-1136. doi: 10.1007/s10237-024-01827-5
- Chen D, Jarrell A, Guo C, Lang R, Atit R. Dermal β-catenin activity in response to epidermal Wnt ligands is required for fibroblast proliferation and hair follicle initiation. Development. 2012;139(8):1522-1533. doi: 10.1242/dev.076463
- Qu X, Xie Z, Zhang J, et al. Regulating Mitochondrial Aging via Targeting the Gut-Bone Axis in BMSCs With Oral Hydrogel Microspheres to Inhibit Bone Loss. Small. 2025;21(4):e2409936. doi: 10.1002/smll.202409936
- Bai L, Han Q, Han Z, et al. Stem Cells Expansion Vector via Bioadhesive Porous Microspheres for Accelerating Articular Cartilage Regeneration. Adv Healthc Mater. 2024;13(3):e2302327. doi: 10.1002/adhm.202302327
- Millar SE. Molecular mechanisms regulating hair follicle development. J Invest Dermatol. 2002;118(2):216-225. doi: 10.1046/j.0022-202x.2001.01670.x
- Ji S, Li Y, Xiang L, et al. Cocktail Cell-Reprogrammed Hydrogel Microspheres Achieving Scarless Hair Follicle Regeneration. Adv Sci (Weinh). 2024;11(12):e2306305. doi: 10.1002/advs.202306305
- Li C, Ma Z, Sun X, et al. Activation of bone tumor-eating macrophages via assembling and co-delivering 3D printed scaffold. Biomaterials. 2026;324:123495. doi: 10.1016/j.biomaterials.2025.123495
- Zhang P, Li L, Yan C, et al. 3D printed scaffolds regulated by neural-bone metabolic coupling promote bone unit regeneration. Bioact Mater. 2026;57:284-304. doi: 10.1016/j.bioactmat.2025.10.031
- Hong ZX, Zhu ST, Li H, et al. Bioengineered skin organoids: from development to applications. Mil Med Res. 2023;10(1):40. doi: 10.1186/s40779-023-00475-7
- Ma X, Zhu X, Lv S, et al. 3D bioprinting of prefabricated artificial skin with multicomponent hydrogel for skin and hair follicle regeneration. Theranostics. 2025;15(7):2933- 2950. doi: 10.7150/thno.104854
- Abaci HE, Coffman A, Doucet Y, et al. Tissue engineering of human hair follicles using a biomimetic developmental approach. Nat Commun. 2018;9(1):5301. doi: 10.1038/s41467-018-07579-y
- Aliyazdi S, Frisch S, Neu T, et al. A Novel 3D Printed Model of Infected Human Hair Follicles to Demonstrate Targeted Delivery of Nanoantibiotics. ACS Biomater Sci Eng. 2024;10(8):4947-4957. doi: 10.1021/acsbiomaterials.4c00570
- Li J, Fu S, Lu KW, et al. Engineering functional skin constructs: A quantitative comparison of three-dimensional bioprinting with traditional methods. Exp Dermatol. 2022;31(4):516- 527. doi: 10.1111/exd.14488
- Huang J, Fu D, Wu X, et al. One-step generation of core-shell biomimetic microspheres encapsulating double-layer cells using microfluidics for hair regeneration. Biofabrication. 2023;15(2):025007. doi: 10.1088/1758-5090/acb107
- Li J, Ma J, Cao R, et al. A skin organoid-based infection platform identifies an inhibitor specific for HFMD. Nat Commun. 2025;16(1):2513. doi: 10.1038/s41467-025-57610-2
- Brusatin G, Panciera T, Gandin A, Citron A, Piccolo S. Biomaterials and engineered microenvironments to control YAP/TAZ-dependent cell behaviour. Nat Mater. 2018;17(12):1063-1075. doi: 10.1038/s41563-018-0180-8
- Yang C, Zhang L, Zhou A, et al. Cryomicroneedle Arrays for Biotherapeutics Delivery. Small Sci. 2025;5(8):2500009. doi: 10.1002/smsc.202500009
- Kageyama T, Anakama R, Togashi H, Fukuda J. Impacts of manipulating cell sorting on in vitro hair follicle regeneration. J Biosci Bioeng. 2022;134(6):534-540. doi: 10.1016/j.jbiosc.2022.09.004
- Hu Z, Qian S, Liao B, et al. Apoptotic Vesicle Membrane- Mediated Targeted Endothelial Mitochondrial Transplantation-Clearance Therapy for Diabetic Wound Healing. Research. 2025;9:1042. doi: 10.34133/research.1042
- Yi W, Han X, Wang F, et al. Acousto-Electric Conversion Fiber Networks via Regional Activation of Schwann Cell-Derived Exosomes for Neurogenic Bone Regeneration. Research (Wash D C). 2025;8:0769. doi: 10.34133/research.0769
- Qian S, Dai S, Guo C, et al. Apoptotic Bodies Restore NAD and Mitochondrial Homeostasis in Fibroblasts. Adv Sci (Weinh). 2025;12(29):e15691. doi: 10.1002/advs.202415691
- Nilforoushzadeh MA, Aghdami N, Taghiabadi E. Human Hair Outer Root Sheath Cells and Platelet-Lysis Exosomes Promote Hair Inductivity of Dermal Papilla Cell. Tissue Eng Regen Med. 2020;17(4):525-536. doi: 10.1007/s13770-020-00266-4
- Hu Z, Qian S, Zhao Q, et al. Engineering strategies for apoptotic bodies. Smart Med. 2024;3(3):e20240005. doi: 10.1002/SMMD.20240005
- Zhao Q, Lu B, Qian S, et al. Biogenerated Oxygen- Related Environmental Stressed Apoptotic Vesicle Targets Endothelial Cells. Adv Sci (Weinh). 2024;11(20):e2306555. doi: 10.1002/advs.202306555
- Li J, Zhao B, Dai Y, Zhang X, Chen Y, Wu X. Exosomes Derived from Dermal Papilla Cells Mediate Hair Follicle Stem Cell Proliferation through the Wnt3a/β-Catenin Signaling Pathway. Oxid Med Cell Longev. 2022;2022:9042345. doi: 10.1155/2022/9042345
- Zhou L, Wang H, Jing J, Yu L, Wu X, Lu Z. Regulation of hair follicle development by exosomes derived from dermal papilla cells. Biochem Biophys Res Commun. 2018;500(2):325- 332. doi: 10.1016/j.bbrc.2018.04.067
- Shimizu Y, Ntege EH, Sunami H, Inoue Y. Regenerative medicine strategies for hair growth and regeneration: A narrative review of literature. Regen Ther. 2022;21:527-539. doi: 10.1016/j.reth.2022.10.005
- Kim H, Jang Y, Kim EH, et al. Potential of Colostrum-Derived Exosomes for Promoting Hair Regeneration Through the Transition From Telogen to Anagen Phase. Front Cell Dev Biol. 2022;10:815205. doi: 10.3389/fcell.2022.815205
- Lei M, Guo H, Qiu W, et al. Modulating hair follicle size with Wnt10b/DKK1 during hair regeneration. Exp Dermatol. 2014;23(6):407-413. doi: 10.1111/exd.12416
- Papukashvili D, Rcheulishvili N, Liu C, et al. Perspectives on miRNAs Targeting DKK1 for Developing Hair Regeneration Therapy. Cells. 2021;10(11):2957. doi: 10.3390/cells10112957
- Mahmoud EA, Elgarhy LH, Hasby EA, Mohammad L. Dickkopf-1 Expression in Androgenetic Alopecia and Alopecia Areata in Male Patients. Am J Dermatopathol. 2019;41(2):122-127. doi: 10.1097/dad.0000000000001266
- Kageyama T, Seo J, Yan L, Fukuda J. Cinnamic acid promotes elongation of hair peg-like sprouting in hair follicle organoids via oxytocin receptor activation. Sci Rep. 2024;14(1):4709. doi: 10.1038/s41598-024-55377-y
- Kageyama T, Miyata H, Seo J, Nanmo A, Fukuda J. In vitro hair follicle growth model for drug testing. Sci Rep. 2023;13(1):4847. doi: 10.1038/s41598-023-31842-y

