Inner ear organoids: From three-dimensional bioprinting to vascularization

The inner ear is essential for auditory perception and balance, yet damage to cochlear hair cells remains a major cause of irreversible hearing loss worldwide. As the number of individuals affected by sensorineural deafness continues to rise, there is a growing need for deeper pathophysiological insight and the development of effective therapies. Inner ear organoids, which recapitulate key aspects of the in vivo inner ear microenvironment, have emerged as powerful platforms for disease modeling, high-throughput drug screening, and regenerative medicine. This review summarizes recent progress in the development of inner ear organoids, with a focus on strategies to improve their structural complexity and functional maturity. In particular, we highlight innovations in vascularization and three-dimensional bioprinting that are advancing organoid scalability and integration potential. Meanwhile, commonly used biomaterials in three-dimensional bioprinting for inner ear organoids are systematically compared. Finally, we discuss current challenges and future directions for translating these technologies into clinical applications.
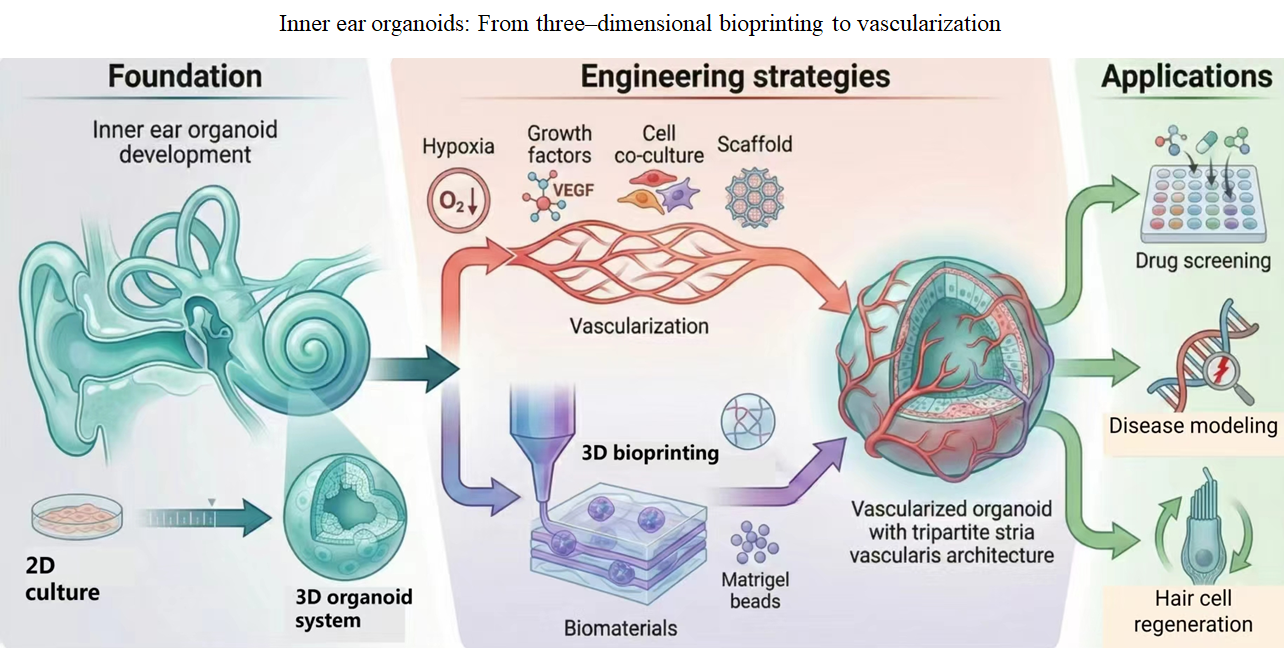
- Qi J, Zhang L, Wang X, et al. Modeling, applications and challenges of inner ear organoid. Smart Med. 2024;3(1):e20230028. doi: 10.1002/SMMD.20230028
- Wagner EL, Shin J-B. Mechanisms of hair cell damage and repair. Trends Neurosci. 2019;42(6):414-424. doi: 10.1016/j.tins.2019.03.006
- World Health Organization. Deafness and hearing loss. Available from: https://www.who.int/news-room/fact- sheets/detail/deafness-and-hearing-loss [Last accessed on August 6, 2025].
- Lu Y-C, Wu C-C, Yang T-H, et al. Differences in the pathogenicity of the p. H723R mutation of the common deafness-associated SLC26A4 gene in humans and mice. PLoS ONE. 2013;8(6):e64906. doi: 10.1371/journal.pone.0064906
- Yi Z, Wang X, Yin G, Sun Y. The Blood-Labyrinth Barrier: Non-Invasive Delivery Strategies for Inner Ear Drug Delivery. Pharmaceutics. 2025;17(4):482. doi: 10.3390/pharmaceutics17040482
- Koehler KR, Nie J, Longworth-Mills E, et al. Generation of inner ear organoids containing functional hair cells from human pluripotent stem cells. Nat Biotechnol. 2017;35(6):583- 589. doi: 10.1038/nbt.3840
- Castaño-González K, Köppl C, Pyott SJ. The crucial role of diverse animal models to investigate cochlear aging and hearing loss. Hear Res. 2024;445:108989. doi: 10.1016/j.heares.2024.108989
- Qin X, Fu L, Li C, Tan X, Yin X. Optimized inner ear organoids for efficient hair cell generation and ototoxicity response modeling. Sci China Life Sci. 2025;68(5):1369-1383. doi: 10.1007/s11427-024-2803-1
- van der Valk WH, Nist-Lund C, Zhang J, et al. Generation and characterization of vestibular inner ear organoids from human pluripotent stem cells. Nat Protoc. 2025;21(2):391- 428. doi: 10.1038/s41596-025-01191-3
- Lou Y, Liu Y, Wu M, Jia G, Xia M, Li W. Inner ear organoids: Recent progress and potential applications. Fundam Res. 2025;5(6):2926-2936. doi: 10.1016/j.fmre.2023.07.013
- Wang Y, Chen M, Pan Y, Li X, Lou X. Inner Ear Organoid as a Preclinical Model of Hearing Regeneration: Progress and Applications. Stem Cell Rev Rep. 2025;21:2031-2042. doi: 10.1007/s12015-025-10941-5
- Moeinvaziri F, Zarkesh I, Pooyan P, Nunez DA, Baharvand H. Inner ear organoids: Progress and outlook, with a focus on the vascularization. FEBS J. 2022;289(23):7368-7384. doi: 10.1111/febs.16146
- Pianigiani G, Roccio M. Inner Ear Organoids: Strengths and Limitations. J Assoc Res Otolaryngol. 2024;25(1):5-11. doi: 10.1007/s10162-024-00929-2
- Streit A. Origin of the vertebrate inner ear: Evolution and induction of the otic placode. J Anat. 2001;199(Pt 1-2):99- 103. doi: 10.1046/j.1469-7580.2001.19910099.x
- Choo D. The role of the hindbrain in patterning of the otocyst. Dev Biol. 2007;308(2):257-265. doi: 10.1016/j.ydbio.2007.05.035
- Ohyama T, Groves AK, Martin K. The first steps towards hearing: Mechanisms of otic placode induction. Int J Dev Biol. 2007;51(6-7):463-472. doi: 10.1387/ijdb.072320to
- Kelly MC, Chen P. Development of form and function in the mammalian cochlea. Curr Opin Neurobiol. 2009;19(4):395- 401. doi: 10.1016/j.conb.2009.07.010
- Whitfield TT. Development of the inner ear. Curr Opin Genet Dev. 2015;32:112-118. doi: 10.1016/j.gde.2015.02.006
- Lim R, Brichta AM. Anatomical and physiological development of the human inner ear. Hear Res. 2016;338:9- 21. doi: 10.1016/j.heares.2016.02.004
- Köppl C, Wilms V, Russell IJ, Nothwang HG. Evolution of Endolymph Secretion and Endolymphatic Potential Generation in the Vertebrate Inner Ear. Brain Behav Evol. 2018;92(1-2):1-31. doi: 10.1159/000494050
- Driver EC, Kelley MW. Development of the cochlea. Development. 2020;147(12):dev162263. doi: 10.1242/dev.162263
- Li H, Liu H, Heller S. Pluripotent stem cells from the adult mouse inner ear. Nat Med. 2003;9(10):1293-1299. doi: 10.1038/nm925
- Oshima K, Grimm CM, Corrales CE, et al. Differential distribution of stem cells in the auditory and vestibular organs of the inner ear. J Assoc Res Otolaryngol. 2007;8(1):18- 31. doi: 10.1007/s10162-006-0058-3
- Oshima K, Shin K, Diensthuber M, Peng AW, Ricci AJ, Heller S. Mechanosensitive hair cell-like cells from embryonic and induced pluripotent stem cells. Cell. 2010;141(4):704-716. doi: 10.1016/j.cell.2010.03.035
- Barker N, Van Es JH, Kuipers J, et al. Identification of stem cells in small intestine and colon by marker gene Lgr5. Nature. 2007;449(7165):1003-1007. doi: 10.1038/nature06196
- Koo B-K, Clevers H. Stem cells marked by the R-spondin receptor LGR5. Gastroenterology. 2014;147(2):289-302. doi: 10.1053/j.gastro.2014.05.007
- Chai R, Kuo B, Wang T, et al. Wnt signaling induces proliferation of sensory precursors in the postnatal mouse cochlea. Proc Natl Acad Sci USA. 2012;109(21):8167-8172. doi: 10.1073/pnas.1202774109
- Shi F, Kempfle JS, Edge AS. Wnt-responsive Lgr5-expressing stem cells are hair cell progenitors in the cochlea. J Neurosci. 2012;32(28):9639-9648. doi: 10.1523/JNEUROSCI.1064-12.2012
- Koehler KR, Mikosz AM, Molosh AI, Patel D, Hashino E. Generation of inner ear sensory epithelia from pluripotent stem cells in 3D culture. Nature. 2013;500(7461):217-221. doi: 10.1038/nature12298
- McLean WJ, Yin X, Lu L, et al. Clonal expansion of Lgr5- positive cells from mammalian cochlea and high-purity generation of sensory hair cells. Cell Rep. 2017;18(8):1917- 1929. doi: 10.1016/j.celrep.2017.01.066
- Roccio M, Perny M, Ealy M, Widmer HR, Heller S, Senn P. Molecular characterization and prospective isolation of human fetal cochlear hair cell progenitors. Nat Commun. 2018;9(1):4027. doi: 10.1038/s41467-018-06334-7
- Xia M, Ma J, Wu M, et al. Generation of innervated cochlear organoid recapitulates early development of auditory unit. Stem Cell Rep. 2023;18(1):319-336. doi: 10.1016/j.stemcr.2022.11.024
- Moore ST, Nakamura T, Nie J, et al. Generating high-fidelity cochlear organoids from human pluripotent stem cells. Cell Stem Cell. 2023;30(7):950-961.e7. doi: 10.1016/j.stem.2023.06.006
- Xia M, Wu M, Li Y, et al. Varying mechanical forces drive sensory epithelium formation. Sci Adv. 2023;9(44):eadf2664. doi: 10.1126/sciadv.adf2664
- Matsui TK, Tsuru Y, Hasegawa K, Kuwako K-i. Vascularization of human brain organoids. Stem Cells. 2021;39(8):1017-1024. doi: 10.1002/stem.3368
- Novosel EC, Kleinhans C, Kluger PJ. Vascularization is the key challenge in tissue engineering. Adv Drug Deliv Rev. 2011;63(4-5):300-311.doi: 10.1016/j.addr.2011.03.004
- Nyberg S, Abbott NJ, Shi X, Steyger PS, Dabdoub A. Delivery of therapeutics to the inner ear: The challenge of the blood- labyrinth barrier. Sci Transl Med. 2019;11(482):eaao0935. doi: 10.1126/scitranslmed.aao0935
- Wangemann P. Supporting sensory transduction: Cochlear fluid homeostasis and the endocochlear potential. J Physiol. 2006;576(Pt 1):11-21. doi: 10.1113/jphysiol.2006.112888
- Hansen CE, Hollaus D, Kamermans A, de Vries HE. Tension at the gate: Sensing mechanical forces at the blood– brain barrier in health and disease. J Neuroinflammation. 2024;21(1):325. doi: 10.1186/s12974-024-03321-2
- Poorna Pillutla SV, Kaur C, Roy TS, Jacob TG. Estimation of volume of stria vascularis and the length of its capillaries in the human cochlea. J Microsc Ultrastruct. 2019;7(3):117-123. doi: 10.4103/JMAU.JMAU_12_19
- Li Y, Sun X-Y, Zeng P-M, Luo Z-G. Neural Responses to Hypoxic Injury in a Vascularized Cerebral Organoid Model. Neurosci Bull. 2025:1-13. doi: 10.1007/s12264-024-01346-2
- Shaji M, Tamada A, Fujimoto K, Muguruma K, Karsten SL, Yokokawa R. Deciphering potential vascularization factors of on-chip co-cultured hiPSC-derived cerebral organoids. Lab Chip. 2024;24(4):680-696. doi: 10.1039/d3lc00930k
- Shi Y, Sun L, Wang M, et al. Vascularized human cortical organoids (vOrganoids) model cortical development in vivo. PLoS Biol. 2020;18(5):e3000705. doi: 10.1371/journal.pbio.3000705
- Salehi A, Rutz L, Ulbrich K, et al. Surface-modified gelatin hydrogel scaffolds with imprinted microgrooves: Physical characterization and study on endothelial cell interaction. J Biomater Sci Polym Ed. 2025;36(5):1-36. doi: 10.1080/09205063.2025.2527912
- Mastrullo V, Cathery W, Velliou E, Madeddu P, Campagnolo P. Angiogenesis in tissue engineering: As nature intended? Front Bioeng Biotechnol. 2020;8:188. doi: 10.3389/fbioe.2020.00188
- Walaas GA, Gopalakrishnan S, Bakke I, et al. Physiological hypoxia improves growth and functional differentiation of human intestinal epithelial organoids. Front Immunol. 2023;14:1095812. doi: 10.3389/fimmu.2023.1095812
- Kumano K, Nakahashi H, Louphrasitthiphol P, et al. Hypoxia at 3D organoid establishment selects essential subclones within heterogenous pancreatic cancer. Front Cell Dev Biol. 2024;12:1327772. doi: 10.3389/fcell.2024.1327772
- Min S, Ko IK, Yoo JJ. State-of-the-art strategies for the vascularization of three-dimensional engineered organs. Vasc Specialist Int. 2019;35(2):77-89. doi: 10.5758/vsi.2019.35.2.77
- Almubarak S, Nethercott H, Freeberg M, et al. Tissue engineering strategies for promoting vascularized bone regeneration. Bone. 2016;83:197-209. doi: 10.1016/j.bone.2015.11.011
- Martino MM, Hubbell JA. The 12th–14th type III repeats of fibronectin function as a highly promiscuous growth factor‐binding domain. FASEB J. 2010;24(12):4711-4721. doi: 10.1096/fj.09-151282
- Ebeid M, Huh S-H. FGF signaling: Diverse roles during cochlear development. BMB Rep. 2017;50(10):487-495. doi: 10.5483/BMBRep.2017.50.10.164
- Huh S-H, Warchol ME, Ornitz DM. Cochlear progenitor number is controlled through mesenchymal FGF receptor signaling. eLife. 2015;4:e05921. doi: 10.7554/eLife.05921
- Yang LM, Cheah KSE, Huh S-H, Ornitz DM. Sox2 and FGF20 interact to regulate organ of Corti hair cell and supporting cell development in a spatially-graded manner. PLoS Genet. 2019;15(7):e1008254. doi: 10.1371/journal.pgen.1008254
- Jacques BE, Montcouquiol ME, Layman EM, Lewandoski M, Kelley MW. Fgf8 induces pillar cell fate and regulates cellular patterning in the mammalian cochlea. Development. 2007;134(16):3021-3029. doi: 10.1242/dev.02874
- Zhang J, Hou Z, Wang X, et al. VEGFA165 gene therapy ameliorates blood-labyrinth barrier breakdown and hearing loss. JCI Insight. 2021;6(8):e143285. doi: 10.1172/jci.insight.143285
- Wan L, Lovett M, Warchol ME, Stone JS. Vascular endothelial growth factor is required for regeneration of auditory hair cells in the avian inner ear. Hear Res. 2020;385:107839. doi: 10.1016/j.heares.2019.107839
- Wang K, Lin R-Z, Hong X, et al. Robust differentiation of human pluripotent stem cells into endothelial cells via temporal modulation of ETV2 with modified mRNA. Sci Adv. 2020;6(30):eaba7606. doi: 10.1126/sciadv.aba7606
- Skylar-Scott MA, Huang JY, Lu A, et al. Orthogonally induced differentiation of stem cells for the programmatic patterning of vascularized organoids and bioprinted tissues. Nat Biomed Eng. 2022;6(4):449-462. doi: 10.1038/s41551-022-00856-8
- Palikuqi B, Nguyen D-HT, Li G, et al. Adaptable haemodynamic endothelial cells for organogenesis and tumorigenesis. Nature. 2020;585(7825):426-432. doi: 10.1038/s41586-020-2712-z
- Kim TM, Lee RH, Kim MS, Lewis CA, Park C. ETV2/ER71, the key factor leading the paths to vascular regeneration and angiogenic reprogramming. Stem Cell Res Ther. 2023;14(1):41. doi: 10.1186/s13287-023-03267-x
- Cakir B, Xiang Y, Tanaka Y, et al. Engineering of human brain organoids with a functional vascular-like system. Nat Methods. 2019;16(11):1169-1175. doi: 10.1038/s41592-019-0586-5
- Cai H, Tian C, Chen L, et al. Vascular network-inspired diffusible scaffolds for engineering functional midbrain organoids. Cell Stem Cell. 2025;32(5):824-837.e5. doi: 10.1016/j.stem.2025.02.010
- Nwokoye PN, Abilez OJ. Bioengineering methods for vascularizing organoids. Cell Rep Methods. 2024;4(6):100779. doi: 10.1016/j.crmeth.2024.100779
- Han X, Saiding Q, Cai X, et al. Intelligent vascularized 3D/4D/5D/6D-printed tissue scaffolds. Nano Micro Lett. 2023;15(1):239. doi: 10.1007/s40820-023-01187-2
- Arkenberg MR, Jafarkhani M, Lin CC, Hashino E. Chemically defined and dynamic click hydrogels support hair cell differentiation in human inner ear organoids. Stem Cell Rep. 2025;20(2):102386. doi: 10.1016/j.stemcr.2024.12.001
- Park S, Kim YJ, Sharma H, et al. Graphene hybrid inner ear organoid with enhanced maturity. Nano Lett. 2023;23(12):5573-5580. doi: 10.1021/acs.nanolett.3c00988
- Hu Y, Xing J, Zhang H, et al. Electroacoustic Responsive Cochlea‐on‐a‐Chip. Adv Mater. 2024;36(24):2309002. doi: 10.1002/adma.202309002
- Gavara N, Manoussaki D, Chadwick RS. Auditory mechanics of the tectorial membrane and the cochlear spiral. Curr Opin Otolaryngol Head Neck Surg. 2011;19(5):382-387. doi: 10.1097/MOO.0b013e32834a5bc9
- Reichenbach T, Hudspeth AJ. The physics of hearing: Fluid mechanics and the active process of the inner ear. Rep Prog Phys. 2014;77(7):076601. doi: 10.1088/0034-4885/77/7/076601
- Ji S, Almeida E, Guvendiren M. 3D bioprinting of complex channels within cell-laden hydrogels. Acta Biomater. 2019;95:214-224. doi: 10.1016/j.actbio.2019.02.038
- Brassard JA, Nikolaev M, Hübscher T, Hofer M, Lutolf MP. Recapitulating macro-scale tissue self-organization through organoid bioprinting. Nat Mater. 2021;20(1):22-29. doi: 10.1038/s41563-020-00803-5
- Homan KA, Gupta N, Kroll KT, et al. Flow-enhanced vascularization and maturation of kidney organoids in vitro. Nat Methods. 2019;16(3):255-262. doi: 10.1038/s41592-019-0325-y
- Onesto MM, Kim J-i, Pasca SP. Assembloid models of cell- cell interaction to study tissue and disease biology. Cell Stem Cell. 2024;31(11):1563-1573. doi: 10.1016/j.stem.2024.09.017
- Kang H-W, Lee SJ, Ko IK, Kengla C, Yoo JJ, Atala A. A 3D bioprinting system to produce human-scale tissue constructs with structural integrity. Nat Biotechnol. 2016;34(3):312-319. doi: 10.1038/nbt.3413
- Yue K, Trujillo-de Santiago G, Alvarez MM, Tamayol A, Annabi N, Khademhosseini A. Synthesis, properties, and biomedical applications of gelatin methacryloyl (GelMA) hydrogels. Biomaterials. 2015;73:254-271. doi: 10.1016/j.biomaterials.2015.08.045
- Miller JS, Stevens KR, Yang MT, et al. Rapid casting of patterned vascular networks for perfusable engineered three-dimensional tissues. Nat Mater. 2012;11(9):768-774. doi: 10.1038/nmat3357
- Kolesky DB, Homan KA, Skylar-Scott MA, Lewis JA. Three- dimensional bioprinting of thick vascularized tissues. Proc Natl Acad Sci USA. 2016;113(12):3179-3184. doi: 10.1073/pnas.1521342113
- Lee A, Hudson AR, Shiwarski DJ, et al. 3D bioprinting of collagen to rebuild components of the human heart. Science. 2019;365(6452):482-487. doi: 10.1126/science.aav9051
- Skylar-Scott MA, Uzel SG, Nam LL, et al. Biomanufacturing of organ-specific tissues with high cellular density and embedded vascular channels. Sci Adv. 2019;5(9):eaaw2459. doi: 10.1126/sciadv.aaw2459
- Murphy SV, Atala A. 3D bioprinting of tissues and organs. Nat Biotechnol. 2014;32(8):773-785. doi: 10.1038/nbt.2958
- Ozbolat IT, Peng W, Ozbolat V. Application areas of 3D bioprinting. Drug Discov Today. 2016;21(8):1257-1271. doi: 10.1016/j.drudis.2016.04.006
- Gungor-Ozkerim PS, Inci I, Zhang YS, Khademhosseini A, Dokmeci MR. Bioinks for 3D bioprinting: An overview. Biomater Sci. 2018;6(5):915-946. doi: 10.1039/c7bm00765e
- Pedde RD, Mirani B, Navaei A, et al. Emerging biofabrication strategies for engineering complex tissue constructs. Adv Mater. 2017;29(19):1606061. doi: 10.1002/adma.201606061
- Bernal PN, Bouwmeester M, Madrid-Wolff J, et al. Volumetric Bioprinting of Organoids and Optically Tuned Hydrogels to Build Liver-Like Metabolic Biofactories. Adv Mater. 2022;34(15):e2110054. doi: 10.1002/adma.202110054
- Aisenbrey EA, Murphy WL. Synthetic alternatives to Matrigel. Nat Rev Mater. 2020;5(7):539-551. doi: 10.1038/s41578-020-0199-8
- Zhang Z, Gao S, Hu YN, et al. Ti₃C₂Tx MXene composite 3D hydrogel potentiates mTOR signaling to promote the generation of functional hair cells in cochlea organoids. Adv Sci. 2022;9(32):e2203557. doi: 10.1002/advs.202203557
- Wang Y, Li H, Zhang J, Chen M, Pan Y, Lou X. 3D Bioprinting Inner Ear Organ of Corti Organoids Induce Hair Cell Regeneration. J Biomed Mater Res A. 2025;113(3):e37892. doi: 10.1002/jbm.a.37892
- Zhang J, Liu L, Shen R, Lou X. Construction of organ of Corti organoid to study the effects of berberine sulfate on damaged auditory cells. J Biomed Mater Res B Appl Biomater. 2024;112(7):e35439. doi: 10.1002/jbm.b.35439
- McNamara LE, McMurray RJ, Biggs MJ, Kantawong F, Oreffo RO, Dalby MJ. Nanotopographical control of stem cell differentiation. J Tissue Eng. 2010;1(1):120623. doi: 10.4061/2010/120623
- Daley WP, Peters SB, Larsen M. Extracellular matrix dynamics in development and regenerative medicine. J Cell Sci. 2008;121(Pt 3):255-264. doi: 10.1242/jcs.006064
- Zhu F, Nie G, Liu C. Engineered biomaterials in stem cell- based regenerative medicine. Life Med. 2023;2(4):lnad027. doi: 10.1093/lifemedi/lnad027
- Kleinman HK, Martin GR. Matrigel: Basement membrane matrix with biological activity. Semin Cancer Biol. 2005;15(5):378-386. doi: 10.1016/j.semcancer.2005.05.004
- Nie J, Koehler KR, Hashino E. Directed differentiation of mouse embryonic stem cells into inner ear sensory epithelia in 3D culture. In: Bhargava N, editor. Organ Regeneration: 3D Stem Cell Culture & Manipulation. New York, NY, USA: Humana Press; 2017:67-83. doi: 10.1007/978-1-4939-6949-4_6
- Schneeberger K, Spee B, Costa P, Sachs N, Clevers H, Malda J. Converging biofabrication and organoid technologies: The next frontier in hepatic and intestinal tissue engineering? Biofabrication. 2017;9(1):013001. doi: 10.1088/1758-5090/aa6121
- Asim S, Tabish TA, Liaqat U, Ozbolat IT, Rizwan M. Advances in Gelatin Bioinks to Optimize Bioprinted Cell Functions. Adv Healthc Mater. 2023;12(17):e2203148. doi: 10.1002/adhm.202203148
- Sakai Y, Yamato R, Onuma M, Kikuta T, Watanabe M, Nakayama T. Non-antigenic and low allergic gelatin produced by specific digestion with an enzyme-coupled matrix. Biol Pharm Bull. 1998;21(4):330-334. doi: 10.1248/bpb.21.330
- Li N, Zhang Z, Zhang L, et al. Establishment of a cochlear organoid platform for remodeling the extracellular matrix. ACS Nano. 2025;19(29):26542-26561. doi: 10.1021/acsnano.5c04897
- Gerchman E, Hilfer SR, Brown JW. Involvement of extracellular matrix in the formation of the inner ear. Dev Dyn. 1995;202(4):421-432. doi: 10.1002/aja.1002020411
- Fareez UNM, Naqvi SAA, Mahmud M, Temirel M. Computational Fluid Dynamics (CFD) Analysis of Bioprinting. Adv Healthc Mater. 2024;13(20):e2400643. doi: 10.1002/adhm.202400643
- Goto-Silva L, Ayad NME, Herzog IL, et al. Computational fluid dynamic analysis of physical forces playing a role in brain organoid cultures in two different multiplex platforms. BMC Dev Biol. 2019;19(1). doi: 10.1186/s12861-019-0183-y
- Kanda GN, Tsuzuki T, Terada M, et al. Robotic search for optimal cell culture in regenerative medicine. eLife. 2022;11. doi: 10.7554/eLife.77007
- Abdul L, Xu J, Sotra A, et al. D-CryptO: Deep learning-based analysis of colon organoid morphology from brightfield images. Lab Chip. 2022;22(21):4118-4128. doi: 10.1039/d2lc00596d
- Ou HC, Santos F, Raible DW, Simon JA, Rubel EW. Drug screening for hearing loss: Using the zebrafish lateral line to screen for drugs that prevent and cause hearing loss. Drug Discov Today. 2010;15(7-8):265-271. doi: 10.1016/j.drudis.2010.01.001
- Dekkers JF, Wiegerinck CL, De Jonge HR, et al. A functional CFTR assay using primary cystic fibrosis intestinal organoids. Nat Med. 2013;19(7):939-945. doi: 10.1038/nm.3201
- Liu Q, Zhang L, Zhu M-S, Wan G. High-throughput screening on cochlear organoids identifies VEGFR-MEK- TGFB1 signaling promoting hair cell reprogramming. Stem Cell Rep. 2021;16(9):2257-2273. doi: 10.1016/j.stemcr.2021.08.010
- Romano DR, Hashino E, Nelson RF. Deafness-in-a-dish: Modeling hereditary deafness with inner ear organoids. Hum Genet. 2022;141(3):347-362. doi: 10.1007/s00439-021-02325-9
- Cong L, Ran FA, Cox D, et al. Multiplex genome engineering using CRISPR/Cas systems. Science. 2013;339(6121):819- 823. doi: 10.1126/science.1231143
- Takahashi K, Tanabe K, Ohnuki M, et al. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell. 2007;131(5):861-872. doi: 10.1016/j.cell.2007.11.019
- Zhong C, Chen Z, Luo X, et al. Barhl1 is required for the differentiation of inner ear hair cell-like cells from mouse embryonic stem cells. Int J Biochem Cell Biol. 2018;96:79-89. doi: 10.1016/j.biocel.2018.01.013
- Tang P-C, Nie J, Lee J, et al. Defective Tmprss3-associated hair cell degeneration in inner ear organoids. Stem Cell Rep. 2019;13(1):147-162. doi: 10.1016/j.stemcr.2019.05.014
- White PM, Doetzlhofer A, Lee YS, Groves AK, Segil N. Mammalian cochlear supporting cells can divide and trans- differentiate into hair cells. Nature. 2006;441(7096):984-987. doi: 10.1038/nature04849
- Tao L, Yu HV, Llamas J, et al. Enhancer decommissioning imposes an epigenetic barrier to sensory hair cell regeneration. Dev Cell. 2021;56(17):2471-2485.e5. doi: 10.1016/j.devcel.2021.07.003
- Sun Q, Tan F, Wang X, et al. AAV-regulated Serpine2 overexpression promotes hair cell regeneration. Mol Ther Nucleic Acids. 2024;35(4):102396. doi: 10.1016/j.omtn.2024.102396
- Li X-J, Doetzlhofer A. LIN28B/let-7 control the ability of neonatal murine auditory supporting cells to generate hair cells through mTOR signaling. Proc Natl Acad Sci USA. 2020;117(36):22225-22236. doi: 10.1073/pnas.2000417117
- Urciuolo A, Giobbe GG, Dong Y, et al. Hydrogel-in-hydrogel live bioprinting for guidance and control of organoids and organotypic cultures. Nat Commun. 2023;14(1):3128. doi: 10.1038/s41467-023-37953-4
- DeForest CA, Tirrell DA. A photoreversible protein- patterning approach for guiding stem cell fate in three- dimensional gels. Nat Mater. 2015;14(5):523-531. doi: 10.1038/nmat421901

