Removal of antimonite by low-temperature Fe-modified biochar

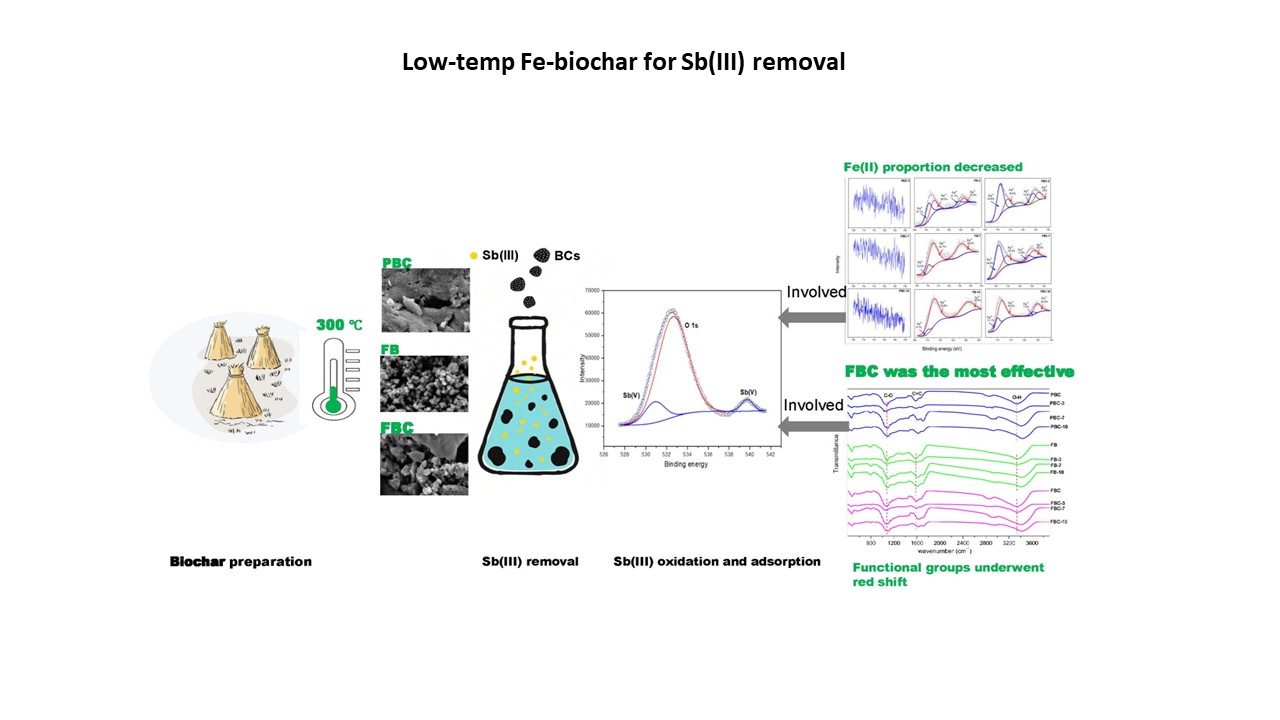
High-temperature biochar (BC) has been extensively studied for treating wastewater contaminated with antimonite (Sb(III)). In contrast, low-temperature BCs are more energy-efficient and eco-friendly, making them more promising for remediating contaminated environmental media; however, their performance and mechanisms for Sb(III) removal remain largely unexplored. To provide a theoretical basis for remediating antimony–contaminated natural environments, the present study investigated the Sb(III) removal performance and underlying mechanisms of two low-temperature (300 °C) BCs, namely ferric chloride (FeCl3) co-pyrolyzed BC (FB) and FeCl3-impregnated pristine BC (FBC), with pristine BC (PBC) as the control. The Sb(III) removal performance was evaluated using adsorption kinetic and isotherm models, and the mechanisms were elucidated via adsorption experiments across a pH range of 3–10. Results showed that the maximum adsorption capacities of PBC, FB, and FBC were 12.93, 9.01, and 14.57 mg·g−1, with FBC exhibiting superior Sb(III) removal performance. The kinetic model indicated that Sb(III) removal by FB and FBC was dominated by chemisorption. Characterization of the solid phases in the adsorption systems showed that functional groups were mainly responsible for Sb(III) adsorption/oxidation at pH 3, whereas co-oxidation of ferrous iron (Fe(II)) and Sb(III) was dominant at pH 10. Moreover, at an initial Sb(III) concentration of 5 mg·L−1, the residual aqueous Sb(III) in FBC-treated adsorption systems ranged from 0.015 to 0.036 mg·L−1 over the pH range of 3–10, indicating FBC’s high versatility in Sb(III) removal owing to its abundant functional groups and high Fe(II) content. Thus, FBC exhibits substantial potential to remediate Sb(III) contamination in environmental media across a typical environmental pH range of 3.0–10.0.
- Zhang Y, O’Loughlin EJ, Kwon MJ. Antimony redox processes in the environment: A critical review of associated oxidants and reductants. J Hazard Mater. 2022;431:128607. doi: 10.1016/j.jhazmat.2022.128607
- Li C, Ran Y, Wu P, et al. Antimony and arsenic migration in a heterogeneous subsurface at an abandoned antimony smelter under rainfall. J Hazard Mater. 2024;470:134156. doi: 10.1016/j.jhazmat.2024.134156
- Herath I, Vithanage M, Bundschuh J. Antimony as a global dilemma: Geochemistry, mobility, fate and transport. Environ Pollut. 2017;223:545-559. doi: 10.1016/j.envpol.2017.01.057
- Liu L, Supe Tulcan RX, He M, Ouyang W, Zhang Q, Huarez Yarleque, CM, et al. Antimony pollution threatens soils and riverine habitats across China: An analysis of antimony concentration changes and risks. Crit Rev Environ Sci. Technol. 2023;54(10):797-816. doi: 10.1080/10643389.2023.2279882
- He M, Wang N, Long X, et al. Antimony speciation in the envFement: Recent advances in understanding the biogeochemical processes and ecological effects. J Environ Sci. 2019;75:14-39. doi: 10.1016/j.jes.2018.05.023
- Bolan N, Kumar M, Singh E, et al. Antimony contamination and its risk management in complex environmental settings: A review. EnvFe Int. 2022;158:106908. doi: 10.1016/j.envint.2021.106908
- Li L, Tu H, Zhang S, et al. Geochemical behaviors of antimony in mining-affected water environment (Southwest China). Environ Geochem Health. 2019;41(6):2397-2411. doi: 10.1007/s10653-019-00285-8
- Filella M, Belzile N, Chen YW. Antimony in the environment: a review focused on natural waters: I. Occurrence. Earth-Sci Rev. 2002;57(1-2):125-176. doi: 10.1016/S0012-8252(01)00070-8
- Long J, Tan D, Deng S, Li B, Ding D, Lei M. Antimony accumulation and Fe plaque formation at different growth stages of rice (Oryza sativa L.). Environ Pollut. 2019;249:414- 422. doi: 10.1016/j.envpol.2019.03.042
- Gao Y, Chen H, Fang Z, et al. Coupled sorptive and oxidative antimony (III) removal by Fe-modified biochar: Mechanisms of electron-donating capacity and reactive Fe species. Environ Pollut. 2023;337:122637. doi: 10.1016/j.envpol.2023.122637
- Yao B, Li Y, Zeng W, et al. Synergistic adsorption and oxidation of trivalent antimony from groundwater using biochar-supported magnesium ferrite: Performances and mechanisms. Environ Pollut. 2023;323:121318. doi: 10.1016/j.envpol.2023.121318
- Zhang L, Dong Y, Liu J, Liu C, Liu W, Lin H. The effect of co-pyrolysis temperature for Fe-biochar composites on their adsorption behavior of antimonite and antimonate in aqueous solution. Bioresour Technol. 2022;347:126362. doi: 10.1016/j.biortech.2021.126362
- Zhang L, Yang D, Tao Y, et al. Efficiently simultaneous sorption and oxidation of antimonite on nitroso grafting-manganese loading binary-modified biochar from aqueous solution. Sep Purif Technol. 2024;337:126398. doi: 10.1016/j.seppur.2024.126398
- Zhang L, Dong Y, Liu J, Liu W, Lu Y, Lin H. Promotion of higher synthesis temperature for higher-efficient removal of antimonite and antimonate in aqueous solution by Fe-loaded porous biochar. Bioresour Technol. 2022;363:127889. doi: 10.1016/j.biortech.2022.127889
- Ji J, Xu S, Ma Z, Mou Y. Optimisation of preparation conditions and removal mechanism for trivalent antimony by biochar-supported nano zero-valent Fe. EnvFe Technol Innov. 2022;26:102240. doi: 10.1016/j.eti.2021.102240
- Dong Z, Zhou J, Huang T, et al. Effects of oxygen on the adsorption/oxidation of aqueous Sb (III) by Fe-loaded biochar: An X-ray absorption spectroscopy study. Sci Total Environ. 2022;846:157414. doi: 10.1016/j.scitotenv.2022.157414
- Wei D, Li B, Luo L, et al. Simultaneous adsorption and oxidation of antimonite onto nano zero-valent Fe sludge-based biochar: Indispensable role of reactive oxygen species and redox-active moieties. J Hazard Mater. 2020;391:122057. doi: 10.1016/j.jhazmat.2020.122057
- Chen H, Gao Y, Li J, Sun C, Sarkar B, Bhatnagar A. Insights into simultaneous adsorption and oxidation of antimonite [Sb (III)] by crawfish shell-derived biochar: spectroscopic investigation and theoretical calculations. Biochar. 2022;4(1):37. doi: 10.1007/s42773-022-00161-2
- Lai L, Liu X, Ren W, et al. Efficient removal of Sb (III) from water using β-FeOOH-modified biochar: synthesis, performance and mechanism. Chemosphere. 2023;311:137057. doi: 10.1016/j.chemosphere.2022.137057
- Chen H, Gao Y, El-Naggar A, et al. Enhanced sorption of trivalent antimony by chitosan-loaded biochar in aqueous solutions: characterization, performance and mechanisms. J Hazard Mater. 2022;425:127971. doi: 10.1016/j.jhazmat.2021.127971
- Chen Z, Zhang J, Huang, L Yuan Z, Li Z, Liu M. Removal of Cd and Pb with biochar made from dairy manure at low temperature. J Integr Agric. 2019;18(1):201-210. doi: 10.1016/S2095-3119(18)61987-2
- Buss W, Graham MC, MacKinnon G, Mašek O. Strategies for producing biochars with minimum PAH contamination. J Anal Appl Pyrol. 2016;119:24-30. doi: 10.1016/j.jaap.2016.04.001
- De la Rosa JM, Sánchez-Martín AM, Campos P, Miller AZ. Effect of pyrolysis conditions on the total contents of polycyclic aromatic hydrocarbons in biochars produced from organic residues: Assessment of their hazard potential. Sci Total Environ. 2019;667:578-585. doi: 10.1016/j.scitotenv.2019.02.421
- Ahmad M, Lee, SS, Lim JE, Lee SE, Cho JS, Moon DH. Speciation and phytoavailability of lead and antimony in a small arms range soil amended with mussel shell, cow bone and biochar: EXAFS spectroscopy and chemical extractions. Chemosphere. 2014;95:433-441. doi: 10.1016/j.chemosphere.2013.09.077
- Tomczyk A, Sokołowska Z, Boguta P. Biochar physicochemical properties: pyrolysis temperature and feedstock kind effects. Rev Environ Sci Bio/Technol. 2020;19(1):191-215. doi: 10.1007/s11157-020-09523-3
- Lin SL, Zhang H, Chen WH, Song M, Kwon EE. Low-temperature biochar production from torrefaction for wastewater treatment: A review. Bioresour Technol. 2023;387:129588. doi: 10.1016/j.biortech.2023.129588
- Gámiz B, Hall K, Spokas KA, Cox L. Understanding activation effects on low-temperature biochar for optimization of herbicide sorption. Agronomy. 2019;9(10):588. doi: 10.3390/agronomy9100588
- Gruss I, Twardowski JP, Latawiec A, Medyńska-Juraszek A, Królczyk J. Risk assessment of low-temperature biochar used as soil amendment on soil mesofauna. Environ Sci Pollut Res. 2019;26(18):18230-18239. doi: 10.1007/s11356-019-05153-7
- Guo F, Xu F, Cai R, et al. Enhancement of denitrification in biofilters by immobilized biochar under low-temperature stress. Bioresour Technol. 2022;347:126664. doi: 10.1016/j.biortech.2021.126664
- He R, Peng Z, Lyu H, Huang H, Nan Q, Tang J. Synthesis and characterization of an Fe-impregnated biochar for aqueous arsenic removal. Sci Total Environ. 2018;612:1177-1186. doi: 10.1016/j.scitotenv.2017.09.016
- Yang X, Shaheen SM, Wang J, et al. Elucidating the redox-driven dynamic interactions between arsenic and Fe-impregnated biochar in a paddy soil using geochemical and spectroscopic techniques. J Hazard Mater. 2022;422:126808. doi: 10.1016/j.jhazmat.2021.126808
- Yao W, Weng Y, Catchmark JM. Improved cellulose X-ray diffraction analysis using Fourier series modeling. Cellulose. 2020;27(10):5563-5579. doi: 10.1007/s10570-020-03177-8
- Zhang RY, Zheng XX, Chen BH, et al. Enhanced adsorption of sulfamethoxazole from aqueous solution by Fe-impregnated graphited biochar. J Clean Prod. 2020;256:120662. doi: 10.1016/j.jclepro.2020.120662
- Yanniotis, S, Blahovec, J. Model analysis of sorption isotherms. LWT-Food Sci Technol. 2009;42(10):1688-1695. doi: 10.1016/j.lwt.2009.05.010
- Tang J, Zhao B, Lyu H, Li D. Development of a novel pyrite/ biochar composite (BM-FeS₂@BC) by ball milling for aqueous Cr(VI) removal and its mechanisms. J Hazard Mater. 2021;413:125415. doi: 10.1016/j.jhazmat.2021.125415
- Yan B, Niu C, Wang J. Analyses of levofloxacin adsorption on pretreated barley straw with respect to temperature: kinetics, π–π electron-donor–acceptor interaction and site energy distribution. Environ Sci Technol. 2017;51:8048- 8056. doi: 10.1021/acs.est.7b00327
- Yang D, Li J, Luo L, Deng R, He Q, Chen Y. Exceptional levofloxacin removal using biochar-derived porous carbon sheets: Mechanisms and density-functional-theory calculation. Chem Eng J. 2020;387:124103. doi: 10.1016/j.cej.2020.124103
- Berryman OB, Johnson DW. Experimental evidence for interactions between anions and electron-deficient aromatic rings. Chem Commun. 2009;22:3143-3153. doi: 10.1039/B823236A
- Fan P, Sun Y, Qiao J, Imc L, Guan X. Influence of weak magnetic field and tartrate on the oxidation and sequestration of Sb(III) by zerovalent Fe: batch and semi-continuous flow study. J Hazard Mater. 2017;343:266-275. doi: 10.1016/j.jhazmat.2017.09.041
- Wang H, Guo W, Yin R, et al. Biochar-induced Fe (III) reduction for persulfate activation in sulfamethoxazole degradation: Insight into the electron transfer, radical oxidation and degradation pathways. Chem Eng J. 2019;362:561-569. doi: 10.1016/j.cej.2019.01.053
- Huang JZ, Jones A, Waite TD, et al. Fe(II) redox chemistry in the environment. Chem. Rev. 2021;121(13):8161-8233. doi: 10.1021/acs.chemrev.0c01286
- Zhang X, Fu Q, Hu H, Zhu J, Liu Y. Effects of Fe (II) on As (III) oxidation in Fe (II)-As (III) co-oxidation: Limiting and driving roles. J Hazard Mater. 2023;447:130790. doi: 10.1016/j.jhazmat.2023.130790
- Meng F, Tong H, Feng C, et al. Structural Fe (II)-induced generation of reactive oxygen species on magnetite surface for aqueous As (III) oxidation during oxygen activation. Water Res. 2024;252:121232. doi: 10.1016/j.watres.2024.121232
- Wang Y, Kong L, He M, et al. Mechanistic insights into Sb (III) and Fe (II) co-oxidation by oxygen and hydrogen peroxide: Dominant reactive oxygen species and roles of organic ligands. Water Res. 2023;242:120296. doi: 10.1016/j.watres.2023.120296
- Cheng Z, Lyu HH, Shen BX, Tian JY, Sun YF, Wu CF. Removal of antimonite (Sb(III)) from aqueous solution using a magnetic Fe-modified carbon nanotubes (CNTs) composite: experimental observations and governing mechanisms. Chemosphere. 2022;288:132581. doi: 10.1016/j.chemosphere.2021.132581
- Xu ZB, Wan ZH, Sun YQ, et al. Electroactive Fe-biochar for redox-related remediation of arsenic and chromium: distinct redox nature with varying Fe/carbon speciation. J Hazard Mater. 2022;430:128479. doi: 10.1016/j.jhazmat.2022.128479
- Morgan, B, Lahav, O. The effect of pH on the kinetics of spontaneous Fe (II) oxidation by O₂ in aqueous solution-basic principles and a simple heuristic description. Chemosphere. 2007;68(11):2080-2084. doi: 10.1016/j.chemosphere.2007.02.015
- Abhishek K, Parashar N, Patel M, et al. Recent advancements in antimony (Sb) removal from water and wastewater by carbon-based materials: a systematic review. Environ Monit Assess. 2023;195(6):758. doi: 10.1007/s10661-023-11322-6
- Xu S, Zhong Z, Liu W, Deng H, Lin Z. Removal of Sb(III) from wastewater by magnesium oxide and the related mechanisms. Environ Res. 2020;186:109489. doi: 10.1016/j.envres.2020.109489
- Chen W, Lin Z, Chen Z, Weng X, Owens G, Chen Z. Simultaneous removal of Sb(III) and Sb(V) from mining wastewater by reduced graphene oxide/bimetallic nanoparticles. Sci Total Environ. 2022;836:155704. doi: 10.1016/j.scitotenv.2022.155704
- Long J, Zhou D, Wang J, et al. Repeated inoculation of antimony resistant bacterium reduces antimony accumulation in rice plants. Chemosphere. 2023;327:138335. doi: 10.1016/j.chemosphere.2023.138335
- Ceriotti G, Amarasiriwardena D. A study of antimony complexed to soil-derived humic acids and inorganic antimony species along a Massachusetts highway. Microchem J. 2009;91(1):85-93. doi: 10.1016/j.microc.2008.08.010

