Nanomaterials in organoid research: Advancements and applications

In recent years, organoids have demonstrated immense potential as three-dimensional in vitro models of human tissue, capable of simulating physiological and pathological environments. Nanomaterials, owing to their unique physicochemical properties, have become pivotal tools in advancing organoid research and applications. By integrating nanomaterials, scientists can more effectively explore disease mechanisms, optimize drug delivery systems, and develop novel therapeutic strategies. For example, nanoparticle-based metabolic reprogramming techniques offer novel avenues for regulating organ function, while advances in gene editing technologies have markedly enhanced the precision and complexity of organoid models. Furthermore, organoids demonstrate broad application prospects in fields such as virology research, cardiac tissue engineering, and cancer therapy. These advances not only propel fundamental scientific inquiry but also provide crucial support for clinical translation. The future holds promise for achieving more precise, personalized medical solutions through the integration of nanotechnology and organoid models.
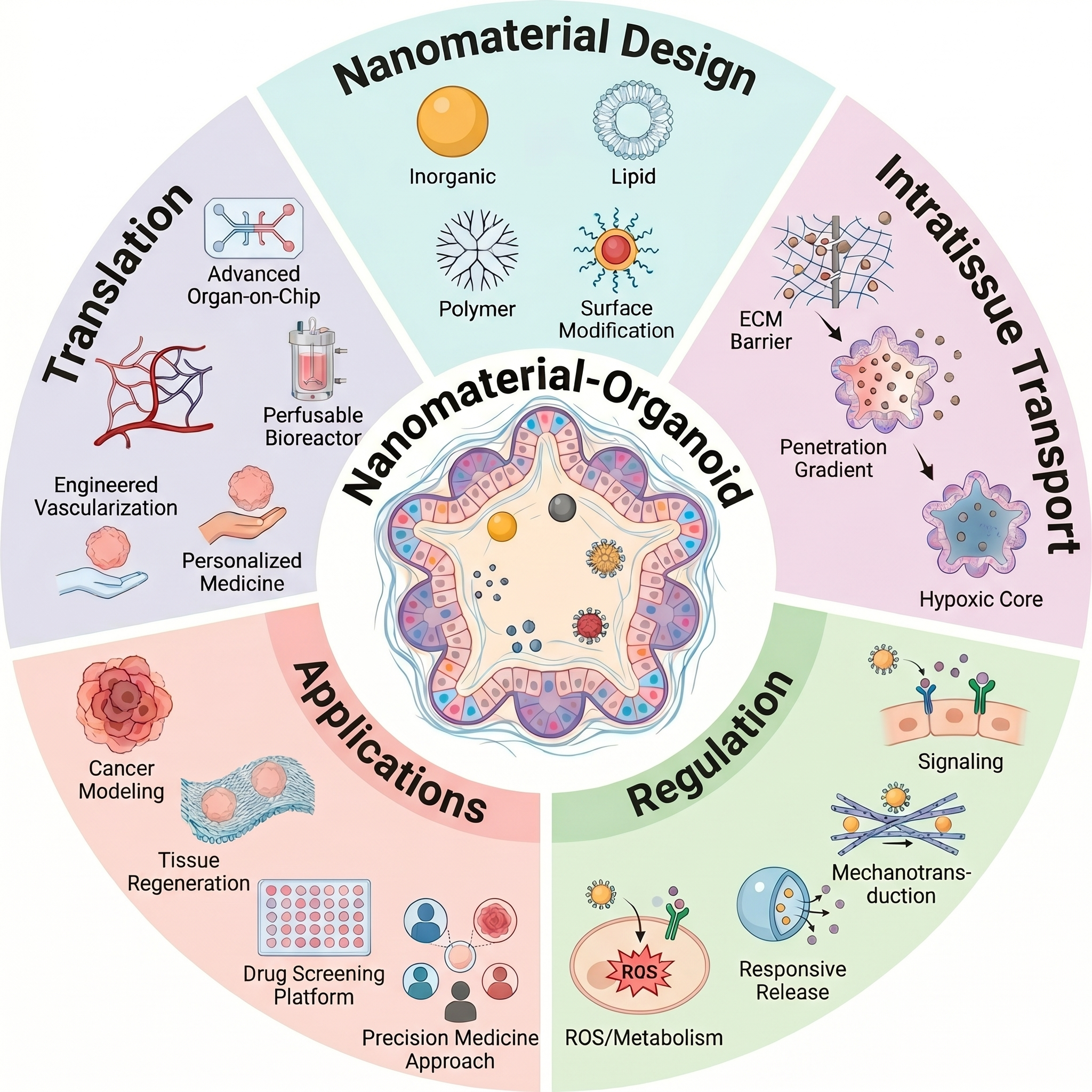
- Schutgens F, Clevers H. Human organoids: Tools for understanding biology and treating diseases. Annu Rev Pathol. 2020;15:211-234. doi: 10.1146/annurev-pathmechdis-012419-032611
- Lancaster MA, Knoblich JA. Organogenesis in a dish: modeling development and disease using organoid technologies. Science. 2014;345(6194):1247125. doi: 10.1126/science.1247125
- Clevers H. Modeling development and disease with organoids. Cell. 2016;165(7):1586-1597. doi: 10.1016/j.cell.2016.05.082
- Kapałczyńska M, Kolenda T, Przybyła W, et al. 2D and 3D cell cultures - a comparison of different types of cancer cell cultures. Arch Med Sci. 2018;14(4):910-919. doi: 10.5114/aoms.2016.63743
- Zhou LB, Lei YT, Han X. Charting bioethical frontiers: China’s human organoid guidelines in a global context. Mil Med Res. 2025;12(1):64. doi: 10.1186/s40779-025-00651-x
- Abdel Fattah AR, Ranga A. Nanoparticles as versatile tools for mechanotransduction in tissues and organoids. Front Bioeng Biotechnol. 2020;8:240. doi: 10.3389/fbioe.2020.00240
- Fei K, Zhang J, Yuan J, Xiao P. Present application and perspectives of organoid imaging technology. Bioengineering (Basel). 2022;9(3):121. doi: 10.3390/bioengineering9030121
- Pomerantseva E, Bonaccorso F, Feng X, Cui Y, Gogotsi Y. Energy storage: The future enabled by nanomaterials. Science. 2019;366(6468):eaan8285. doi: 10.1126/science.aan8285
- Shen C, Zhang ZJ, Li XX, et al. Intersection of nanomaterials and organoids technology in biomedicine. Front Immunol. 2023;14:1172262. doi: 10.3389/fimmu.2023.1172262
- Mahapatra C, Lee R, Paul MK. Emerging role and promise of nanomaterials in organoid research. Drug Discov Today. 2022;27(3):890-899. doi: 10.1016/j.drudis.2021.11.007
- Pampaloni F, Reynaud EG, Stelzer EH. The third dimension bridges the gap between cell culture and live tissue. Nat Rev Mol Cell Biol. 2007;8(10):839-845. doi: 10.1038/nrm2236
- Badir A, Refki S, Sekkat Z. Utilizing gold nanoparticles in plasmonic photothermal therapy for cancer treatment. Heliyon. 2025;11(4):e42738. doi: 10.1016/j.heliyon.2025.e42738
- Narayan R, Nayak UY, Raichur AM, Garg S. Mesoporous silica nanoparticles: A comprehensive review on synthesis and recent advances. Pharmaceutics. 2018;10(3):118. doi: 10.3390/pharmaceutics10030118
- Vallet-Regí M, Schüth F, Lozano D, Colilla M, Manzano M. Engineering mesoporous silica nanoparticles for drug delivery: Where are we after two decades? Chem Soc Rev. 2022;51(13):5365-5451. doi: 10.1039/d1cs00659b
- Miyamoto Y, Koshidaka Y, Murase K, Kanno S, Noguchi H, Miyado K, et al. Functional evaluation of 3D liver models labeled with polysaccharide functionalized magnetic nanoparticles. Materials. 2022;15(21):7823. doi: 10.3390/ma15217823
- Choudhery MS, Umezawa A. Stem cells as biological drugs for incurable diseases. Stem Cell Res Ther. 2025;16(1):482. doi: 10.1186/s13287-025-04632-8
- Shi Y, Han X, Zou S, Liu G. Nanomaterials in organoids: From interactions to personalized medicine. ACS Nano. 2024;18(49):33276-33292. doi: 10.1021/acsnano.4c13330
- Pérez-Piñeiro J, Sánchez-Cea F, Arce MP, et al. Stability study of graphene oxide-bovine serum albumin dispersions. J Xenobiot. 2023;13(1):90-101. doi: 10.3390/jox13010008
- Shah S, Dhawan V, Holm R, Nagarsenker MS, Perrie Y. Liposomes: Advancements and innovation in the manufacturing process. Adv Drug Deliv Rev. 2020;154- 155:102-122. doi: 10.1016/j.addr.2020.07.002
- Lin L, Song X, Dong X, Li B. Nano-photosensitizers for enhanced photodynamic therapy. Photodiagnosis Photodyn Ther. 2021;36:102597.doi: 10.1016/j.pdpdt.2021.102597
- Wang H, Chen Y, Wei R, et al. Synergistic chemoimmunotherapy augmentation via sequential nanocomposite hydrogel-mediated reprogramming of cancer-associated fibroblasts in osteosarcoma. Adv Mater. 2024;36(15):e2309591. doi: 10.1002/adma.202309591
- El Moukhtari SH, Garbayo E, Amundarain A, et al. Lipid nanoparticles for siRNA delivery in cancer treatment. J Control Release. 2023;361:130-146. doi: 10.1016/j.jconrel.2023.07.054
- Dutta Gupta Y, Mackeyev Y, Krishnan S, Bhandary S. Mesoporous silica nanotechnology: promising advances in augmenting cancer theranostics. Cancer Nanotechnol. 2024;15(1):9. doi: 10.1186/s12645-024-00250-w
- Viegas C, Patrício AB, Prata J, et al. Advances in pancreatic cancer treatment by nano-based drug delivery systems. Pharmaceutics. 2023;15(9):2363. doi: 10.3390/pharmaceutics15092363
- Chiu JZ, Tucker IG, McLeod BJ, McDowell A. Arginine-tagging of polymeric nanoparticles via histidine to improve cellular uptake. Eur J Pharm Biopharm. 2015;89:48-55. doi: 10.1016/j.ejpb.2014.11.014
- Dilliard SA, Siegwart DJ. Passive, active and endogenous organ-targeted lipid and polymer nanoparticles for delivery of genetic drugs. Nat Rev Mater. 2023;8(4):282-300. doi: 10.1038/s41578-022-00529-7
- Huang H, Chang A, Peng H, et al. Preparation and anti-tumor effect in hepatocellular carcinoma treatment of AS1411 aptamer-targeted polyphyllin II-loaded PLGA nanoparticles. J Sci Adv Mater Devices. 2024;9(3):100755. doi: 10.1016/j.jsamd.2024.100755
- Shaw I, Aryee AA, Ali YS, et al. Harmonizing tradition and technology: Liposomal nanocarriers unlocking the power of natural herbs in Traditional Chinese Medicine. Chin J Nat Med. 2025;23(6):700-713. doi: 10.1016/s1875-5364(25)60889-2
- Mitchell MJ, Billingsley MM, Haley RM, Wechsler ME, Peppas NA, Langer R. Engineering precision nanoparticles for drug delivery. Nat Rev Drug Discov. 2021;20(2):101-124. doi: 10.1038/s41573-020-0090-8
- Omlor AJ, Nguyen J, Bals R, Dinh QT. Nanotechnology in respiratory medicine. Respir Res. 2015;16(1):64. doi: 10.1186/s12931-015-0223-5
- Barroso L, Viegas C, Vieira J, Ferreira-Pêgo C, Costa J, Fonte P. Lipid-based carriers for food ingredients delivery. J Food Eng. 2021;295:110451. doi: 10.1016/j.jfoodeng.2020.110451
- Bag J, Banerjee S, De A, et al. Nanoengineered approaches to improve the efficacy of targeted drug delivery for the treatment of malignancy: a comprehensive review. Futur J Pharm Sci. 2023;9(1):88. doi: 10.1186/s43094-023-00541-w
- López-Estevez AM, Gref R, Alonso MJ. A journey through the history of PEGylated drug delivery nanocarriers. Drug Deliv Transl Res. 2024;14(8):2026-2031. doi: 10.1007/s13346-024-01608-8
- Vakhshiteh F, Bagheri Z, Soleimani M, et al. Heterotypic tumor spheroids: a platform for nanomedicine evaluation. J Nanobiotechnology. 2023;21(1):249. doi: 10.1186/s12951-023-02021-y
- Lacin ME, Yildirim M. Applications of multiphoton microscopy in imaging cerebral and retinal organoids. Front Neurosci. 2024;18:1360482. doi: 10.3389/fnins.2024.1360482
- Zhao WP, Wang HW, Liu J, et al. Positive PCNA and Ki-67 expression in the testis correlates with spermatogenesis dysfunction in fluoride-treated rats. Biol Trace Elem Res. 2018;186(2):489-497. doi: 10.1007/s12011-018-1338-6
- Mohammed MA, Syeda JTM, Wasan KM, Wasan EK. An overview of chitosan nanoparticles and its application in non-parenteral drug delivery. Pharmaceutics. 2017;9(4):53. doi: 10.3390/pharmaceutics9040053
- Alves-Lopes JP, Söder O, Stukenborg JB. Use of a three-layer gradient system of cells for rat testicular organoid generation. Nat Protoc. 2018;13(2):248-259. doi: 10.1038/nprot.2017.140
- Kita K, Watanabe T, Ohsaka K, et al. Production of functional spermatids from mouse germline stem cells in ectopically reconstituted seminiferous tubules. Biol Reprod. 2007;76(2):211-217. doi: 10.1095/biolreprod.106.056895
- Dobrinski I. De novo morphogenesis of functional testis tissue after ectopic transplantation of isolated cells. Organogenesis. 2007;3(2):79-82. doi: 10.4161/org.3.2.4944
- Sato T, Clevers H. Growing self-organizing mini-guts from a single intestinal stem cell: mechanism and applications. Science. 2013;340(6137):1190-1194. doi: 10.1126/science.1234852
- Proceedings of the 17th Italian convention of FFC investigators in cystic fibrosis. BMC Pulm Med. 2020;20(1):100. doi: 10.1186/s12890-020-1104-3
- Yang T, Wang YY, Ma F, Xu BH, Qian HL. Build the virtual cell with artificial intelligence: a perspective for cancer research. Mil Med Res. 2025;12(1):4. doi: 10.1186/s40779-025-00591-6
- Kaur SD, Kapoor DN. Cancer in translation: cyanine carborane salts: a multifunctional theranostic approach to cancer treatment. EJNMMI Radiopharm Chem. 2025;10(1):36. doi: 10.1186/s41181-025-00360-9
- van Ravensteijn BGP, Vermonden T. Dynamic, tunable, biocompatible: Functional polymers advancing biomedical innovation. Biomacromolecules. 2026;27(1):1-2. doi: 10.1021/acs.biomac.5c02557
- Turpin I, Modrego A, Sarrias AM, et al. Deep learning applied to live imaging for classifying brain organoids derived from neurodevelopmental disorders considering the intrinsic variability of the in vitro model. IBRO Neurosci Rep. 2023;15:S209. doi: 10.1016/j.ibneur.2023.08.331
- Singh A, Lutolf M. Breaking the barriers in engineering organoids and tissues with advanced materials. Adv Funct Mater. 2020;30(48):2008531. doi: 10.1002/adfm.202008531
- Robinson M, Sparanese S, Witherspoon L, Flannigan R. Human in vitro spermatogenesis as a regenerative therapy - where do we stand? Nat Rev Urol. 2023;20(8):461-479. doi: 10.1038/s41585-023-00723-4
- Shankaran A, Prasad K, Chaudhari S, Brand A, Satyamoorthy K. Advances in development and application of human organoids. 3 Biotech. 2021;11(6):257. doi: 10.1007/s13205-021-02815-7
- Takasato M, Er PX, Chiu HS, et al. Kidney organoids from human iPS cells contain multiple lineages and model human nephrogenesis. Nature. 2015;526(7574):564-568. doi: 10.1038/nature15695
- Karkampouna S, La Manna F, Benjak A, et al. Patient-derived xenografts and organoids model therapy response in prostate cancer. Nat Commun. 2021;12(1):1117. doi: 10.1038/s41467-021-21300-6
- Scholler J, Groux K, Goureau O, et al. Dynamic full-field optical coherence tomography: 3D live-imaging of retinal organoids. Light Sci Appl. 2020;9:140. doi: 10.1038/s41377-020-00375-8
- Bruun J, Kryeziu K, Eide PW, et al. Patient-derived organoids from multiple colorectal cancer liver metastases reveal moderate intra-patient pharmacotranscriptomic heterogeneity. Clin Cancer Res. 2020;26(15):4107-4119. doi: 10.1158/1078-0432.CCR-19-3637
- Dorbala S, Ando Y, Bokhari S, et al. ASNC/AHA/ASE/ EANM/HFSA/ISA/SCMR/SNMMI Expert consensus recommendations for multimodality imaging in cardiac amyloidosis: Part 1 of 2-Evidence base and standardized methods of imaging. Circ Cardiovasc Imaging. 2021;14(7):e000029. doi: 10.1161/HCI.0000000000000029
- Xiang X, Pang H, Ma T, et al. Ultrasound targeted microbubble destruction combined with Fe-MOF based bio-/enzyme-mimics nanoparticles for treating of cancer. J Nanobiotechnology. 2021;19(1):92. doi: 10.1186/s12951-021-00835-2
- Achilefu S. Introduction to concepts and strategies for molecular imaging. Chem Rev. 2010;110(5):2575-2578. doi: 10.1021/cr1001113
- Yang J, Lei W, Xiao Y, et al. Generation of human vascularized and chambered cardiac organoids for cardiac disease modelling and drug evaluation. Cell Prolif. 2024;57(8):e13631. doi: 10.1111/cpr.13631
- Shi R, Li Y, Liu H, Chen J. Lung microbiota: Unexploited treasure hidden in the immune microenvironment of lung cancer. Thorac Cancer. 2021;12(21):2964-2966. doi: 10.1111/1759-7714.14159
- Taylor LS, Bergström CAS, Lavasanifar A, Qian F, Suryanarayanan R, Thurecht KJ. Celebrating women in the pharmaceutical sciences. Mol Pharm. 2021;18(4):1487-1490. doi: 10.1021/acs.molpharmaceut.0c01197
- Chen N, Brachmann C, Liu X, et al. Albumin-bound nanoparticle (nab) paclitaxel exhibits enhanced paclitaxel tissue distribution and tumor penetration. Cancer Chemother Pharmacol. 2015;76(4):699-712. doi: 10.1007/s00280-015-2833-5
- Zhang D, Jin W, Cui Y, He Z. Establishment and characterization of testis organoids with proliferation and differentiation of spermatogonial stem cells into spermatocytes and spermatids. Cells. 2024;13(19):1642. doi: 10.3390/cells13191642
- Metselaar JM, Lammers T. Challenges in nanomedicine clinical translation. Drug Deliv Transl Res. 2020;10(3):721-725. doi: 10.1007/s13346-020-00740-5
- Miller MA, Gadde S, Pfirschke C, et al. Predicting therapeutic nanomedicine efficacy using a companion magnetic resonance imaging nanoparticle. Sci Transl Med. 2015;7(314):314ra183. doi: 10.1126/scitranslmed.aac6522
- Pan B, Wei X, Xu X. Patient-derived xenograft models in hepatopancreatobiliary cancer. Cancer Cell Int. 2022;22(1):41. doi: 10.1186/s12935-022-02454-9
- Heo JH, Kang D, Seo SJ, Jin Y. Engineering the extracellular matrix for organoid culture. Int J Stem Cells. 2022;15:60-69. doi: 10.15283/ijsc21190
- Chen X. Biomedical poly(amino acid) materials. Nano Lett. 2026;26(11):3645-3648. doi: 10.1021/acs.nanolett.6c00156
- Chen X. From quantum dots to cancer nanotheranostics. Nano Lett. 2025;25(38):13967-13969. doi: 10.1021/acs.nanolett.5c03619
- Mao Y, Li C, Fan C, Schanze KS, Fernandez L. Forum on materials for brain research. ACS Appl Mater Interfaces. 2023;15(1):1-2. doi: 10.1021/acsami.2c21906
- Singh LS, Singh CS, Premlata T. Magnetically controlled nanocarriers for site-specific drug delivery and theranostics in cancer precision medicine. Mol Imaging Biol. doi: 10.1007/s11307-026-02097-6
- Becker ML, Burdick JA. Introduction: Polymeric biomaterials. Chem Rev. 2021;121(18):10789-10791. doi: 10.1021/acs.chemrev.1c00354
- Sridhar SS, Blais N, Tran B, et al. Efficacy and safety of nab-paclitaxel vs paclitaxel on survival in patients with platinum-refractory metastatic urothelial cancer: The Canadian Cancer Trials Group BL.12 Randomized Clinical Trial. JAMA Oncol. 2020;6(11):1751-1758. doi: 10.1001/jamaoncol.2020.3927
- Takebe T, Wells JM. Organoids by design. Science. 2019;364(6444):956-959. doi: 10.1126/science.aaw7567
- Hammond PT. Shooting for the moon: Nanoscale approaches to cancer. ACS Nano. 2016;10(2):1711-1713. doi: 10.1021/acsnano.6b01095
- Choi Y, Cho BK, Seok SH, Kim C, Ryu JH, Kwon IC. Controlled spatial characteristics of ligands on nanoparticles: Determinant of cellular functions. J Control Release. 2023;360:672-686. doi: 10.1016/j.jconrel.2023.07.020
- Jooken S, Deschaume O, Bartic C. Nanocomposite hydrogels as functional extracellular matrices. Gels. 2023;9(2):153. doi: 10.3390/gels9020153
- Yang Z, Jiang H. Control of hematopoietic stem cell function through epigenetic regulation of energy metabolism and genome integrity. Blood. 2017;130:772. doi: 10.1182/blood.v130.suppl_1.772.772
- Teli MK, Mutalik S, Rajanikant GK. Nanotechnology and nanomedicine: going small means aiming big. Curr Pharm Des. 2010;16(16):1882-1892. doi: 10.2174/138161210791208992
- Wang W, Cai J, Wen J, et al. Boosting ferroptosis via abplatin(iv) for treatment of platinum-resistant recurrent ovarian cancer. Nano Today. 2022;44:101459. doi: 10.1016/j.nantod.2022.101459
- van der Meel R, Sulheim E, Shi Y, Kiessling F, Mulder WJM, Lammers T. Smart cancer nanomedicine. Nat Nanotechnol. 2019;14(11):1007-1017. doi: 10.1038/s41565-019-0567-y
- Yoshida GJ. Applications of patient-derived tumor xenograft models and tumor organoids. J Hematol Oncol. 2020;13(1):4. doi: 10.1186/s13045-019-0829-z
- Zazo H, Colino CI, Lanao JM. Current applications of nanoparticles in infectious diseases. J Control Release. 2016;224:86-102. doi: 10.1016/j.jconrel.2016.01.008
- Hill DR, Spence JR. Gastrointestinal organoids: Understanding the molecular basis of the host-microbe interface. Cell Mol Gastroenterol Hepatol. 2016;3(2):138-149. doi: 10.1016/j.jcmgh.2016.11.007
- Schlaermann P, Toelle B, Berger H, et al. A novel human gastric primary cell culture system for modelling Helicobacter pylori infection in vitro. Gut. 2016;65(2):202-213. doi: 10.1136/gutjnl-2014-307949
- Stanković A, Mirković M, Radović M, et al. Abstracts from the 20th European symposium on radiopharmacy and radiopharmaceuticals. EJNMMI Radiopharm Chem. 2023;8(1):11. doi: 10.1186/s41181-023-00193-4
- Wang J, Wang F, Jiang Y, Bai L, Chen X, Su J. Organoid-driven nanomedicine platform development. Biomaterials. 2026;325:123611. doi: 10.1016/j.biomaterials.2025.123611
- Giobbe GG, Bonfante F, Jones BC, et al. SARS-CoV-2 infection and replication in human gastric organoids. Nat Commun. 2021;12(1):6610. doi: 10.1038/s41467-021-26762-2
- Chen YW, Huang SX, de Carvalho ALRT, et al. A three-dimensional model of human lung development and disease from pluripotent stem cells. Nat Cell Biol. 2017;19(5):542- 549. doi: 10.1038/ncb3510
- Li Y, Guo Y, Zhang K, et al. Cell death pathway regulation by functional nanomedicines for robust antitumor immunity. Adv Sci (Weinh). 2024;11(3):e2306580. doi: 10.1002/advs.202306580
- Allenspach K. I32 | 3-Dimensional organoid application in veterinary medicine – focus on oncology. J Vet Pharmacol Ther. 2023;46(S1):21-23. doi: 10.1111/jvp.13162
- Sun M, Lee J, Chen Y, Hoshino K. Studies of nanoparticle delivery with in vitro bio-engineered microtissues. Bioact Mater. 2020;5(4):924-937. doi: 10.1016/j.bioactmat.2020.06.016
- Wang Y, Jeon H. 3D cell cultures toward quantitative high-throughput drug screening. Trends Pharmacol Sci. 2022;43(7):569-581. doi: 10.1016/j.tips.2022.03.014
- Shapiro RL, DeLong K, Zulfiqar F, Carter D, Better M, Ensign LM. In vitro and ex vivo models for evaluating vaginal drug delivery systems. Adv Drug Deliv Rev. 2022;191:114543. doi: 10.1016/j.addr.2022.114543
- Palma M, Qi B. Advancing phage therapy: A comprehensive review of the safety, efficacy, and future prospects for the targeted treatment of bacterial infections. Infect Dis Rep. 2024;16(6):1127-1181. doi: 10.3390/idr16060092
- Hu T, Brinker CJ, Chan WCW, et al. Publishing translational research of nanomedicine in ACS Nano. ACS Nano. 2022;16(11):17479-17481. doi: 10.1021/acsnano.2c10967
- Liang W, Zhou C, Liu X, et al. Current status of nano-embedded growth factors and stem cells delivery to bone for targeted repair and regeneration. J Orthop Translat. 2025;50:257-273. doi: 10.1016/j.jot.2024.12.006
- Abbasalizadeh S, Larijani MR, Samadian A, Baharvand H. Bioprocess development for mass production of size-controlled human pluripotent stem cell aggregates in stirred suspension bioreactor. Tissue Eng Part C Methods. 2012;18(11):831-851. doi: 10.1089/ten.TEC.2012.0161
- Carvalho DJ, Kip AM, Tegel A, et al. A modular microfluidic organoid platform using LEGO-like bricks. Adv Healthc Mater. 2024;13(13):e2303444. doi: 10.1002/adhm.202303444
- Sakamoto JH, van de Ven AL, Godin B, et al. Enabling individualized therapy through nanotechnology. Pharmacol Res. 2010;62(2):57-89. doi: 10.1016/j.phrs.2009.12.011
- Yang W, Liu H, Liu X, et al. Biomimetic fucoidan nanoparticles with regulation of macrophage polarization for targeted therapy of acute lung injury. Carbohydr Polym. 2025;351:122967. doi: 10.1016/j.carbpol.2024.122967
- Fancher IS, Rubinstein I, Levitan I. Potential strategies to reduce blood pressure in treatment-resistant hypertension using Food and Drug Administration-approved nanodrug delivery platforms. Hypertension. 2019;73(2):250-257. doi: 10.1161/HYPERTENSIONAHA.118.12005
- Ju S, Mu J, Dokland T, et al. Grape exosome-like nanoparticles induce intestinal stem cells and protect mice from DSS-induced colitis. Mol Ther. 2013;21(7):1345-1357. doi: 10.1038/mt.2013.64
- Smith TT, Stephan SB, Moffett HF, et al. In situ programming of leukaemia-specific T cells using synthetic DNA nanocarriers. Nat Nanotechnol. 2017;12(8):813-820. doi: 10.1038/nnano.2017.57
- Abdel Fattah AR, Kolaitis N, Van Daele K, Daza B, Rustandi AG, Ranga A. Targeted mechanical stimulation via magnetic nanoparticles guides in vitro tissue development. Nat Commun. 2023;14(1):5281. doi: 10.1038/s41467-023-41037-8
- Ye L, Jiang S, Hu J, et al. Induction of metabolic reprogramming in kidney by singlet diradical nanoparticles. Adv Mater. 2023;35(36):e2301338. doi: 10.1002/adma.202301338
- Tang XY, Wu S, Wang D, et al. Human organoids in basic research and clinical applications. Signal Transduct Target Ther. 2022;7(1):168. doi: 10.1038/s41392-022-01024-9
- Moradi S, Mahdizadeh H, Šarić T, et al. Research and therapy with induced pluripotent stem cells (iPSCs): social, legal, and ethical considerations. Stem Cell Res Ther. 2019;10(1):341. doi: 10.1186/s13287-019-1455-y
- Sean G, Banes AJ, Gangaraju R. Organoids and tissue/organ chips. Stem Cell Res Ther. 2024;15(1):241. doi: 10.1186/s13287-024-03859-1
- Hong Y, Gao D, Zhao B, Ma J, Yang Z, Guo H. Thermal immuno-nanomedicine: A new strategy for cancer treatment. Clin Transl Med. 2023;13(5):e1256. doi: 10.1002/ctm2.1256
- Patton J, Smith E, Smith Z, et al. Characterization of cytotoxic macrophages in a pre-clinical model of Epstein- Barr Virus (EBV)-driven lymphoproliferative disease. Blood. 2021;138:3502. doi: 10.1182/blood-2021-154161
- Jin H, Xue Z, Liu J, Ma B, Yang J, Lei L. Advancing organoid engineering for tissue regeneration and biofunctional reconstruction. Biomater Res. 2024;28:0016. doi: 10.34133/bmr.0016
- Di X, Gao X, Peng L, et al. Cellular mechanotransduction in health and diseases: from molecular mechanism to therapeutic targets. Signal Transduct Target Ther. 2023;8(1):282. doi: 10.1038/s41392-023-01501-9
- Chhabra A, Song HG, Grzelak KA, et al. A vascularized model of the human liver mimics regenerative responses. Proc Natl Acad Sci USA. 2022;119(28):e2115867119. doi: 10.1073/pnas.2115867119
- Chooi WH, Ng CY, Ow V, et al. Defined alginate hydrogels support spinal cord organoid derivation, maturation, and modeling of spinal cord diseases. Adv Healthc Mater. 2023;12(9):e2202342. doi: 10.1002/adhm.202202342
- Ohnsorg ML, Hushka EA, Anseth KS. Photoresponsive chemistries for user-directed hydrogel network modulation to investigate cell-matrix interactions. Acc Chem Res. 2025;58(1):47-60. doi: 10.1021/acs.accounts.4c00548
- Thang NH, Chien TB, Cuong DX. Polymer-based hydrogels applied in drug delivery: An overview. Gels. 2023;9(7):523. doi: 10.3390/gels9070523
- Singh R, Mori N, Kida YS. Methodological innovations in perfusable vascular networks: Advancing high-density tissue models. Regen Ther. 2026;32:101109. doi: 10.1016/j.reth.2026.101109
- Kaur N, Yang D, Ali J. Abstract 1292: Innovative 3D ECM platforms for tumoroid development, drug screening, and personalized treatment. Cancer Res. 2025;85(5_Suppl):1292. doi: 10.1158/1538-7445.am2025-1292
- Zhou R, Brislinger D, Fuchs J, et al. Vascularised organoids: Recent advances and applications in cancer research. Clin Transl Med. 2025;15(3):e70258. doi: 10.1002/ctm2.70258
- Li G, Zhou D, Sheng S, et al. Hydrogel for bone microenvironment: Strategy and application. Chem Eng J. 2024;499:156554. doi: 10.1016/j.cej.2024.156554
- Luce E, Duclos-Vallee JC. Stem cells and organoids: A paradigm shift in preclinical models toward personalized medicine. Pharmaceuticals (Basel). 2025;18(7):992. doi: 10.3390/ph18070992
- András IE, Cho HJ, Toborek M. Abstracts from the International Symposium “Signal Transduction at the Blood– Brain Barriers” 2022. Fluids Barriers CNS. 2023;20(1):26. doi: 10.1186/s12987-023-00417-4
- Ma W, Dong Z, Zheng Z, Bai L, Zhang X, Su J. Multiscale construction, evaluation, and application of organoids. Adv Sci (Weinh). 2025;12(47):e08534. doi: 10.1002/advs.202508534
- Vallmajo-Martin Q, Broguiere N, Millan C, Zenobi-Wong M, Ehrbar M. PEG/HA hybrid hydrogels for biologically and mechanically tailorable bone marrow organoids. Adv Funct Mater. 2020;30(48):1910282. doi: 10.1002/adfm.201910282
- Joddar B, Natividad-Diaz SL, Padilla AE, et al. Engineering approaches for cardiac organoid formation and their characterization. Transl Res. 2022;250:46-67. doi: 10.1016/j.trsl.2022.08.009
- Shaw CA, Mortimer GM, Deng ZJ, et al. Protein corona formation in bronchoalveolar fluid enhances diesel exhaust nanoparticle uptake and pro-inflammatory responses in macrophages. Nanotoxicology. 2016;10(7):981-991. doi: 10.3109/17435390.2016.1155672
- Wang C, Ma X. The role of acetylation and deacetylation in cancer metabolism. Clin Transl Med. 2025;15(1):e70145. doi: 10.1002/ctm2.70145
- Bilardo R, Traldi F, Vdovchenko A, Resmini M. Influence of surface chemistry and morphology of nanoparticles on protein corona formation. Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2022;14(4):e1788. doi: 10.1002/wnan.1788
- Chen C, Leong D, Lynch I. Rethinking nanosafety Part II: Leveraging progress to pioneer new approaches and solutions. Small. 2020;16(36):2004934. doi: 10.1002/smll.202004934
- Ghosh S, Pati F. Decellularized extracellular matrix and silk fibroin-based hybrid biomaterials: A comprehensive review on fabrication techniques and tissue-specific applications. Int J Biol Macromol. 2023;253(Pt 8):127410. doi: 10.1016/j.ijbiomac.2023.127410
- Khodayari H, Khodayari S, Rezaee M, et al. Promotion of cardiac microtissue assembly within G-CSF-enriched collagen I-cardiogel hybrid hydrogel. Regen Biomater. 2024;11:rbae072. doi: 10.1093/rb/rbae072
- Khetani SR, Berger DR, Ballinger KR, Davidson MD, Lin C, Ware BR. Microengineered liver tissues for drug testing. SLAS Technol. 2015;20(3):216-250. doi: 10.1177/2211068214566939
- Zhao R, Feng Q, Xia Y, Liao L, Xie S. Iteration of tumor organoids in drug development: Simplification and integration. Pharmaceuticals. 2025;18(10):1540. doi: 10.3390/ph18101540
- Jun HR, Chang SY, Kim H, et al. Abstract 5199: Enhancing precision medicine in advanced lung, gastric and rectal cancer: Exploring the reliability of Patient-derived organoids. Cancer Res. 2025;85(5_Suppl):5199. doi: 10.1158/1538-7445.am2025-5199
- Vives J, Batlle-Morera L. The challenge of developing human 3D organoids into medicines. Stem Cell Res Ther. 2020;11(1):72. doi: 10.1186/s13287-020-1586-1
- Sato T, Vries RG, Snippert HJ, et al. Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature. 2009;459(7244):262-265. doi: 10.1038/nature07935
- Franco C, Rezzani R. Methods and models for studying mycobacterium tuberculosis in respiratory infections. Int J Mol Sci. 2025;26(1):18. doi: 10.3390/ijms26010018
- Lin YJ, Komperda I. Biomimetic targeting and autonomous optimization in nanomedicine. ChemRxiv. Preprint posted online 2025. doi: 10.26434/chemrxiv-2025-fxnmb
- Lancaster MA, Renner M, Martin CA, et al. Cerebral organoids model human brain development and microcephaly. Nature. 2013;501(7467):373-379. doi: 10.1038/nature12517
139. Huch M, Gehart H, van Boxtel R, et al. Long-term culture of genome-stable bipotent stem cells from adult human liver. Cell. 2015;160(1):299-312. doi: 10.1016/j.cell.2014.11.050

