Virgin coconut oil attenuates cognitive dysfunction and oxidative stress in streptozotocin-induced diabetic rats

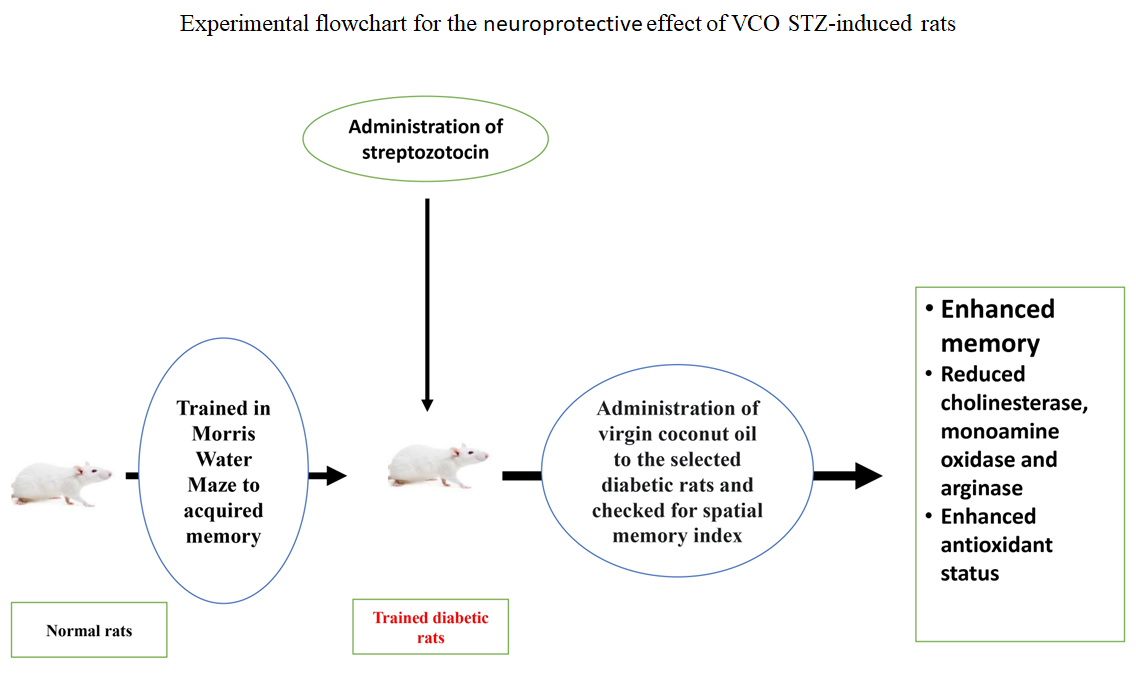
Hyperglycemia contributes to neuronal damage and cognitive impairment. Although virgin coconut oil (COL) is traditionally used to enhance memory, its neuroprotective mechanisms remain inadequately understood. This study investigates the fasting blood glucose (FBG)-lowering and neuroprotective effects of COL in streptozotocin (STZ)-induced diabetic rats. Adult Wistar rats were trained for memory tasks and rendered diabetic with STZ (45 mg/kg, intraperitoneal injection). Seventy-two hours post-induction, rats with FBG levels exceeding 250 mg/dL were assigned to five groups: control, untreated diabetic, metformin-treated diabetic, and diabetic rats treated with COL (0.5 or 1.0 mL/kg body weight). FBG was monitored on days 1, 7, and 14, and spontaneous escape latency (SEL) was assessed. Brain and other tissues were homogenized and analyzed for biochemical and antioxidant markers. Untreated diabetic rats exhibited significantly (p < 0.05) elevated FBG and prolonged SEL, indicating impaired glycemic control and memory performance. Treatment with metformin or COL markedly reduced FBG and improved SEL. Untreated diabetic rats showed increased α-amylase, α-glucosidase, cholinesterase, monoamine oxidase, and arginase activities with reduced antioxidant enzyme levels. Meanwhile, COL treatment reversed these alterations, enhancing antioxidant defense and modulating key metabolic enzymes. COL demonstrated significant antihyperglycemic and neuroprotective effects, suggesting its therapeutic potential in mitigating diabetes-induced oxidative stress and memory dysfunction.
- Ab-Hamid N, Omar N, Ismail CAN, Long I. Diabetes and cognitive decline: challenges and future direction. World J Diabetes. 2023;14(6):795-807. doi: 10.4239/wjd.v14.i6.795
- Kodl CT, Seaquist ER. Cognitive dysfunction and diabetes mellitus. Endocr Rev. 2008;29(4):494-511. doi: 10.1210/er.2007-0034
- Agunloye OM, Oboh G. Effect of diet supplemented with P. ostreatus and L. subnudus on memory index and key enzymes linked with Alzheimer’s disease in streptozotocin-induced diabetes rats. J Food Biochem. 2020;44(8):e13355. doi: 10.1111/jfbc.13355
- Markesbery WR. Oxidative stress hypothesis in Alzheimer’s disease. Free Radic Biol Med. 1997;23(1):134-147. doi: 10.1016/s0891-5849(96)00629-6
- Simó R, Ciudin A, Simó-Servat O, Hernández C. Cognitive impairment and dementia: a new emerging complication of type 2 diabetes—the diabetologist’s perspective. Acta Diabetol. 2017;54:417-424. doi: 10.1007/s00592-017-0970-5
- Niedzielska E, Smaga I, Gawlik M, et al. Oxidative stress in neurodegenerative diseases. Mol Neurobiol. 2016;53:4094- 4125. doi: 10.1007/s12035-015-9337-5
- Franzoni F, Scarfò G, Guidotti S, Fusi J, Asomov M, Pruneti C. Oxidative stress and cognitive decline: the neuroprotective role of natural antioxidants. Front Neurosci. 2021;13:729757. doi: 10.3389/fnins.2021.729757
- DebMandal M, Mandal S. Coconut (Cocos nucifera L.: Arecaceae): in health promotion and disease prevention. Asian Pac J Trop Med. 2011;4(3):241-247. doi: 10.1016/S1995-7645(11)60078-3
- Krishna AG, Gaurav R, Singh BA, Kumar PP, Preeti C. Coconut oil: chemistry, production and its applications—a review. Indian Coconut J. 2010;53(3):15. Available from: https://www.researchgate.net/publication/235780162_ Coconut_Oil_Chemist ry_Production_and_Its_ Applications_-_A_Review.
- Conrado SD. Coconut oil: atherogenic or not? (What therefore causes atherosclerosis?). Philipp J Cardiol. 2003;31(3):97-104.
- Ghani NAA, Channip AA, Chok HHP, Ja’afar F, Yasin FHM, Usman A. Physicochemical properties, antioxidant capacities, and metal contents of virgin coconut oil produced by wet and dry processes. Food Sci Nutr. 2018;6(5):1298- 1306. doi: 10.1002/fsn3.671
- Nevin KG, Rajamohan T. Beneficial effects of virgin coconut oil on lipid parameters and in vitro LDL oxidation. Clin Biochem. 2004;37(9):830-835. doi: 10.1016/j.clinbiochem.2004.04.010
- Offor SJ, Tologbonse AA, Iseh PU, Okoh DE, Akpan S. Hypoglycaemic Activity of Extra Virgin Avocado (Persea americana) Oil and Extra Virgin Coconut (Cocos nucifera) Oil on Alloxan-induced Diabetic Rats. JOCAMR. 2021;16(4):241-250. doi: 10.9734/jocamr/2021/v16i430313
- Alatawi KA, Alshubaily FA. Coconut products alleviate hyperglycaemic, hyperlipidaemic and nephropathy indices in streptozotocin-induced diabetic Wistar rats. Saudi J Biol Sci. 2021;28(8):4224-4231. doi: 10.1016/j.sjbs.2021.06.060
- Agarwal RK, Bosco SJD. Extraction processes of virgin coconut oil. MOJ Food Process Technol. 2017;4(2):54-56. doi: 10.15406/mojfpt.2017.04.00087
- Abioye AIR, Sulyman OR. Histoarchitecture and biochemical effects of coconut oil extract on the ovary of adult Wistar rats. Acta Sci Anat. 2022;1(8):3-8.
- Tuzcu M, Baydas G. Effect of melatonin and vitamin E on diabetes-induced learning and memory impairment in rats. Eur J Pharmacol. 2006;537(1-3):106-110. doi: 10.1016/j.ejphar.2006.03.024
- Worthington V. Amylase, Alpha. In: Worthington Enzyme Manual. Worthington Biochemical Corp; 1993:63-71.
- Apostolidis E, Kwon YI, Shetty K. Inhibitory potential of herb, fruit, and fungal-enriched cheese against key enzymes linked to type 2 diabetes and hypertension. Innov Food Sci Emerg Technol. 2007;8:46-54. doi: 10.1016/j.ifset.2006.06.001
- Ellman GL, Courtney KD, Andres V Jr, Featherstone RM. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol. 1961;7(2):88-95. doi: 10.1016/0006-2952(61)90145-9
- Green AL, Haughton TM. A colorimetric method for the estimation of monoamine oxidase. Biochem J. 1961;78(1):172-176. doi: 10.1042/bj0780172
- Kaysen GA, Strecker HJ. Purification and properties of arginase of rat kidney. Biochem J. 1973;133(4):779-788. doi: 10.1042/bj1330779
- Miranda KM, Espey MG, Wink DA. A rapid, simple spectrophotometric method for simultaneous detection of nitrate and nitrite. Nitric Oxide. 2001;5(1):62-71. doi: 10.1006/niox.2000.0319
- Misra HP, Fridovich I. The role of superoxide anion in the autoxidation of epinephrine and a simple assay for superoxide dismutase. J Biol Chem. 1972;247:3170-3175. doi: 10.1016/S0021-9258(19)45228-9
- Aebi H. Catalase in vitro. Methods Enzymol. 1984;105:121- 126. doi: 10.1016/s0076-6879(84)05016-3
- Rotruck JT, Pope AL, Ganther HE, Swanson AB, Hafeman DG, Hoekstra WG. Selenium: biochemical role as a component of glutathione peroxidase. Science. 1973;179(4073):588-590. doi: 10.1126/science.179.4073.588
- Ellman GL. Tissue sulfhydryl groups. Arch Biochem Biophys. 1959;82(1):70-77. doi: 10.1016/0003-9861(59)90090-6
- Ohkawa H, Ohishi N, Yagi K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem. 1979;95:351-358. doi: 10.1016/0003-2697(79)90738-3
- Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976;72:248-254. doi: 10.1016/0003-2697(76)90527-3
- American Diabetes Association. Diagnosis and classification of diabetes mellitus. Diabetes Care. 2006;29(Suppl 1):S43- S48. doi: 10.2337/diacare.29.s1.06.s43
- AlGhamdi S. Potential antidiabetic and antioxidant effects of coconut oil on streptozotocin-induced diabetes in male Sprague-Dawley rats. Int J Pharm Phytopharmacol Res. 2019;9(5):68-76.
- Iranloye B, Oludare G, Olubiyi M. Antidiabetic and antioxidant effects of virgin coconut oil in alloxan-induced diabetic male Sprague Dawley rats. J Diabetes Mellitus. 2013;3(4):221-228. doi: 10.4236/jdm.2013.34034
- Wickimayr M, Dietze G, Gunther B, Bottger I, Mayer L, Janetschek P. Improvement of glucose assimilation and protein degradation by bradykinin in maturity onset diabetics and in surgical patients. In: Fujii S, Moriya H, Suzuki T, eds. Kinins—II. Adv Exp Med Biol. 1979;120:637- 645. doi: 10.1007/978-1-4757-0926-1_52
- Alqahtani AS, Hidayathulla S, Rehman MT, et al. Alpha-amylase and alpha-glucosidase enzyme inhibition and antioxidant potential of 3-oxolupenal and katononic acid isolated from Nuxia oppositifolia. Biomolecules. 2019;10(1):61. doi: 10.3390/biom10010061
- Siddalingaswamy M, Rayaorth A, Khanum F. Antidiabetic effects of cold and hot extracted virgin coconut oil. J Diabetes Mellitus. 2011;1(4):118-123. doi: 10.4236/jdm.2011.14016
- Zhao W, Varghese M, Vempati P, et al. Caprylic triglyceride as a novel therapeutic approach to effectively improve performance and attenuate symptoms due to motor neuron loss in ALS disease. PLoS One. 2012;7(11):e49191. doi: 10.1371/journal.pone.0049191
- Grzelczyk J, Budryn G, Peña-García J, et al. Evaluation of the inhibition of monoamine oxidase A by bioactive coffee compounds protecting serotonin degradation. Food Chem. 2021;348:129108. doi: 10.1016/j.foodchem.2021.129108
- Durante W, Johnson FK, Johnson RA. Arginase: a critical regulator of nitric oxide synthesis and vascular function. Clin Exp Pharmacol Physiol. 2007;34(9):906-911. doi: 10.1111/j.1440-1681.2007.04638.x
- Steinert JR, Chernova T, Forsythe ID. Nitric oxide signaling in brain function, dysfunction, and dementia. Neuroscientist. 2010;16:435-452. doi: 10.1177/1073858410366481
- Xu J, Begley P, Church SJ, et al. Graded perturbations of metabolism in multiple regions of human brain in Alzheimer’s disease: Snapshot of a pervasive metabolic disorder. Biochim Biophys Acta Mol Basis Dis. 2016;1862:1084-1092. doi: 10.1016/j.bbadis.2016.03.001
- Narayanankutty A, Illam SP, Raghavamenon AC. Health impacts of different edible oils prepared from coconut (Cocos nucifera): a comprehensive review. Trends Food Sci Technol. 2018;80:1-7. doi: 10.1016/j.tifs.2018.07.025
- Illam SP, Narayanankutty A, Raghavamenon AC. Polyphenols of virgin coconut oil prevent pro-oxidant mediated cell death. Toxicol Mech Methods. 2017;27(6):442- 450. doi: 10.1080/15376516.2017.1320458

