Molecular subtype-guided precision immunotherapy with sintilimab for gastric cancer: Efficacy, heterogeneity, mechanisms, and clinical perspectives

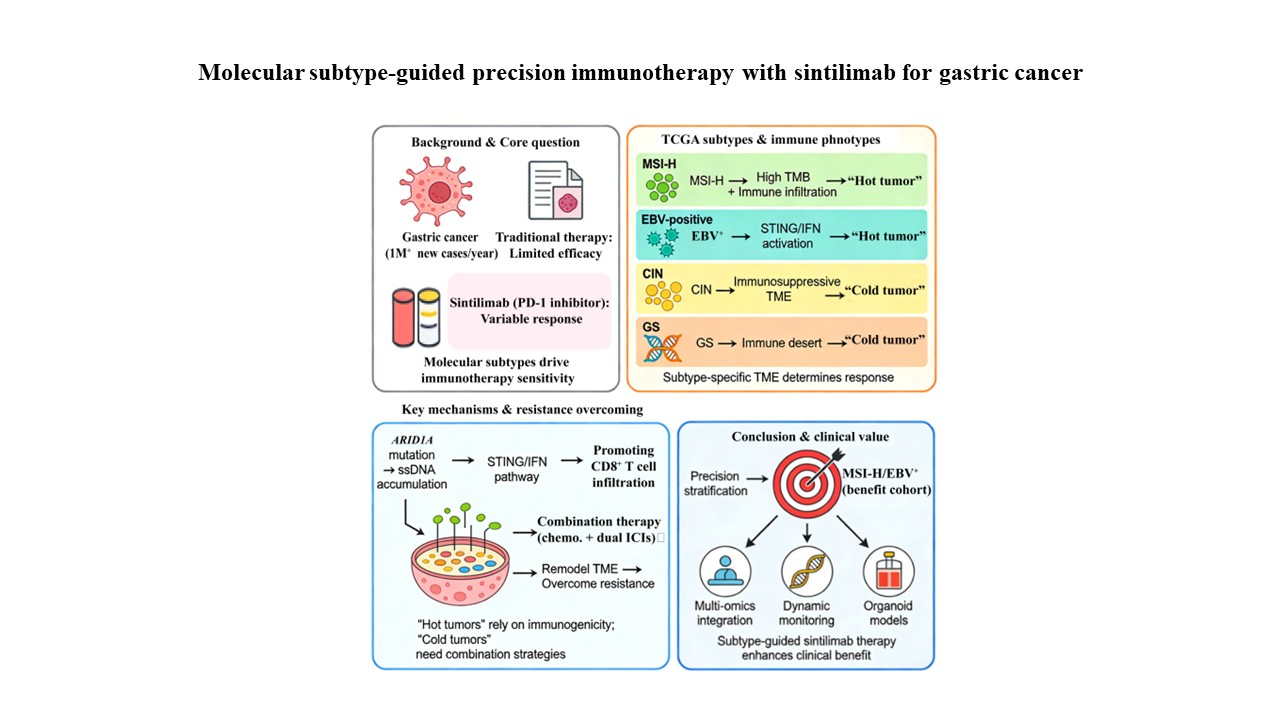
Despite the paradigm-shifting impact of immune checkpoint inhibitors in advanced gastric cancer, a significant proportion of patients exhibit primary or acquired resistance to therapy, largely driven by the disease’s profound molecular heterogeneity. This narrative review synthesizes current evidence on how The Cancer Genome Atlas–defined molecular subtypes–microsatellite instability-high (MSI-H), Epstein–Barr virus-positive (EBV+), chromosomally unstable (CIN), and genomically stable (GS)—orchestrate distinct tumor immune microenvironments that critically determine sensitivity to sintilimab, a programmed cell death protein 1 inhibitor widely adopted in China. We highlight that MSI-H and EBV+ tumors typically foster an immunologically “hot” milieu, characterized by high tumor mutational burden, abundant CD8+ T-cell infiltration, and interferon-gamma signaling, rendering them highly responsive to sintilimab monotherapy or dual checkpoint blockade. In contrast, CIN and GS subtypes often present as “cold” or immune-excluded, necessitating rational combinations with chemotherapy or anti-angiogenic agents such as apatinib. However, we also critically appraise contradictory data, noting that not all MSI-H tumors respond equally and that the “cold” versus “hot” dichotomy represents a spectrum with significant intratumoral heterogeneity. Notably, loss-of-function mutations in AT-rich interaction domain 1A serve as a pivotal link between genomic instability and immune activation, potentially enhancing sintilimab efficacy by driving single-stranded DNA accumulation, stimulating the stimulator of interferon genes pathway, and driving subsequent type I interferon production. While landmark trials such as ORIENT-16 have validated sintilimab-based regimens, challenges persist in managing organ-specific metastases and overcoming dynamic mechanisms of resistance. We propose that integrating comprehensive molecular profiling into routine clinical practice is essential for precision immunotherapy, and future efforts should focus on validating novel biomarkers and leveraging multi-omics platforms to guide individualized treatment strategies.
- Xu J, Jiang H, Pan Y, et al. Sintilimab Plus Chemotherapy for Unresectable Gastric or Gastroesophageal Junction Cancer: The ORIENT-16 Randomized Clinical Trial. JAMA. 2023;330(21):2064-2074. doi: 10.1001/jama.2023.19918
- Zhang Z, Wu C, Hu X, Zhang L, Dong X. Mechanisms and therapeutic strategies for immunotherapy resistance in gastric cancer. Cancer Cell Int. 2025;26(1). doi: 10.1186/s12935-025-04110-4
- Patel AK, Sethi NS, Park H. Gastric Cancer: A Review. JAMA. 2026;335(5):439-450. doi: 10.1001/jama.2025.20034
- Zeng Y, Jin RU. Molecular pathogenesis, targeted therapies, and future perspectives for gastric cancer. Semin Cancer Biol. 2022;86(Pt 3):566-582. doi: 10.1016/j.semcancer.2021.12.004
- Qi H, Ma X, Ma Y, Jia L, Liu K, Wang H. Mechanisms of HIF1A-mediated immune evasion in gastric cancer and the impact on therapy resistance. Cell Biol Toxicol. 2024;40(1):87. doi: 10.1007/s10565-024-09917-x
- Chen W, Zhang L, Gao M, et al. Role of tertiary lymphoid structures and B cells in clinical immunotherapy of gastric cancer. Front Immunol. 2024;15:1519034. doi: 10.3389/fimmu.2024.1519034
- Luo D, Zhou J, Ruan S, et al. Overcoming immunotherapy resistance in gastric cancer: insights into mechanisms and emerging strategies. Cell Death Dis. 2025;16(1):75. doi: 10.1038/s41419-025-07385-7
- Li N, Li Z, Fu Q, et al. Efficacy and safety of neoadjuvant sintilimab in combination with FLOT chemotherapy in patients with HER2-negative locally advanced gastric or gastroesophageal junction adenocarcinoma: an investigator-initiated, single-arm, open-label, phase II study. Int J Surg. 2024;110(4):2071-2084. doi: 10.1097/JS9.0000000000001119
- Jiang H, Yu X, Li N, et al. Efficacy and safety of neoadjuvant sintilimab, oxaliplatin and capecitabine in patients with locally advanced, resectable gastric or gastroesophageal junction adenocarcinoma: early results of a phase 2 study. J Immunother Cancer. 2022;10(3):e003635. doi: 10.1136/jitc-2021-003635
- Nie Y, Zhao W, Lu L, Zhou F. Predictive biomarkers and new developments of immunotherapy in gastric cancer: a 2023 update. Am J Cancer Res. 2023;13(7):3169-3184.
- Qian X, Hu W, Yan J. Nano-Chemotherapy synergize with immune checkpoint inhibitor- A better option. Front Immunol. 2022;13:963533. doi: 10.3389/fimmu.2022.963533
- Bos J, Groen-van Schooten TS, Brugman CP, Jamaludin FS, van Laarhoven H, Derks S. The tumor immune composition of mismatch repair deficient and Epstein-Barr virus-positive gastric cancer: A systematic review. Cancer Treat Rev. 2024;127:102737. doi: 10.1016/j.ctrv.2024.102737
- Zhang C, Zhang X, Liang J, et al. Subacute cutaneous lupus erythematosus triggered by sintilimab: a case report. Front Immunol. 2025;16:1544312. doi: 10.3389/fimmu.2025.1544312
- Wei J, Lu X, Liu Q, et al. Neoadjuvant sintilimab in combination with concurrent chemoradiotherapy for locally advanced gastric or gastroesophageal junction adenocarcinoma: a single-arm phase 2 trial. Nat Commun. 2023;14(1):4904. doi: 10.1038/s41467-023-40480-x
- Liu J, Liu D, Hu G, et al. Circulating memory PD-1(+) CD8(+) T cells and PD-1(+)CD8(+)T/PD-1(+)CD4(+)T cell ratio predict response and outcome to immunotherapy in advanced gastric cancer patients. Cancer Cell Int. 2023;23(1):274. doi: 10.1186/s12935-023-03137-9
- Lin GT, Yan C, Li LJ, et al. Combining Apatinib and Oxaliplatin Remodels the Immunosuppressive Tumor Microenvironment and Sensitizes Desert-Type Gastric Cancer to Immunotherapy. Cancer Res. 2025;85(11):2117- 2133. doi: 10.1158/0008-5472.CAN-24-2697
- Cui K, Yao S, Liu B, et al. A novel high-risk subpopulation identified by CTSL and ZBTB7B in gastric cancer. Br J Cancer. 2022;127(8):1450-1460. doi: 10.1038/s41416-022-01936-x
- Ma F, Ren M, Li Z, et al. ARID1A is a coactivator of STAT5 that contributes to CD8(+) T cell dysfunction and anti-PD-1 resistance in gastric cancer. Pharmacol Res. 2024;210:107499. doi: 10.1016/j.phrs.2024.107499
- Xu C, Huang KK, Law JH, et al. Comprehensive molecular phenotyping of ARID1A-deficient gastric cancer reveals pervasive epigenomic reprogramming and therapeutic opportunities. Gut. 2023;72(9):1651-1663. doi: 10.1136/gutjnl-2022-328332
- Niu Y, Ding C, Wang Q, et al. TRIM6 ablation reverses ICB resistance in MSS gastric cancer by unleashing cGAS-STING-dependent antitumor immunity. J Exp Clin Cancer Res. 2025;44(1):242. doi: 10.1186/s13046-025-03513-5
- Khalique S, Nash S, Natrajan R. Definitive study shows no association between ARID1A mutation status and clinical outcome in endometriosis-related ovarian cancers‡. J Pathol. 2022;258(1):1-3. doi: 10.1002/path.5973
- Chen JY, Ke TW, Chiang SF, et al. CHK1 inhibition increases the therapeutic response to radiotherapy via antitumor immunity in ARID1A-deficient colorectal cancer. Cell Death Dis. 2025;16(1):584. doi: 10.1038/s41419-025-07912-6
- Yu ZC, Li T, Tully E, et al. Temozolomide Sensitizes ARID1A-Mutated Cancers to PARP Inhibitors. Cancer Res. 2023;83(16):2750-2762. doi: 10.1158/0008-5472.CAN-22-3646
- Maxwell MB, Hom-Tedla MS, Yi J, et al. ARID1A suppresses R-loop-mediated STING-type I interferon pathway activation of anti-tumor immunity. Cell. 2024;187(13):3390- 3408.e19. doi: 10.1016/j.cell.2024.04.025
- Zhou X, Yang J, Lu Y, et al. Relationships of tumor differentiation and immune infiltration in gastric cancers revealed by single-cell RNA-seq analyses. Cell Mol Life Sci. 2023;80(2):57. doi: 10.1007/s00018-023-04702-1
- Mao D, Zhou Z, Chen H, et al. Pleckstrin-2 promotes tumour immune escape from NK cells by activating the MT1-MMP-MICA signalling axis in gastric cancer. Cancer Lett. 2023;572:216351. doi: 10.1016/j.canlet.2023.216351
- Yong X, Mu D, Ni H, et al. Regulation of the CD8(+) T cell and PDL1/PD1 axis in gastric cancer: Unraveling the molecular landscape. Crit Rev Oncol Hematol. 2025;212:104750. doi: 10.1016/j.critrevonc.2025.104750
- Zhang Y, Huang Y, Yu D, et al. Demethylzeylasteral induces PD-L1 ubiquitin-proteasome degradation and promotes antitumor immunity via targeting USP22. Acta Pharm Sin B. 2024;14(10):4312-4328. doi: 10.1016/j.apsb.2024.08.004
- Lu C, Liu Z, Klement JD, et al. WDR5-H3K4me3 epigenetic axis regulates OPN expression to compensate PD-L1 function to promote pancreatic cancer immune escape. J Immunother Cancer. 2021;9(7):e002624. doi: 10.1136/jitc-2021-002624
- Liu X, Zhou J, Wu H, et al. Fibrotic immune microenvironment remodeling mediates superior anti-tumor efficacy of a nano-PD-L1 trap in hepatocellular carcinoma. Mol Ther. 2023;31(1):119-133. doi: 10.1016/j.ymthe.2022.09.012
- Wang H, Wu J, Ling R, et al. Fibroblast-derived LPP as a biomarker for treatment response and therapeutic target in gastric cancer. Mol Ther Oncolytics. 2022;24:547-560. doi: 10.1016/j.omto.2022.01.008
- Zhang Y, Yang Y, Chen Y, et al. PD-L1: Biological mechanism, function, and immunotherapy in gastric cancer. Front Immunol. 2022;13:1060497. doi: 10.3389/fimmu.2022.1060497
- Luo H, Wu J, Yan Y, et al. Mechanisms and therapeutic strategies to reveal and overcome T-cell dysfunction in gastric cancer: translation from basic research to clinical application. Front Immunol. 2025;16:1681539. doi: 10.3389/fimmu.2025.1681539
- Liu S, Liu H, Song X, et al. Adoptive CD8(+)T-cell grafted with liposomal immunotherapy drugs to counteract the immune suppressive tumor microenvironment and enhance therapy for melanoma. Nanoscale. 2021;13(37):15789- 15803. doi: 10.1039/D1NR04036G
- Yang SF, Weng MT, Liang JD, et al. Neoantigen vaccination augments antitumor effects of anti-PD-1 on mouse hepatocellular carcinoma. Cancer Lett. 2023;563:216192. doi: 10.1016/j.canlet.2023.216192
- Huang M, Xiong D, Pan J, et al. Prevention of Tumor Growth and Dissemination by In Situ Vaccination with Mitochondria-Targeted Atovaquone. Adv Sci (Weinh). 2022;9(12):e2101267. doi: 10.1002/advs.202101267
- Hagenstein J, Burkhardt S, Sprezyna P, Tasika E, Tiegs G, Diehl L. CD44 expression on murine hepatic stellate cells promotes the induction of monocytic and polymorphonuclear myeloid-derived suppressor cells. J Leukoc Biol. 2024;116(1):177-185. doi: 10.1093/jleuko/qiae053
- Zhang W, Li X, Jiang M, et al. SOCS3 deficiency-dependent autophagy repression promotes the survival of early-stage myeloid-derived suppressor cells in breast cancer by activating the Wnt/mTOR pathway. J Leukoc Biol. 2023;113(5):445-460. doi: 10.1093/jleuko/qiad020
- Liu M, Wei F, Wang J, et al. Myeloid-derived suppressor cells regulate the immunosuppressive functions of PD-1(-) PD-L1(+) Bregs through PD-L1/PI3K/AKT/NF-kappaB axis in breast cancer. Cell Death Dis. 2021;12(5):465. doi: 10.1038/s41419-021-03745-1
- He Y, Hong Q, Chen S, Zhou J, Qiu S. Reprogramming tumor-associated macrophages in gastric cancer: a pathway to enhanced immunotherapy. Front Immunol. 2025;16:1558091. doi: 10.3389/fimmu.2025.1558091
- Wang W, Gao Y, Xu J, et al. A NRF2 Regulated and the Immunosuppressive Microenvironment Reversed Nanoplatform for Cholangiocarcinoma Photodynamic-Gas Therapy. Adv Sci (Weinh). 2024;11(14):e2307143. doi: 10.1002/advs.202307143
- Liu H, Huang K, Zhang H, Liu X, Jiang H, Wang X. Photo- Driven In Situ Solidification of Whole Cells through Inhibition of Trogocytosis for Immunotherapy. Research (Wash D C). 2024;7:0318. doi: 10.34133/research.0318
- Tang Y, Cai Q, Tian Z, Chen W, Tang H. Crosstalk between Gut Microbiota and Cancer Immunotherapy: Present Investigations and Future Perspective. Research (Wash D C). 2025;8:0600. doi: 10.34133/research.0600
- Lubiński J, Lener MR, Marciniak W, et al. Serum Essential Elements and Survival after Cancer Diagnosis. Nutrients. 2023;15(11):2611. doi: 10.3390/nu15112611
- Eiman L, Moazzam K, Anjum S, Kausar H, Sharif E, Ibrahim WN. Gut dysbiosis in cancer immunotherapy: microbiota-mediated resistance and emerging treatments. Front Immunol. 2025;16:1575452. doi: 10.3389/fimmu.2025.1575452
- Zhao J, Li X, Sun X, et al. Combination of cadonilimab (PD-1/CTLA-4 bispecific antibody) and apatinib as salvage therapy achieves partial response in MSI-H advanced gastric cancer: a case report. Front Immunol. 2025;16:1533700. doi: 10.3389/fimmu.2025.1533700
- Liu Z, Liu A, Li M, Xiang J, Yu G, Sun P. Efficacy and safety of sintilimab combined with trastuzumab and chemotherapy in HER2-positive advanced gastric or gastroesophageal junction cancer. Front Immunol. 2025;16:1545304. doi: 10.3389/fimmu.2025.1545304
- Mao C, Xiong A, Qian J, et al. Dual inhibition of LAG-3 and PD-1 with IBI110 and sintilimab in advanced solid tumors: the first-in-human phase Ia/Ib study. J Hematol Oncol. 2024;17(1):132. doi: 10.1186/s13045-024-01651-5
- Randon G, Montroni I, Pietrantonio F. Bringing immunotherapy to clinical practice in dMMR/MSI-high colon cancer. Cancer Cell. 2025;43(10):1789-1791. doi: 10.1016/j.ccell.2025.08.010
- Liu R, Ji Z, Wang X, et al. Regorafenib plus sintilimab as a salvage treatment for microsatellite stable metastatic colorectal cancer: a single-arm, open-label, phase II clinical trial. Nat Commun. 2025;16(1):1481. doi: 10.1038/s41467-025-56748-3
- Cao LL, Lu H, Soutto M, et al. Multivalent tyrosine kinase inhibition promotes T cell recruitment to immune-desert gastric cancers by restricting epithelial-mesenchymal transition via tumour-intrinsic IFN-gamma signalling. Gut. 2023;72(11):2038-2050. doi: 10.1136/gutjnl-2022-329134
- Zhang X, Wang WB, Cai XY, et al. MNDA promotes immunosuppression in microsatellite instability-high colorectal cancer by facilitating PMN-MDSC infiltration via H3K18 lactylation. J Transl Med. 2025;23(1):1049. doi: 10.1186/s12967-025-07097-8
- Huang W, Zhang Y, Chen S, et al. Personalized immune subtypes based on machine learning predict response to checkpoint blockade in gastric cancer. Brief Bioinform. 2023;24(1):bbac554. doi: 10.1093/bib/bbac554
- Zu M, Hao X, Ning J, et al. Patient-derived organoid culture of gastric cancer for disease modeling and drug sensitivity testing. Biomed Pharmacother. 2023;163:114751. doi: 10.1016/j.biopha.2023.114751
- Wang J, Kunzke T, Prade VM, et al. Spatial Metabolomics Identifies Distinct Tumor-Specific Subtypes in Gastric Cancer Patients. Clin Cancer Res. 2022;28(13):2865-2877. doi: 10.1158/1078-0432.CCR-21-4383
- Joshi SS, Badgwell BD. Current treatment and recent progress in gastric cancer. CA Cancer J Clin. 2021;71(3):264- 279. doi: 10.3322/caac.21657
- Kwon M, An M, Klempner SJ, et al. Determinants of Response and Intrinsic Resistance to PD-1 Blockade in Microsatellite Instability-High Gastric Cancer. Cancer Discov. 2021;11(9):2168-2185. doi: 10.1158/2159-8290.CD-21-0219
- Zhang J, Dong Y, Yu S, et al. IL-4/IL-4R axis signaling drives resistance to immunotherapy by inducing the upregulation of Fcgamma receptor IIB in M2 macrophages. Cell Death Dis. 2024;15(7):500. doi: 10.1038/s41419-024-06875-4
59. Wang Y, Jin RU, Xu J, et al. Harnessing technologies to unravel gastric cancer heterogeneity. Trends Cancer. 2025;11(8):753-769. doi: 10.1016/j.trecan.2025.04.011

